Atrial fibrillation (AF) is the most common sustained arrhythmia, and is associated with increased cardiovascular morbidity and mortality, and preventable stroke. The incidence and prevalence of AF rise with age, with a prevalence of 8% in people older than 80 years.1 In addition, the age-adjusted incidence in the Framingham study increased significantly from the 1960s to the 1980s, and has increased further from 1980 to 2000.2 This may be due in part to the population increase in obesity and obstructive sleep apnoea.3
Here we summarise updated concepts in the management of AF as recently published in the revised guidelines of the American College of Cardiology, American Heart Association and European Society of Cardiology (ACC/AHA/ESC).4
Use of angiotensin-converting enzyme inhibitors, angiotensin II receptor blockers,5 and statins may reduce the incidence of AF, as may fish oils, which alter atrial membrane composition.4 After cardiac surgery, where postoperative AF occurs in about 25% of patients, pretreatment with β-blockers, sotalol, amiodarone, and statins has been shown to reduce the incidence.4,6,7
Objectives in the management of AF are to:
identify and treat associated or causative factors — this may abort the arrhythmia;
decide on “rate control or rhythm control”, and implement treatment either to control heart rate or to achieve and maintain sinus rhythm; and
prevent thromboembolism, balancing the risk of stroke against the risk of bleeding on warfarin.
AF is most commonly caused by hypertension, ischaemic heart disease, heart failure, valvular heart disease, and thyrotoxicosis, but other treatable causes exist (Box 1). The minimal clinical evaluation comprises a history, physical examination, electrocardiography, transthoracic echocardiography, and blood tests of thyroid, renal and hepatic function. Additional tests may be required to exclude other conditions according to clinical suspicion.
Most patients with AF require control of the heart rate for symptomatic relief, and to prevent tachycardia-induced cardiomyopathy. Digoxin is no longer the drug of first choice for rate control (I/C; see Box 2 for key to evidence levels);4 β-blockers are the most effective agents for monotherapy, followed by verapamil and diltiazem,8 as these drugs control both exertional and resting heart rate (I/B).
The decision to attempt cardioversion and maintain sinus rhythm, rather than just control heart rate, depends on the long-term frequency and hazards of AF, and risks of cardioversion and antiarrhythmic therapy. Older patients (> 65 years) with recurrent persistent AF at high risk of stroke (CHADS2 score ≥ 1, see “Stroke risk stratification” below) have similar outcomes (stroke or mortality) with either rate or rhythm control, and a trend towards fewer hospitalisations for those managed with rate control.9 Rate control is more suitable for older patients with asymptomatic persistent or permanent AF, whereas young patients with highly symptomatic paroxysmal AF may require rhythm control.
Sinus rhythm probably does confer a benefit, particularly for patients with heart failure. However, because drugs are relatively ineffective at long-term maintenance of sinus rhythm (60% in sinus rhythm at 1 year on amiodarone and 40% with sotalol),9 and have significant cardiac and extra-cardiac toxicities, including ventricular tachycardia and pulmonary fibrosis, this benefit is negated. A rhythm control strategy is therefore associated with frequent AF recurrences, which may be asymptomatic, and places the patient at risk of stroke unless continuously treated with effective antithrombotic prophylaxis.
Electrical cardioversion remains the mainstay for conversion of persistent AF to sinus rhythm, but there are significant issues of anticoagulation, as discussed below. Cardioversion can also be achieved with amiodarone and flecainide, but up to two-thirds of paroxysmal episodes revert spontaneously within 24 hours. Use of flecainide as a “pill in the pocket” (an oral dose taken at the onset of symptoms) is now endorsed for infrequent highly symptomatic AF (IIb/C).4 However, concomitant use of atrio-ventricular nodal slowing drugs is advised, and first use recommended in hospital while monitored. Flecainide is also contraindicated with structural heart disease.
This procedure involves application of radiofrequency ablation to electrically isolate the pulmonary veins, with or without other lesions to attempt cure of AF. Success rates are variable, but approximate 75%, although this may require multiple procedures.10 If AF recurs, episodes may be less symptomatic or asymptomatic. Catheter ablation is associated with a 3%–6% risk of major complications, including pulmonary vein stenosis, thromboembolism, and the rare (0.6%) but often fatal atrio-oesophageal fistula. It is safest and most successful in patients younger than 70 years with paroxysmal AF for whom anti-arrhythmic therapy has been ineffective and who have a left atrial diameter < 5 cm and left ventricular ejection fraction > 40%. Greater understanding of the mechanisms underlying AF may increase the efficacy of this procedure, but currently ablation is not applicable to the large numbers of elderly patients who develop this arrhythmia.
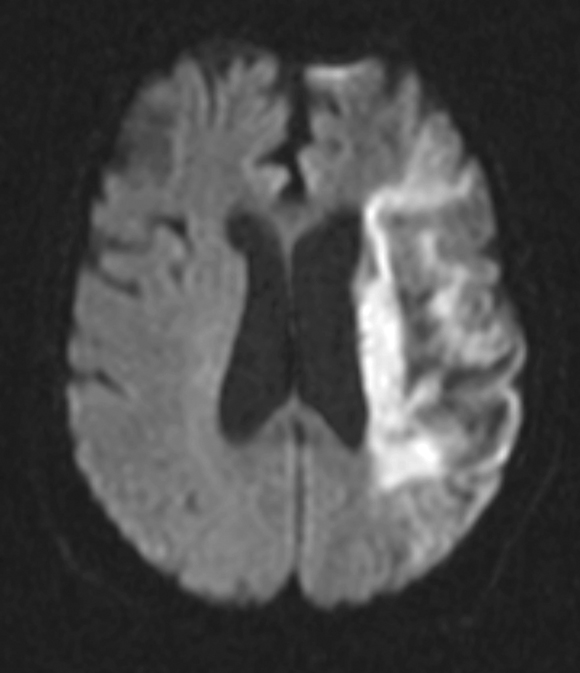
AF predisposes to the formation of blood clots within the left atrium (LA) and particularly the left atrial appendage (LAA) (Box 3), and these may embolise to the systemic circulation. Consequently, AF is an independent risk factor for cardioembolic ischaemic stroke. Although most strokes in people with AF are embolic from the LA/LAA, about a quarter may originate elsewhere — from the left ventricle, heart valves, and extracranial and intracranial arteries. Cardioembolic strokes in patients with AF are typically larger, associated with higher early mortality, and occur in older patients compared with strokes in patients with sinus rhythm (Box 4).
Optimal thromboprophylaxis for patients with AF is individualised and requires an assessment of:
the patient’s risk of thromboembolism, particularly ischaemic stroke;
the potential benefits of anticoagulant compared with antiplatelet therapy;
the competing risk of major haemorrhage with antithrombotic therapy; and
the patient’s preference.
Among patients with non-valvular AF, the risk of ischaemic stroke averages 5% per year (range, 3%–8%), about 3–5 times that of people in sinus rhythm.11 However, the risk is greatly influenced by individual patient characteristics (Box 5). For patients with lone AF (younger than 60 years with no clinical history or echocardiographic signs of cardiopulmonary disease), the cumulative risk of stroke over 15 years is very low (about 1.3%).12 For patients with non-valvular AF, the strongest independent predictor of stroke is prior stroke or transient ischaemic attack (TIA) (relative risk [RR], 1.9–3.7), which increases the annual risk of subsequent stroke to about 12% per year with no antithrombotic therapy, and about 10% per year with aspirin.13 Increasing age increases the annual risk of stroke from 1.5% in ages 50–59 years to 23% in those aged 80–89 years.11 Other independent risk factors for ischaemic stroke are heart failure with systolic dysfunction, hypertension, and diabetes mellitus (Box 5).14
The revised guidelines promote the simple CHADS2 score to estimate stroke risk. This is an acronym for an index that gives 1 point for Congestive heart failure, Hypertension, Age ≥ 75 years, and Diabetes mellitus, and 2 points for prior Stroke/TIA. This score successfully distinguishes between patients at high risk and those at low risk of stroke ().15-17
Adjusted-dose warfarin reduces the relative risk of stroke by 62%,18 with absolute risk reductions of 2.7% per year for primary prevention and 8.4% per year for secondary prevention. Major extracranial bleeding is increased by warfarin therapy (absolute risk increase, 0.3% per year).18 Aspirin is less efficacious, with only 22% relative risk reduction, and absolute risk reductions of 1.5% and 2.5% per year for primary and secondary prevention, respectively.19 Aspirin appears to prevent non-disabling non-cardioembolic ischaemic strokes more than disabling cardioembolic strokes.19 Treating 1000 patients with AF for 1 year with oral anticoagulant rather than aspirin would prevent 23 ischaemic strokes while causing 9 additional major bleeds.
Warfarin (INR, 2–3) is superior to clopidogrel 75 mg plus aspirin 75–100 mg for prevention of vascular events in patients with AF at high risk of stroke (CHADS2 score ≥ 1), reducing the annual risk of stroke from 5.6% with dual antiplatelet therapy to 3.9% with warfarin (RR, 0.69; 95% CI, 0.57–0.85).20 However, these results apply primarily to patients previously exposed to warfarin therapy. For patients new to both treatments, post hoc analysis found the benefits of warfarin were not well defined. In routine practice, the superiority of warfarin could be negated by poor INR control, whereas the major bleeding rate associated with clopidogrel plus aspirin may approximate that of warfarin.
Low rates of major haemorrhage in the trials are unlikely to reflect common practice. Predictors of major bleeding include increasing age, hypertension, elevated INR, and previous cerebral ischaemia,21,22 as well as combining antiplatelet agents with anticoagulation and use of dual antiplatelet therapy.23
Adjusted odds ratios associating drug use with gastrointestinal bleeding are between 1 and 2 for low dose aspirin, clopidogrel and warfarin alone, but corresponding figures for aspirin in combination with clopidogrel or warfarin are 7.4 and 5.3, respectively.24
Keeping the INR < 3, lowering blood pressure to below 135 mmHg systolic, and avoiding antiplatelet drugs decreases intracranial haemorrhage.23
The threshold of stroke risk where the benefit of anticoagulation exceeds the risk is controversial, but is somewhere between 3% and 5% per year, depending on the bleeding risks and preferences of the patient. This equates to a CHADS2 score of ≥ 2 (). Patients with a CHADS2 score of 0 do not require anticoagulation, and can be managed with aspirin 81–325 mg (I/A). The recommendations for those with a CHADS2 score of 1 now state that either aspirin or warfarin may be used (IIa/B).
Newly diagnosed or prior AF occurs in 15%–38% of patients presenting with ischaemic stroke, and increases mortality and disability (Professor G Donnan, NEMESIS study, personal communication).25 Therapeutic anticoagulation reduces the risk of disabling stroke; however, only about 10%–20% of patients with known AF are adequately anticoagulated immediately before their stroke.26,27 In a recent study, a third of patients with AF were on no treatment at the time of stroke, a third were on antiplatelet treatment, and a quarter were on warfarin with subtherapeutic INR; only one-eighth were on adequate warfarin at the time of the stroke.26
The reasons for undertreatment with antithrombotic agents are complex, but include a lack of knowledge about trials and guidelines,27 perceived “potential contraindications”, and fear of bleeding. Warfarin use increases in patients reviewed by cardiologists and by younger GPs.27
Elderly patients with AF have a greater net benefit from anticoagulation, at the expense of an increased risk of major haemorrhage.28,29 Cognitive function, falls risk, compliance, access to INR testing facilities, drug interactions due to polypharmacy, and the required changes in diet and lifestyle must be taken into account before committing an elderly patient to indefinite anticoagulation.
Warfarin use is very unlikely in patients older than 85 years, despite evidence of its safety in selected patients.30 Aiming for an INR of 2 may be a reasonable benefit–risk trade-off for primary prevention in elderly patients with non-valvular AF,4 although usual practice is to give aspirin in the very elderly.
The annual stroke risk is similar for patients with paroxysmal and permanent AF,19 so recommendations on antithrombotic therapy apply to both (IIa/B).
Although data on atrial flutter are sparse, anticoagulation guidelines are the same as for AF (I/C).
Patients on warfarin for AF may require additional antiplatelet therapy, such as after angioplasty. There is little evidence to guide management of anticoagulation and antiplatelet therapy in this context, and there is considerable variability in the approach taken by cardiologists.31 A balance is needed between preventing stent thrombosis and stroke, and minimising bleeding risk. In the absence of warfarin, dual antiplatelet therapy is required for longer with drug-eluting than bare metal stents (1 year, compared with about 4 weeks).32,33 When the presentation is an acute coronary syndrome, clopidogrel is beneficial for up to 1 year. Patients with a CHADS2 score ≤ 1 may have adequate protection against stroke during this time with antiplatelet drugs, and warfarin may be resumed thereafter. Patients with a CHADS2 score ≥ 2 or valvular AF may require a limited period with triple therapy until one or both antiplatelet drugs can be safely ceased (IIb/C).
1 Causes and predisposing factors for atrial fibrillation
Reversible causes of atrial fibrillation
Atrial fibrillation with associated heart disease
Hypertension (particularly when left ventricular hypertrophy is present)
Valvular heart disease (mitral or tricuspid valve disease)
Myocardial disease leading to systolic or diastolic dysfunction (ischaemic cardiomyopathy, dilated cardiomyopathy, hypertrophic cardiomyopathy)
Intracardiac tumours or thrombi
Amyloidosis
Age-induced atrial fibrotic changes
Haemochromatosis
Endomyocardial fibrosis
Primary or metastatic disease in or adjacent to the atrial wall
Electrophysiological abnormalities
Atrial fibrillation without associated heart disease
2 Recommendation and evidence codes
Codes are presented as Recommendation class/Evidence level according to the ACC/AHA/ESC 2006 classification scheme.4
IIa: Weight of evidence/opinion is in favour of usefulness/efficacy.
IIb: Usefulness/efficacy is less well established by evidence/opinion.
A: Data from multiple randomised clinical trials or meta-analyses.
B: Data from a single randomised trial or non-randomised studies.
C: Consensus opinion of experts, case studies, or standard of care.
5 Prognostic factors for ischaemic stroke and systemic embolism in patients with non-valvular atrial fibrillation
- Caroline Medi1
- Graeme J Hankey2,3
- Saul B Freedman1
- 1 Department of Cardiology, Concord Repatriation General Hospital, University of Sydney, Sydney, NSW.
- 2 Stroke Unit, Royal Perth Hospital, Perth, WA.
- 3 School of Medicine and Pharmacology, University of Western Australia, Perth, WA.
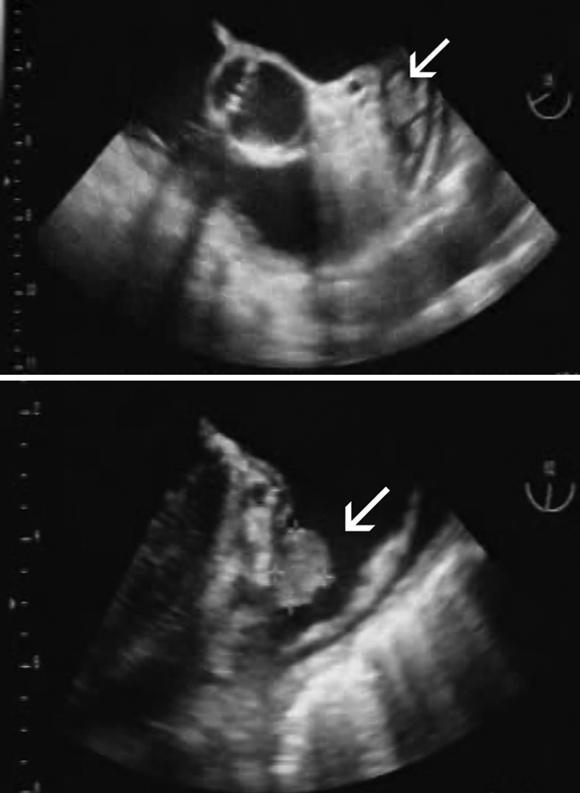
We gratefully acknowledge the assistance of Dr Andy Yong for providing the transoesophageal echo image of a left atrial appendage thrombus.
Saul Freedman received an honorarium for being on the AstraZeneca international advisory board for ximelagatran, a drug which has been removed from the market for liver toxicity as mentioned in the article. Graeme Hankey is a member of the executive committee of the rivaroxaban in atrial fibrillation trial (Johnson & Johnson Pharmaceutical Research and Development, USA), the steering committee of the atrial fibrillation trial of monitored, adjusted dose vitamin K antagonist, comparing efficacy and safety with unadjusted SanOrg 34006/idraparinux (AMADEUS) trial (Sanofi Aventis), and the stroke advisory committee of atrial fibrillation clopidogrel trial with irbesartan for prevention of vascular events (ACTIVE) trial (Sanofi Aventis). He was a member of international and national advisory boards for ximelagatran (AstraZeneca), for which he received honoraria and travel expenses to attend meetings.
- 1. Furberg CD, Psaty BM, Manolio TA, et al. Prevalence of atrial fibrillation in elderly subjects (the Cardiovascular Health Study). Am J Cardiol 1994; 74: 236-241.
- 2. Miyasaka Y, Barnes ME, Gersh BJ, et al. Secular trends in incidence of atrial fibrillation in Olmstead County, Minnesota, 1980 to 2000, and implications on the projections for future prevalence. Circulation 2006; 114: 119-125.
- 3. Mehra R, Benjamin EJ, Shahar E, et al. Association of nocturnal arrhythmias with sleep-disordered breathing. The Sleep Heart Health Study. Am J Resp Crit Care Med 2006; 173: 910-916.
- 4. Fuster V, Ryden LE, Cannom DS, et al. ACC/AHA/ESC 2006 Guidelines for the management of patients with atrial fibrillation: a report of the American College of Cardiology/ American Heart Association Task Force on Practice Guidelines and the European Society of Cardiology Committee for Practice Guidelines (Writing Committee to Revise the 2001 Guidelines for the Management of Patients With Atrial Fibrillation). Circulation 2006; 114: e257-e354.
- 5. Madrid AH, Peng J, Zamora J, et al. The role of angiotensin receptor blockers and/or angiotensin converting enzyme inhibitors in the prevention of atrial fibrillation in patients with cardiovascular diseases: meta-analysis of randomized clinical trials. Pacing Clin Electrophysiol 2004; 27: 1405-1410.
- 6. Patti G, Chello M, Candura D, et al. Randomized trial of atorvastatin for reduction of postoperative atrial fibrillation in patients undergoing cardiac surgery: results of the ARMYDA-3 (Atorvastatin for Reduction of MYocardial Dysrhythmia After cardiac surgery) Study. Circulation 2006; 114: 1455-1461.
- 7. Burgess DC, Kilborn MJ, Keech AC. Interventions for prevention of post-operative atrial fibrillation and its complications after cardiac surgery: a meta-analysis. Eur Heart J 2006; 27: 2846-2857.
- 8. Olshansky B, Rosenfeld LE, Warner AL, et al. The atrial fibrillation follow-up investigation of rhythm management (AFFIRM) study: approaches to control of rate in atrial fibrillation. J Am Coll Cardiol 2004; 43: 1201-1208.
- 9. Atrial Fibrillation Follow-Up Investigation of Rhythm Management (AFFIRM) Investigators. A comparison of rate control and rhythm control in patients with atrial fibrillation. N Engl J Med 2002; 347: 1825-1833.
- 10. Cappato R, Calkins H, Chen SA, et al. Worldwide survey on the methods, efficacy and safety of catheter ablation for human atrial fibrillation. Circulation 2005; 111: 1100-1105.
- 11. Wolf PA, Abbott RD, Kannel WD. Atrial fibrillation as an independent risk factor for stroke: the Framingham Study. Stroke 1991; 22: 983-988.
- 12. Kopecky SL, Gersh BJ, McGoon MD, et al. The natural history of lone atrial fibrillation. A population-based study over three decades. N Engl J Med 1987; 317: 669-674.
- 13. EAFT (European Atrial Fibrillation Trial) Study Group. Secondary prevention in non-rheumatic atrial fibrillation after transient ischaemic attack or minor stroke. Lancet 1993; 342: 1255-1262.
- 14. Risk factors for stroke and efficacy of antithrombotic therapy in atrial fibrillation. Analysis of pooled data from five randomized controlled trials. Arch Intern Med 1994; 154: 1449-1457.
- 15. Gage BF, van Walraven C, Pearce L, et al. Selecting patients with atrial fibrillation for anticoagulation: stroke risk stratification in patients taking aspirin. Circulation 2004; 110: 2287-2292.
- 16. Gage BF, Waterman AD, Shannon W, et al. Validation of clinical classification schemes for predicting stroke: results from the National Registry of Atrial Fibrillation. JAMA 2001; 285: 2864-2870.
- 17. van Walraven C, Hart RG, Wells GA, et al. A clinical prediction rule to identifiy patients with atrial fibrillation and a low risk of stroke while taking aspirin. Arch Intern Med 2003; 163: 936-943.
- 18. Hart RG, Benavente O, McBride R, et al. Antithrombotic therapy to prevent stroke in patients with atrial fibrillation: a meta-analysis. Ann Intern Med 1999; 131: 492-501.
- 19. Hart RG, Pearce LA, Rothbart RM, et al. Stroke with intermittent atrial fibrillation: incidence and predictors during aspirin therapy. Stroke Prevention in Atrial Fibrillation Investigators. J Am Coll Cardiol 2000; 35: 183-187.
- 20. Connolly S, Pogue J, Hart RG, et al. Clopidogrel plus aspirin versus oral anticoagulation for atrial fibrillation in the Atrial fibrillation Clopidogrel Trial with Irbesartan for prevention of Vascular Events (ACTIVE W): a randomised controlled trial. Lancet 2006; 367: 1903-1912.
- 21. Fang MC, Chang Y, Hylek EM, et al. Advanced age, anticoagulation intensity, and risk for intracranial hemorrhage among patients taking warfarin for atrial fibrillation. Ann Intern Med 2004; 141: 745-752.
- 22. Oden A, Fahlen M, Hart RG. Optimal INR for the prevention of stroke and death in atrial fibrillation: a critical appraisal. Thromb Res 2006; 117: 493-499.
- 23. Hart RG, Tonarelli SB, Pearce LA. Avoiding central nervous system bleeding during antithrombotic therapy: recent data and ideas. Stroke 2005; 36: 1588-1593.
- 24. Hallas J, Dall M, Andries A, et al. Use of single and combined antithrombotic therapy and risk of serious upper gastrointestinal bleeding: population based case–control study. BMJ 2006; 333: 726.
- 25. Paciaroni M, Agnelli G, Caso V, et al. Atrial fibrillation in patients with first-ever stroke: frequency, antithrombotic treatment before the event and effect on clinical outcome. J Thromb Haemost 2005; 3: 1218-1223.
- 26. O’Donnell M, Oczkowski W, Fang J, et al. Preadmission antithrombotic treatment and stroke severity in patients with atrial fibrillation and acute ischaemic stroke: an observational study. Lancet Neurol 2006; 5: 749-754.
- 27. Deplanque D, Leys D, Parnetti L, et al. Stroke prevention and atrial fibrillation: reasons leading to an inappropriate management. Main results of the SAFE II study. Br J Clin Pharmacol 2004; 57: 798-806.
- 28. van Walraven C, Hart RG, Singer DE, et al. Oral anticoagulants versus aspirin for stroke prevention in patients with non-valvular atrial fibrillation: the verdict is in. Card Electrophysiol Rev 2003; 7: 374-378.
- 29. Gage BF, Fihn SD, White RH. Warfarin therapy for an octogenarian who has atrial fibrillation. Ann Intern Med 2001; 134: 465-474.
- 30. Kagansky N, Knobler H, Rimon E, et al. Safety of anticoagulation therapy in well-informed older patients. Arch Intern Med 2004; 164: 2044-2050.
- 31. Lip GY, Karpha M. Anticoagulant and antiplatelet therapy use in patients with atrial fibrillation undergoing percutaneous coronary intervention: the need for consensus and a management guideline. Chest 2006; 130: 1823-1827.
- 32. Carrozza JP. Duration of clopidogrel therapy with drug-eluting stents. J Intervent Cardiol 2006; 19 (5 Suppl): s40-s45.
- 33. Smith SC, Feldman TE, Hirshfeld JW. ACC/AHA/SCAI 2005 guideline update for percutaneous coronary intervention-summary article. A report of the American College of Cardiology/American Heart Association Task Force on practice guidelines (ACC/AHA/SCAI writing committee to update the 2001 guidelines for percutaneous coronary intervention). J Am Coll Cardiol 2006; 47: 216-235.





Abstract
The incidence and prevalence of atrial fibrillation are increasing because of both population ageing and an age-adjusted increase in incidence of atrial fibrillation.
Deciding between a rate control or rhythm control approach depends on patient age and comorbidities, symptoms and haemodynamic consequences of the arrhythmia, but either approach is acceptable.
Digoxin is no longer a first-line drug for rate control: β-blockers and verapamil and diltiazem control heart rate better during exercise.
Anti-arrhythmic drugs have only a 40%–60% success rate of maintaining sinus rhythm at 1 year, and have significant side effects.
The selection of optimal antithrombotic prophylaxis depends on the patient’s risk of ischaemic stroke and the benefits and risks of long-term warfarin versus aspirin, but is independent of rate or rhythm control strategy.
Ischaemic stroke risk is best estimated with the CHADS2 score (Congestive heart failure, Hypertension, Age ≥ 75 years, Diabetes, 1 point each; prior Stroke or transient ischaemic attack, 2 points).
For patients with valvular atrial fibrillation or a CHADS2 score ≥ 2, anticoagulation with warfarin is recommended (INR 2–3, higher for mechanical valves) unless contraindicated or annual major bleeding risk > 3%.
Aspirin or warfarin may be used when the CHADS2 score = 1.
Aspirin, 81–325 mg daily, is recommended in patients with a CHADS2 score of 0 or if warfarin is contraindicated.
Stroke rate is similar for paroxysmal, persistent, and permanent atrial fibrillation, and probably for atrial flutter.