Cape York Peninsula and the Torres Strait Islands are home to about 20 000 people, up to 75% of whom are Indigenous Aboriginals and Torres Strait Islanders.1 Although acute rheumatic fever is considered a rare disease in other developed countries, it has an unusually high incidence in this population and places a significant health burden on these communities.2-4 The annual incidence of acute rheumatic fever in children aged 5–14 years has been estimated at 107/100 000 in Cape York Peninsula and 136/100 000 in the Torres Strait Islands.5 This is less than in the Top End of the Northern Territory, where incidence is reported to be between 202/100 0005 and 245/100 000.6 Many of these patients eventually require surgery for rheumatic valvular disease.6
Mortality data were expressed in accordance with published guidelines for reporting survival analyses.7 Deaths were classified as either valve-related or non-valve-related. Major complications were reported according to the American College of Cardiology/American Heart Association guidelines for reporting complications of prosthetic heart valves.8
Valve dysfunction was attributed to rheumatic heart disease (RHD) at the discretion of experienced echocardiographers using accepted morphological criteria.9 Portable Doppler echocardiography was only available from 2000.
Forty-seven patients were included in our analysis (Box 1). Of 30 patients who had echocardiographic evidence of RHD, 20 gave a history of past rheumatic fever.
Thirty-seven patients had valve replacements, 29 with mechanical valves and eight with bioprosthetic valves (Box 2). The mechanical valves used were produced by CarboMedics Inc (Austin, Tex, USA) (18 patients), St Jude Medical (St Paul, Minn, USA) (eight patients) and ATS Medical Inc (Minneapolis, Minn, USA) (three patients). The eight bioprosthetic valves were all singleton replacements.
Of patients with RHD, 29/30 had mitral valve procedures (Box 3). One patient with RHD had a homograft aortic valve and was still alive at 94 months’ follow-up. At 5 years’ follow-up, all seven patients who had undergone valve repair or valvotomy alone were still alive.
The six other patients were Indigenous people, of whom five had RHD. One patient had a homograft aortic valve replaced. Histological examination of the explanted valve showed non-Hodgkins lymphoma. In total, three of the eight bioprosthetic valves, one of the six open mitral repairs and two of four mitral balloon valvotomies required subsequent replacement as a result of valve dysfunction (Box 2).
Ours is the first study to document outcomes of cardiac surgery in Cape York Peninsula and the Torres Strait Islands. The 5-year survival rate of 83% in our study is similar to or better than survival figures published for valve surgery in older, non-Indigenous patients who are more likely to have degenerative valvular disease, in whom the 5-year survival rate is 70%–80%.10-15 Younger patients with rheumatic mitral valve repair or replacement alone typically have a 5-year survival rate of over 90%.16-20 These figures vary greatly depending on the type of surgery, the number of valves involved and the population studied. The question is whether outcomes in the population we studied could be improved, given that the median age of our cohort was only 40 years.
The only other comparable study of Indigenous patients undergoing cardiac valve replacement for RHD was done in the Northern Territory.21 The 5-year survival rate of 79% in the NT study was similar to the rate of 83% in our cohort. The 10-year survival rate was also similar (68% in the NT study compared with 61% in our study). Ninety-three per cent of the NT patients had mechanical valve replacements. As follow-up and data collection were noted to be very difficult in the NT cohort, it is possible that complication rates were under-reported.
The main limitation of our study was that it was too small to perform meaningful regression analysis to identify high-risk groups. For example, it would have been useful to know whether those who received anticoagulant had significantly higher rates of complications. We did observe that no valve-related deaths occurred in patients with normal or mildly elevated PAP. High PAP may be a surrogate marker for proceeding to “last resort” surgery in some patients. LV dysfunction has previously been shown to predict poorer outcomes,21 but 71% of our patients had normal preoperative LV function. Despite this, a large number of patients were symptomatic by the time of surgery. Another limitation of our study was the wide variation in review intervals: some patients would come for frequent assessment for a short time, then the review interval would be extended. The median follow-up interval of 20 months is still longer than ideal. The availability of close follow-up and ambulatory echocardiography is important for monitoring valve dimensions, LV function, PAP, symptoms, and the need for intervention.
A number of studies, including a recent meta-analysis,22 suggest that repair of rheumatic mitral valves is superior to replacement.20,22-27 The number of patients in our cohort was not high enough for our results to contribute directly to this comparison. However, it is worth noting that, of the seven patients who had dual mechanical valves placed, three have died and one is in a vegetative state after multiple embolic strokes. A further patient had dual mechanical valves placed at reoperation, then had a valvular thrombosis and died shortly afterwards. Thus, five of eight patients with dual mechanical prostheses had a poor outcome, the four deaths occurring within 5 years. In contrast, all seven patients with valve repairs or valvotomies and no valve replacement were alive at 5 years. The proportion of procedures that were repairs in our study is lower than that reported in some centres,20 and it may be that conservative surgery should be more readily available. Repair of rheumatic valves can be difficult, and perhaps procedures should be performed in a small number of centres by surgeons with expertise and extensive experience in this field.
Careful selection of patients is important, particularly if multiple valves require replacement. The many complications of anticoagulation therapy can be a problem in remote areas. Some patients manage anticoagulation reliably and should not be denied best treatment. Others find compliance very difficult. Even hospital anticoagulation clinics in the United Kingdom have historically achieved poor results with anticoagulation therapy, with less than 50% of patients achieving therapeutic international normalised ratios (INRs).28 Using “finger prick” INR testing is being trialled in some communities to provide timely feedback on anticoagulation.
2 Original procedures, number of reoperations and number of valve-related deaths for 47 patients undergoing cardiac valve procedures, 1992–2004
Received 20 May 2006, accepted 12 March 2007
- Anna McLean1
- Michael Waters2
- Emma Spencer3
- Clive Hadfield1
- 1 Cairns Base Hospital, Cairns, QLD.
- 2 Lyell McEwin Hospital, Adelaide, SA.
- 3 Royal Darwin Hospital, Darwin, NT.
We would like to acknowledge the assistance of Brad McCulloch of the Queensland Health Tropical Public Health Unit with statistical analysis.
None identified.
- 1. Australian Bureau of Statistics. Population distribution, Indigenous Australians, 2001. Canberra: ABS, 2001. (ABS Cat. No. 4705.0.)
- 2. Carapetis JR, McDonald M, Wilson NJ. Acute rheumatic fever. Lancet 2005; 366: 155-168.
- 3. Carapetis JR, Currie BJ. Mortaliy due to acute rheumatic fever and rheumatic heart disease in the Northern Territory: a preventable cause of death in Aboriginal people. Aust N Z J Public Health 1999; 23: 159-163.
- 4. Carapetis JR, Currie BJ. Preventing rheumatic heart disease in Australia. Med J Aust 1998; 168: 428-429. <MJA full text>
- 5. Hanna JN, Heazlewood RJ. The epidemiology of acute rheumatic fever in Indigenous people in north Queensland. Aust N Z J Public Health 2005; 29: 313-317.
- 6. Australian Institute of Health and Welfare. Rheumatic heart disease: all but forgotten in Australia except among Aboriginal and Torres Strait Islander peoples. Bulletin 16. Canberra: AIHW, 2004. http://www.aihw.gov.au/publications/aus/bulletin16/bulletin16.pdf (accessed May 2007).
- 7. Altman DG, De Stavola BL, Love SB, Stepniewska KA. Review of survival analyses published in cancer journals. Br J Cancer 1995; 72: 511-518.
- 8. Edmunds LH Jr, Clark RE, Cohn LH, et al. Guidelines for reporting morbidity and mortality after cardiac valvular operations. The American Association for Thoracic Surgery, Ad Hoc Liaison Committee for Standardizing Definitions of Prosthetic Heart Valve Morbidity. Ann Thorac Surg 1996; 62: 932-935.
- 9. Feigenbaum H, Armstrong W, Ryan T, editors. Feigenbaum’s echocardiography. 6th ed. Philadelphia: Lippincott Williams & Wilkins, 2005.
- 10. Bessell JR, Gower G, Craddock DR, et al. Thirty years experience with heart valve surgery: isolated aortic valve replacement. ANZ J Surg 1996; 66: 799-805.
- 11. Bessell JR, Gower G, Craddock DR, et al. Thirty years experience with heart valve surgery: isolated mitral valve replacement. ANZ J Surg 1996; 66: 806-812.
- 12. Ikonomidis JS, Kratz JM, Crumbley AJ 3rd, et al. Twenty-year experience with the St Jude Medical mechanical valve prosthesis. J Thorac Cardiovasc Surg 2003; 126: 2022-2031.
- 13. Calafiore AM, Di Mauro M, Gallina S, et al. Mitral valve surgery for chronic ischemic mitral regurgitation. Ann Thorac Surg 2004; 77: 1989-1997.
- 14. Cohn LH, Rizzo RJ, Adams DH, et al. The effect of pathophysiology on the surgical treatment of ischemic mitral regurgitation: operative and late risks of repair versus replacement. Eur J Cardiothorac Surg 1995; 9: 568-574.
- 15. Mantovani V, Mariscalco G, Leva C, et al. Long- term results of the surgical treatment of chronic ischemic mitral regurgitation: comparison of repair and prosthetic replacement. J Heart Valve Dis 2004; 13: 421-429.
- 16. De Santo L, Romano G, Della Corte A, et al. Mitral mechanical replacement in young rheumatic women: analysis of long-term survival, valve-related complications, and pregnancy outcomes over a 3707-patient-year follow-up. J Thorac Cardiovasc Surg 2005; 130: 13-19.
- 17. Pomerantzeff PM, Brandao CM, Faber CM, et al. Mitral valve repair in rheumatic patients. Heart Surg Forum 2000; 3: 273-276.
- 18. Choudhary SK, Talwar S, Dubey B, et al. Mitral valve repair in a predominantly rheumatic population. Long-term results. Tex Heart Inst J 2001; 28: 8-15.
- 19. Chauvad S, Fuzellier JF, Berrebi A, et al. Long-term (29 years) results of reconstructive surgery in rheumatic mitral valve insufficiency. Circulation 2001; 104 (12 Suppl 1): I12-I15.
- 20. Yau TM, El-Ghoneimi YA, Armstrong S, et al. Mitral valve repair and replacement for rheumatic disease. J Thorac Cardiovasc Surg 2000; 119: 53-60.
- 21. Carapetis JR, Powers JR, Currie BJ, et al. Outcomes of cardiac valve replacement for rheumatic heart disease in Aboriginal Australians. Asia Pacific Heart J 1999; 8: 138-147.
- 22. Shuhaiber J, Anderson RJ. Meta-analysis of clinical outcomes following surgical mitral valve repair or replacement. Eur J Cardiothorac Surg 2007; 31: 267-275.
- 23. Walkes J, Reardon MJ. Status of mitral valve surgery. Curr Opin Cardiol 2004; 19: 117-122.
- 24. Moss RR, Humphries KH, Gao M, et al. Outcome of mitral valve repair or replacement: a comparison by propensity score analysis. Circulation 2003; 108 Suppl 1: 1190-1197.
- 25. Song JK, Kim HS, Song JM, et al. Comparison of clinical and echocardiographic outcomes after valve repair: degenerative versus rheumatic mitral regurgitation. J Korean Med Sci 2003; 18: 344-348.
- 26. Talwar S, Rajesh MR, Subramanian A, et al. Mitral valve repair in children with rheumatic heart disease. J Thorac Cardiovasc Surg 2005; 129: 875-879.
- 27. DeAnda A Jr, Kasirajan V, Higgins R. Mitral valve replacement versus repair in 2003: where do we stand? Curr Opin Cardiol 2003; 18: 102-105.
- 28. Fitzmaurice DA, Raftery JP, Bryan S. Policy dilemmas for oral anticoagulation management. Br J Gen Pract 2000; 50: 779-780.





Abstract
Objective: To describe the outcome of valve surgery, for rheumatic heart disease (RHD) and non-RHD, in residents of Cape York Peninsula and the Torres Strait Islands referred to the Cairns Base Hospital specialist outreach service.
Design and participants: Retrospective review of medical records on all patients residing in the outreach area who had surgery for valvular heart disease between 1 January 1992 and 31 December 2004.
Main outcome measures: Operation type and perioperative characteristics; 5- and 10-year survival rates; reoperation rates; complications.
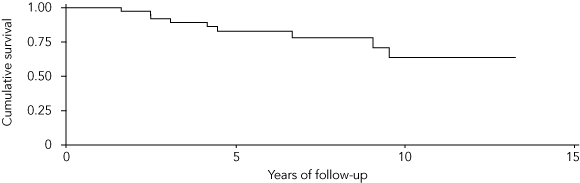
Results: Forty-seven patients met the selection criteria; the median age was 40 years (range, 4–76 years); and 39 patients were Indigenous. RHD was the predominant cause of valve dysfunction (30/47 patients). Thirty-seven patients had valve replacements, six had valve repair and four had balloon valvotomy as the initial procedure. There were three bleeding complications, two episodes of operated valve endocarditis, and six embolic complications. There were nine valve-related deaths (six in the first 5 years). At 5 years, all seven patients who had had valve repair or balloon valvotomy were alive. Seven of the 47 patients required reoperation. Survival analysis showed freedom from valve-related deaths to be 83% (95% CI, 66%–92%) at 5 years and 61% (95% CI, 33%–80%) at 10 years. Freedom from reoperation at 5 years was 88% (95% CI, 71%–95%). Among the 30 patients with RHD, freedom from valve-related death was 80% (95% CI, 60%–92%) at 5 years and 52% (95% CI, 21%–75%) at 10 years. In patients with RHD, freedom from reoperation at 5 years was 87% (95% CI, 65%–96%).
Conclusion: Valvular heart disease results in substantial morbidity and mortality, despite intervention. Efforts need to focus on prevention of rheumatic fever and closer follow-up.