Eczema is a papulovesicular dermatitis that occurs in 15%–20% of infants and young children. The onset of eczema (also known as atopic dermatitis or atopic eczema/dermatitis syndrome) frequently occurs in the first few months of life and usually within the first year of life. The exact cause of eczema is unknown, but factors that may precipitate or aggravate the condition include food and environmental allergens/irritants, heat or cold, stress, and genetic predisposition. Often dismissed as a trivial disorder, severe eczema can cause significant morbidity and have major social, emotional and financial impacts on children and their families.1
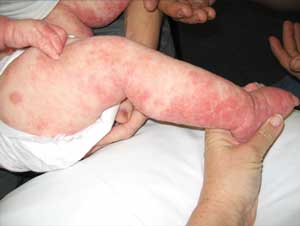
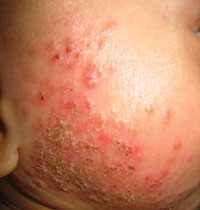
The diagnosis of eczema is made clinically, on the basis of the distribution of skin involvement and symptoms. The hallmarks are pruritus and inflammation. In infants, there is usually involvement of the extensor surfaces, face, neck and trunk (Box 1). As the child gets older, eczema more commonly involves the flexural surfaces, but can be widespread and also affect the hands, feet, face and trunk. In acute lesions, there are usually erythematous papules on a background of erythematous skin and pruritus, and sometimes vesiculation and ooze, but, over time, excoriation, scaling, lichenification and thickened plaques may develop (Box 2). The clinical course can be quite varied, often with periods of remission and exacerbation due to a variety of factors.
Eczema is not a single entity; rather, it is a group of inflammatory disorders of the skin involving a genetically determined skin barrier defect. Recent data suggest that, as for other diseases such as asthma, there are different forms of eczema. Eczema associated with IgE antibodies is called atopic eczema and eczema without IgE antibodies is called non-atopic eczema.2,3 Attempts to further delineate the different types of eczema will have important implications for management and prognosis of the disease. Atopic eczema is associated with other atopic diseases such as food allergy, allergic rhinitis and asthma, which may become evident at a later stage.
Management of eczema principally involves explanation to patients and parents of the chronic nature of the disease, advice on avoiding irritants, and symptom control (Box 1). Irritants such as soaps, perfumes, sand, extreme temperatures and some clothing fabrics should be avoided. As soap strips oils from the skin, adding oils to the bath and using aqueous creams as soap substitutes often works well. Bacterial infection, especially with Staphylococcus aureus, can often cause exacerbations of eczema. This can be difficult to diagnose clinically, and empirical treatment with antibiotics may be justified.
Oral antihistamines: Oral antihistamines may be useful in reducing pruritus in some patients. Older, sedating antihistamines should not be used, especially in young children, as there have been reports of severe and even life-threatening side effects.4
Emollients: There have been very few randomised controlled trials of the use of emollients in eczema, but those that have been done confirm their usefulness.5 Moisturising the skin improves its barrier function and reduces itch and irritation. There are a huge number of bathing and moisturising products available for treating eczema, and patients will often try many before finding one that suits. Products should not be continued if they cause burning, stinging or increased itching. Soft white paraffin is one of the more suitable moisturisers and is best applied immediately after getting out of the bath, while the patient is still wet and warm. Wet wraps may be effective in patients with moderate to severe eczema and provide a partial barrier to scratching.
Topical steroids: Inflammation needs to be controlled, and topical steroid creams and ointments are effective for this purpose.5 A moderate to potent steroid should be applied at the initial signs of inflammation. As the inflammation subsides, steroid use should be tapered off to prevent rebound. This will generally provide better resolution and require a lower total dose of steroid than if a low potency steroid is used alone for a prolonged period. Parental concern about the effects of steroid use, including thinning of the skin, can lead to problems with compliance. However, if steroids are used appropriately for short periods, there is little evidence to support this concern.6
Tacrolimus ointment has been found to be safe and effective in patients with moderate to severe eczema.7 Transient burning and pruritus can occur when the ointment is initially applied. Pimecrolimus is similar in action to tacrolimus but has only been assessed in mild to moderate disease. It can be used to prevent flaring of eczema,8 and may be useful in body areas where there is concern about applying steroids (eg, on the face, around the eyes and around the groin).
Safety concerns about topical calcineurin inhibitors have been raised with respect to increased risk of skin malignancy (on the basis of animal studies), but a review of the literature suggests that “black box” warnings are not indicated.9 Nevertheless, calcineurin inhibitors should not be used in clinically infected areas. Further, there are few studies assessing the effectiveness of these newer agents compared with appropriate strength topical steroids.7
The role of food allergy in causing or exacerbating eczema is controversial, perhaps because many studies of food avoidance have used an unselected population of children with and without sensitisation to foods. Also, foods used in the control group (eg, soy instead of cows milk) may themselves have elicited reactions. Recently, numerous double-blind, placebo-controlled food studies have demonstrated that about 35%–40% of children with moderate to severe eczema have food allergy, and that eliminating the causative food from their diet can bring about significant improvement in the severity and extent of eczema (Box 3).11-14 However, eczema is often multifactorial, and elimination of allergenic foods does not provide complete resolution in all infants. Food allergy is rare in adult-onset eczema.
Allergy to egg is the most common food allergy associated with eczema. Other commonly associated foods include milk, soy, wheat and peanuts. Thus, food allergy screening tests such as skin prick tests and radioallergosorbent tests should be performed in young infants with severe eczema. False negative tests are uncommon, but false positive tests may occur, particularly in older children.15 Food challenges are more accurate; reactions may be immediate or delayed. Food allergens from the maternal diet may be excreted in breastmilk and cause eczema in a breastfed infant.16 A trial of restriction of maternal diet may be useful if an infant is shown to be allergic to a particular food.
Patients with eczema may have sensitisation to environmental allergens such as house dust mite (HDM) or pet dander. There is strong circumstantial evidence that HDM is an important precipitant for atopic eczema. However, there are relatively few randomised controlled trials looking at the role of HDM eradication in treatment of eczema. The limited studies suggest that there is some benefit if a very low level of HDM can be achieved.5
Based on the concept that probiotics, which are normal commensal bacteria of the healthy human gut, play a role in the development of normal immune tolerance and may be immunomodulatory, recent studies have looked at probiotics in the treatment and prevention of allergy.17,18 These studies look promising, but further work is required to clarify whether these agents should be recommended and which is most effective. There is currently no evidence for the use of other dietary supplements such as evening primrose oil, fish oils or omega-3 fatty acids.
For most infants and children, eczema is a mild disease that will resolve during childhood, but for some patients, the condition is severe and may continue into adulthood. The prognosis for eczema has been shown to correlate with its severity and the presence of atopic sensitivity.19 Patients with chronic, severe eczema and their families need adequate support.
The significance of chronic urticaria is sometimes trivialised because it is a non-life-threatening disease. However, the unrelenting pruritus and disfiguring, random occurrence of unsightly weals can cause patients great misery. Chronic urticaria has a major impact on quality of life, with impact on activities of daily living similar to that experienced by patients with heart disease.20
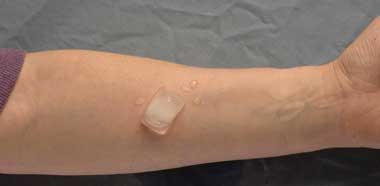
Urticaria with a physical cause: A physical cause for chronic urticaria is probably present in about 20% of cases (Box 4, Box 5). Dermatographism and cholinergic urticaria are the most common forms of urticaria with a physical cause and have characteristic appearances. Various types of urticaria may coexist in the same patient. However, once the typical history is elicited, no further investigations are required.
Urticarial vasculitis: An atypical appearance, long-lasting lesions or a lack of responsiveness to antihistamine treatment suggests urticarial vasculitis, a rare condition seen in less than 1% of people with chronic urticaria. Patients suspected of having this condition require further investigation (Box 6).
Some years ago, there were reports that addition of an H2 antagonist could help control chronic urticaria. The benefit is small, but the treatment may be worth trying, as it may help selected patients.21 A number of studies have demonstrated benefit from the use of newer, non-sedating antihistamines over that derived from placebo.22-25
Leukotriene inhibitors: Theoretically, there may be a use for leukotriene antagonists in the treatment of chronic urticaria. However, randomised controlled trials comparing the use of an antihistamine alone with use of a combined treatment (antihistamine plus montelukast) have shown conflicting evidence of efficacy.23,26 Currently, montelukast is not listed for use in chronic urticaria and is an expensive treatment.
There has been concern, based on anecdotal reports, that chronic urticaria may be an indicator of an underlying malignancy, particularly in older patients. A very large Swedish epidemiological study has shown that there is little evidence to support this fear.27 However, acquired angioedema associated with C1-esterase inhibitor deficiency is associated with malignancy, particularly lymphoma.27
Idiosyncratic reactions to food additives have been suggested as an important cause of chronic urticaria, but this area suffers from a lack of rigorous placebo-controlled studies and also from the inherent problem of challenging patients with chronic urticaria and then trying to interpret the results. Early studies reporting reactions to tartrazine and benzoates in patients with chronic urticaria were not placebo-controlled.28 Other complicating factors include whether or not an elimination diet was used before challenging, which food additives were included in the challenge battery, the dosages of food additives used, and whether medication was ceased before challenging (and if so, for how long). Based on the few rigorous studies that have been performed, food additives appear to be a distinctly uncommon cause of chronic urticaria.29
The question of whether Helicobacter pylori infection is a cause of chronic urticaria remains controversial. A link between the two, first described in the European literature, has not been confirmed by other studies.30 Reports of improvement in chronic urticaria after eradication of H. pylori infection have added to the debate.31 Although H. pylori seropositivity is not higher in patients with chronic urticaria than in other patients, eradication of the infection where it coexists appears worthwhile. Patients presenting with a history of reflux and urticaria may be considered for investigation for H. pylori and appropriate treatment if found.
A link between chronic urticaria and thyroid autoimmunity has been known of for more than 20 years.32 Kikuchi et al33 reported that patients with chronic urticaria had an increased prevalence of IgG antithyroid antibodies (usually antithyroid peroxidase) compared with controls (10%–13% v 3%–5%). They demonstrated that antithyroid antibodies are definitely associated with the presence of antibodies to the IgE receptor, and concluded that antithyroid antibodies and antibodies to the IgE receptor are associated immune abnormalities, rather than one being the cause of the other.
The use of thyroxine to treat patients with chronic urticaria and thyroid autoimmunity in the absence of thyroid disease remains controversial. Some investigators have reported improvement in chronic urticaria symptoms with thryoxine treatment.34 In a recent study that included both euthyroid and hypothyroid women, 16 of 20 women with chronic urticaria who were given thyroxine to suppress thyroid-stimulating hormone showed improvement in chronic urticarial symptoms after 12 weeks.35
Fact or fiction — true or false?
1. All eczema is allergy-based (T/F)
2. Food allergy plays no role in eczema (T/F)
3. Food allergy is an important cause of chronic urticaria (T/F)
4. Five per cent of the population suffers from dermatographism (T/F)
2. False. 35%–40% of children with moderate to severe eczema have food allergy.
In the past decade, 35%–45% of patients with chronic urticaria have been found to have an autoimmune basis for their condition. An IgG antibody directed to the alpha subunit of the IgE receptor has been demonstrated in 35% of patients, and a functional anti-IgE antibody has been found in a further 5%–10%.36 Measurement of these antibodies is confined to research laboratories. However, an intradermal skin test with autologous sera can induce a delayed weal with a macroscopic and microscopic appearance similar to that seen in chronic urticaria. The stimulus for this autoantibody production is unknown.
The notion that chronic urticaria may be an autoimmune disease opens up the possibility of using immunomodulatory treatment when antihistamines have failed to control the condition. There is one controlled trial demonstrating a positive effect from the use of hydroxychloroquine.37 Cyclosporin A has been shown to be an effective treatment, but its use should be restricted and requires careful monitoring because of its potential to cause renal and other side effects.38 Plasmapheresis or administration of intravenous gammaglobulin or low-dose tacrolimus have also been tried, with some benefit, in small numbers of patients with autoimmune chronic urticaria.
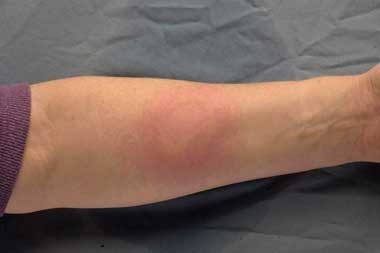
2 Chronic eczema in a 10-year-old child
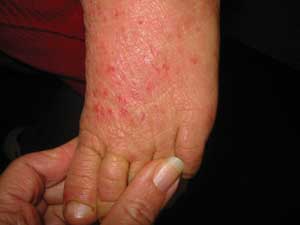
Over time, with continued inflammation and scratching, the skin becomes very thick.
3 Evidence-based practice tips*
Patients with atopic eczema have a higher likelihood of developing other atopic diseases such as asthma and allergic rhinitis (Level III-2).
Removal of an identified allergenic food from the diet can significantly improve eczema in an individual patient (Level III-2).
* Based on National Health and Medical Research Council levels of evidence.10
4 Physical forms of chronic urticaria
6 Urticarial vasculitis*
Lesions lasting longer than 24 hours in the one place
Condition painful rather than itchy
Bruising or scarring after occurrence of lesions
Presence of arthralgia or arthritis
Presence of constitutional symptoms
Lack of response to antihistamines
Investigations when urticarial vasculitis is suspected
Skin biopsy to demonstrate vasculitic changes in postcapillary venules (leukocytoclastic vasculitis)
Full blood count and erythrocyte sedimentation rate
Urinalysis
Antinuclear antibodies to extractable nuclear antigens
Complement studies
- Constance H Katelaris1
- Jane E Peake2
- 1 Allergy and Immunology Services, Westmead Specialist Medical Centre, Sydney, NSW.
- 2 Department of Paediatrics and Child Health, University of Queensland, Brisbane, QLD.
None identified.
- 1. Paller AS, McAlister RO, Doyle JJ, Jackson A. Perceptions of physicians and pediatric patients about atopic dermatitis, its impact, and its treatment. Clin Pediatr (Phila) 2002; 41: 323-332.
- 2. Kusel MM, Holt PG, de Klerk N, Sly PD. Support for 2 variants of eczema. J Allergy Clin Immunol 2005; 116: 1067-1072.
- 3. Johansson SG, Bieber T, Dahl R, et al. Revised nomenclature for allergy for global use: report of the Nomenclature Review Committee of the World Allergy Organization, October 2003. J Allergy Clin Immunol 2004; 113: 832-836.
- 4. Starke PR, Weaver J, Chowdhury BA. Boxed warning added to promethazine labeling for pediatric use. N Engl J Med 2005; 352: 2653.
- 5. Hoare C, Li Wan Po A, Williams H. Systematic review of treatments for atopic eczema. Health Technol Assess 2000; 4(37). http://www.ncchta.org/execsumm/summ437.htm (accessed Sep 2006).
- 6. Atherton DJ. Topical corticosteroids in atopic dermatitis. BMJ 2003; 327: 942-943.
- 7. Conroy S. New products for eczema. Arch Dis Child Educ Pract Ed 2004; 89: ep23-ep26.
- 8. Wahn U, Bos JD, Goodfield M, et al. Efficacy and safety of pimecrolimus cream in the long-term management of atopic dermatitis in children. Pediatrics 2002; 110 (1 Pt 1): e2.
- 9. Fonacier L, Spergel J, Charlesworth EN, et al. Report of the Topical Calcineurin Inhibitor Task Force of the American College of Allergy, Asthma and Immunology and the American Academy of Allergy, Asthma and Immunology. J Allergy Clin Immunol 2005; 115: 1249-1253.
- 10. National Health and Medical Research Council. How to use the evidence: assessment and application of scientific evidence. Canberra: NHMRC, 2000. http://www.nhmrc.gov.au/publications/_files/cp69.pdf (accessed Jul 2006).
- 11. Sampson HA, Scanlon SM. Natural history of food hypersensitivity in children with atopic dermatitis. J Pediatr 1989; 115: 23-27.
- 12. Sampson HA, McCaskill CC. Food hypersensitivity and atopic dermatitis: evaluation of 113 patients. J Pediatr 1985; 107: 669-675.
- 13. Lever R, MacDonald C, Waugh P, Aitchison T. Randomised controlled trial of advice on an egg exclusion diet in young children with atopic eczema and sensitivity to eggs. Pediatr Allergy Immunol 1998; 9: 13-19.
- 14. Eigenmann PA, Sicherer SH, Borkowski TA, et al. Prevalence of IgE-mediated food allergy among children with atopic dermatitis. Pediatrics 1998; 101(3): e8.
- 15. Sampson HA, Albergo R. Comparison of results of skin tests, RAST, and double-blind, placebo-controlled food challenges in children with atopic dermatitis. J Allergy Clin Immunol 1984; 74: 26-33.
- 16. Cavagni G, Paganelli R, Caffarelli C, et al. Passage of food antigens into circulation of breast-fed infants with atopic dermatitis. Ann Allergy 1988; 61: 361-365.
- 17. Weston S, Halbert A, Richmond P, Prescott SL. Effects of probiotics on atopic dermatitis: a randomised controlled trial. Arch Dis Child 2005; 90: 892-897.
- 18. Isolauri E, Arvola T, Sutas Y, et al. Probiotics in the management of atopic eczema. Clin Exp Allergy 2000; 30: 1604-1610.
- 19. Illi S, von Mutius E, Lau S, et al. The natural course of atopic dermatitis from birth to age 7 years and the association with asthma. J Allergy Clin Immunol 2004; 113: 925-931.
- 20. O’Donnell BF, Lawler F, Simpson J, et al. The impact of chronic urticaria on quality of life. Br J Dermatol 1997; 136: 197-201.
- 21. Commens CA, Greaves MW. Cimetidine in chronic idiopathic urticaria: a randomised double-blind study. Br J Dermatol 1978; 99: 675-676.
- 22. Nelson HS, Reynolds R, Mason J. Fexofenadine HCl is safe and effective for treatment of chronic idiopathic urticaria. Ann Allergy Asthma Immunol 2000; 84: 517-522.
- 23. Di Lorenzo G, Pacor ML, Mansueto P, et al. Randomised placebo-controlled trial comparing desloratadine and montelukast in monotherapy and desloratadine plus montelukast in combined therapy for chronic idiopathic urticaria. J Allergy Clin Immunol 2004; 114: 619-625.
- 24. Paul E. Loratadine and terfenadine in the treatment of chronic urticaria. Allergy 1988; 43 Suppl 7: 122.
- 25. Charlesworth EN, Massey WA, Kagey-Sobotka A, et al. Effect of H1 receptor blockade on the early and late response to cutaneous allergen challenge. J Pharmacol Exp Ther 1992; 262: 964-970.
- 26. Sanada S, Tanaka T, Kameyoshi Y, Hide M. The effectiveness of montelukast for the treatment of anti-histamine-resistant chronic urticaria. Arch Dermatol Res 2005; 297: 134-138.
- 27. Sigurgeirsson B. Skin disease and malignancy: an epidemiological study. Acta Derm Venereol Suppl (Stockh) 1992; 178: 1-110.
- 28. Doeglas HMG. Reactions to aspirin and food additives in patients with chronic urticaria, including the physical urticarias. Br J Dermatol 1975; 93: 135-144.
- 29. Simon RA. Additive-induced urticaria: experience with monosodium glutamate. J Nutr 2000; 130: 1063S-1066S.
- 30. Wedi B, Wagner S, Werfel T, et al. Prevalence of Helicobacter pylori-associated gastritis in chronic urticaria. Int Arch Allergy Immunol 1998; 116: 288-294.
- 31. De Koster E, De Bruyne I, Langlet P, Deltenre M. Evidence-based medicine and extradigestive manifestations of Helicobacter pylori. Acta Gastroenterol Belg 2000; 63: 388-392.
- 32. Leznoff A, Josse RG, Denburg J, Dolovich J. Association of chronic urticaria and angioedema and thyroid autoimmunity. Arch Dermatol 1983; 119: 636-640.
- 33. Kikuchi Y, Fann T, Kaplan AP. Antithyroid antibodies in chronic urticaria and angioedema. J Allergy Clin Immunol 2003; 112: 218.
- 34. Rumbyrt JS, Katz JL, Schocket A. Resolution of chronic urticaria in patients with thyroid autoimmunity. J Allergy Clin Immunol 1995; 96: 901-905.
- 35. Alversano M, Caiazzo P, Iorio G, et al. Improvement of chronic urticaria with l-thyroxine: a new TSH role in immune response? Allergy 2005; 60: 489-493.
- 36. Tong LJ, Balakrishnan G, Kochan JP, et al. Assessment of autoimmunity in patients with chronic urticaria. J Allergy Clin Immunol 1997; 99: 461-465.
- 37. Reeves GE, Boyle MJ, Bonfield J, et al. Impact of hydroxychloroquine therapy on chronic urticaria: chronic autoimmune urticaria study and evaluation. Intern Med J 2004; 34: 182-186.
- 38. Grattan CE, O’Donnell BF, Francis DM, et al. Randomized double-blind study of cyclosporin in chronic “idiopathic” urticaria. Br J Dermatol 2000; 143: 365-372.





Abstract
Eczema is common, occurring in 15%–20% of infants and young children. For some infants it can be a severe chronic illness with a major impact on the child’s general health and on the family. A minority of children will continue to have eczema as adults.
The exact cause of eczema is not clear, but precipitating or aggravating factors may include food allergens (most commonly, egg) or environmental allergens/irritants, climatic conditions, stress and genetic predisposition.
Management of eczema consists of education; avoidance of triggers and allergens; liberal use of emollients or topical steroids to control inflammation; use of antihistamines to reduce itch; and treatment of infection if present. Treatment with systemic agents may be required in severe cases, but must be supervised by an immunologist.
Urticaria (“hives”) may affect up to a quarter of people at some time in their lives. Acute urticaria is more common in children, while chronic urticaria is more common in adults. Chronic urticaria is not life-threatening, but the associated pruritus and unsightly weals can cause patients much distress and significantly affect their daily lives.
Angioedema coexists with urticaria in about 50% of patients. It typically affects the lips, eyelids, palms, soles and genitalia.
Management of urticaria is through education; avoidance of triggers and allergens (where relevant); use of antihistamines to reduce itch; and short-term use of corticosteroids when antihistamine therapy is ineffective. Referral is indicated for patients with resistant disease.