Molecular typing methods can help clinicians and public health practitioners to identify clusters of recently acquired TB.1 MTB isolates from epidemiologically linked patients show identical or very closely related genotypic patterns because of the recency of infection transmission.2 The NSW Mycobacterium Reference Laboratory at the Institute of Clinical Pathology and Medical Research, Sydney West Area Health Service, began routine genotyping of MTB isolates in 2003. We describe the application of traditional epidemiological investigation combined with molecular typing to characterise a TB cluster in the Hunter Area.
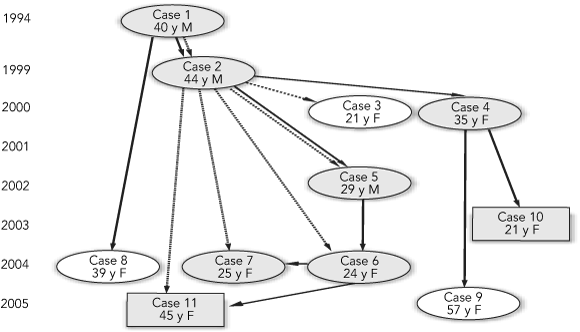
A cluster was first suspected in February 2005, when contact-tracing interviews with an inpatient with pulmonary TB (Case 6 in Box 1) identified a link to an informal music recording studio shared with five other local TB cases whose onset of illness dated back to as early as 1994. The members of this group were younger than typical notified cases and were all born in Australia or New Zealand. We conducted an investigation to determine the extent of the cluster, to identify epidemiological links and risk factors, and to ensure an appropriate public health response.
Learning points
Clinicians should remain vigilant for tuberculosis (TB) in non-typical groups.
The diagnosis of pulmonary TB in a young Australian-born person is an event that warrants detailed investigation, especially if the person is from a suspected socioeconomically vulnerable group.
Genotyping of isolates is useful for detection and investigation of TB clusters.
All MTB isolates were initially genotyped at the NSW Mycobacterium Reference Labor-atory using two nucleic acid amplification-based methods — mycobacterial interspersed repetitive unit (MIRU) typing and spacer oligonucleotide typing (“spoligotyping”) — conducted according to standard protocols.3,4 Clinical isolates with matching MIRU types and spoligotypes were then examined by IS6110 restriction fragment length polymorphism (RFLP) analysis, regarded as the current gold standard for genotyping MTB isolates.5 Comparison of IS6110 RFLP fingerprints was performed using BioNumerics edition 3.0 software (Applied Maths, Koutrai, Belgium).
Genotyping helped to confirm nine cluster cases. All were of the same MIRU profile (223125143312), and spoligotyping showed them to be from the T1 (Haarlem) family of MTB (SpolDB3 classification, probability of 0.99). The RFLP pattern was indistinguishable for seven cases, had one additional band for Case 8, and was not available for Case 5, as insufficient DNA could be recovered from the culture, which had been stored since 2002 (Box 2). All cluster isolates were fully sensitive to first-line antituberculosis therapy.
Clinicians should maintain a high index of suspicion for pulmonary TB in a person presenting with a productive cough lasting more than 3 weeks, weight loss, haemoptysis, night sweats and chest pain, even if the suspected case is atypical (ie, not overseas-born or elderly). This is particularly important if the individual comes from a vulnerable population subgroup.6
In the cluster we examined, epidemiological investigation revealed complex social networks between cases, with a likelihood of several “waves” of transmission between 1994 and 2005. Transmission probably occurred at a small, poorly ventilated recording studio and in social and workplace settings. Although the contribution of recreational marijuana use to the spread of TB is not clear in this cluster, shared smoking equipment has been identified as a potential source of infection in previous clusters.7
Genotyping played a pivotal role in our investigation, and the use of multiple typing methods improved cluster discrimination.8 Confirmed cluster cases were from the relatively uncommon T1 family, identified in fewer than 5% of NSW isolates, increasing the likelihood that the cases were related. Genotyping resulted in the identification of additional cluster cases and the timely exclusion of several young Australian-born cases, thus allowing investigators to focus their resources on identifying epidemiological links between cluster cases. Two cases with isolates whose genotypes were very similar to the cluster strain (with a single band difference in the RFLP pattern) were excluded because no epidemiological links could be identified. However, the possibility that they were linked indirectly to the cluster by currently unidentified cases cannot be excluded.
Routine genotyping has the potential to detect unsuspected clusters, and would likely have identified this group earlier if testing had been available before 2003. The delay in identifying the cluster reinforces the limitations of relying solely on traditional contact tracing, particularly in hard-to-reach populations.9 Molecular epidemiological studies have demonstrated that as few as 5%–10% of clustered cases are identified through routine investigation of close contacts.2,10,11 Factors contributing to this low success rate include the importance of casual contact in transmission and the fact that cases do not necessarily disclose all contacts.
In low-incidence countries such as Australia, secondary TB transmission should ideally be limited by rapid and complete contact investigation. In practice, however, such action is hampered by diagnostic delays and the occurrence of cases in hard-to-reach itinerant groups, in which transmission often occurs beyond recognised “concentric circles” of close contacts.12 A finding of genotypic clustering of MTB isolates could be used as a trigger for a second targeted interview designed to unravel undetected routes of transmission. Such a two-stage contact investigation would add “epidemiologic value”.2,10,13
- Tony D Merritt1
- Vitali Sintchenko2
- Peter Jelfs2
- Margaret Worthing1
- Brian Robinson1
- David N Durrheim1
- Gwendolyn L Gilbert2
- 1 Hunter New England Population Health, Newcastle, NSW.
- 2 Centre for Infectious Diseases and Microbiology — Public Health, Sydney West Area Health Services, Sydney, NSW.
Genotyping of MTB isolates was funded by the NSW Department of Health. Infrastructure support to Hunter Area staff was provided by the Hunter Medical Research Institute.
None identified.
- 1. Dye C, Williams BG, Espinal MA, Raviglione MC. Erasing the world’s slow stain: strategies to beat multidrug-resistant tuberculosis. Science 2002; 295: 2042-2046.
- 2. Van Deutekom H, Hoijng SP, de Haas PEW, et al. Clustered tuberculosis cases: do they represent recent transmission and can they be detected earlier? Am J Respir Crit Care Med 2004; 169: 806-810.
- 3. Supply P, Lesjean S, Savine E, et al. Automated high-throughput genotyping for study of global epidemiology of Mycobacterium tuberculosis based on mycobacterial interspersed repetitive units. J Clin Microbiol 2001; 39: 3563-3571.
- 4. Kamerbeek J, Schouls L, Kolk A, et al. Simultaneous detection and strain differentiation of Mycobacterium tuberculosis for diagnosis and epidemiology. J Clin Microbiol 1997; 35: 907-914.
- 5. Van Embden JD, Cave MD, Crawford JT, et al. Strain identification of Mycobacterium tuberculosis by DNA fingerprinting: recommendations for a standardized methodology. J Clin Microbiol 1993; 31: 406-409.
- 6. Hopewell PC, Pai M. Tuberculosis, vulnerability and access to quality care. JAMA 2005; 293: 2790-2793.
- 7. Munckhof WJ, Konstantinos A, Wamsley M, et al. A cluster of tuberculosis associated with use of a marijuana water pipe. Int J Tuberc Lung Dis 2003; 7: 860-865.
- 8. Nguyen LN, Gilbert GL, Marks GB. Molecular epidemiology of tuberculosis and recent developments in understanding the epidemiology of tuberculosis. Respirology 2004; 9: 313-319.
- 9. Malakmadze N, Gonzalez I, Oemig T, et al. Unsuspected recent transmission of tuberculosis among high-risk groups: implications of universal tuberculosis genotyping in its detection. Clin Infect Dis 2005; 40: 366-373.
- 10. Daley CL. Tuberculosis contact investigations. Please don’t fail me now. Am J Respir Crit Care Med 2004; 169: 779-781.
- 11. Small PM, Hopewell PHC, Singh SP, et al. The epidemiology of tuberculosis in San Francisco: a population-based study using conventional and molecular methods. N Engl J Med 1994; 330: 1703-1709.
- 12. Lambregts-van Weezenbeek CS, Sebek MM, van Gerven PJ, et al. Tuberculosis contact investigation and DNA fingerprint surveillance in The Netherlands: 6 years’ experience with nation-wide cluster feedback and cluster monitoring. Int J Tuberc Lung Dis 2003; 7 (12 Suppl 3): S463-S470.
- 13. Sintchenko V, Gilbert GL. Utility of genotyping of Mycobacterium tuberculosis in the contact investigation: a decision analysis. Tuberculosis (Edinb) 2006 Dec 8; [Epub ahead of print].






Abstract
Objective: To characterise a pulmonary tuberculosis (TB) cluster in the Hunter Area of New South Wales using a combination of traditional epidemiological methods and molecular typing.
Design, setting and participants: Review of all notifications of TB in the Hunter Area between January 1994 and June 2005, with a detailed analysis of cases among people born in Australia or New Zealand.
Main outcome measures: Comparison of genotypes of Mycobacterium tuberculosis isolates; extent of TB cluster.
Results: Over the period studied, there were 72 TB notifications among people born in Australia or New Zealand. Genotypic testing was available for 20 of these cases, of which nine were confirmed to be part of a cluster. Two further cases for which genotyping was not available were epidemiologically linked to the cluster and regarded as probable cluster cases. Members of the cluster were relatively young (median age at diagnosis, 35 years; range, 21–57 years), and eight were women. Over the same period, there were 83 TB notifications among people born overseas, the majority being from Asia (47%) or central and eastern Europe (24%) (median age, 54 years; range, 9–63 years).
Conclusion: Clinicians should maintain a high index of suspicion for pulmonary TB in a person presenting with a productive cough lasting more than 3 weeks, weight loss, haemoptysis, night sweats and chest pain, even if the person is not overseas-born or elderly. A comprehensive tuberculosis genotyping network at regional and national level in Australia could help identify clusters resulting from recent transmission.