Australian general practitioners are responsible for the care of an increasingly multicultural patient population. Between 1998 and 1999, 80% of people arriving in Australia under the Humanitarian Program were from the former Yugoslavia, the Middle East or south-west Asia (including Iraq, Afghanistan, and Iran), while in 2004 to 2005, 80% were from Africa.1 Although many of the health problems of refugees are similar irrespective of their country of origin, the recent changes in migration patterns have resulted in immigrants presenting with more complex conditions that are not commonly encountered in Australia.2
The difficulties faced by GPs servicing refugee communities, which include inadequate remuneration, language difficulties and lack of access to information about health needs, have been previously described.3 Yet in most states in Australia, refugee health care is decentralised, and GPs are relied on to perform important early health screening and assessment as well as ongoing care with little extra support. The recent federal government decision to grant a special refugee Medical Benefits Schedule number recognises the importance of GPs in caring for refugees.4
Policy and planning decisions and refugee health assessment strategies need to be informed by good quantitative data. Studies on the health status of African refugee patients have so far concentrated on infectious diseases,5,6 or have been confined to children.7 We therefore conducted a study in Melbourne to identify the most common conditions diagnosed by GPs in newly arrived African refugees.
Data were collected by six GPs who were identified as providing initial care for African refugee patients. Four GPs worked in two community health centres in the western and northern suburbs of Melbourne and two worked in private practices in the western and south-eastern suburbs.
The GPs identified all the African refugee patients on their registries who arrived in Australia after 1 July 2004 and were seen for the first time between 1 January and 30 June 2005. African refugee patients were identified in various ways, depending on patient-record keeping methods at the health centres or general practices. One recorded the country of birth and extracted records of all those born in Africa, two kept a database of the refugee status of patients, and one doctor depended on recall of individual patients. GPs transcribed data from the medical records of these patients onto data collection forms and recorded information on patient demographics, the reason for the first consultation, laboratory investigation results, and final diagnoses. Diagnoses were described as a disease, symptom or complaint, social problem or ill-defined condition depending on the evidence available at the time of the study. Large private pathology service providers were used for routine laboratory testing, and standard reference ranges were used. Tests for schistosomiasis, strongyloidiasis and positive blood smears for malaria were referred to the Victorian Infectious Diseases Reference Laboratory. Personal identifying information was not recorded on the questionnaires, and only the treating doctor viewed patient files.
Data were entered into a Microsoft Access database (Microsoft Corporation, Redmond, Wash, USA). A relational database was used in which each patient could have many diagnoses. To ensure uniform description of diagnoses, standard entry formats for common diagnoses were listed for the person entering the data. This list was compiled by reviewing likely diagnoses that were found from guidelines and reports.8,9 To standardise entry, a drop-down box with diagnoses listed in ICPC-2-E (an electronic version of the revised and corrected chapters 10 and 11 of the International classification of primary care, second edition)10 was used in conjunction with free text.11
Data were checked for missing entries, duplicates, outliers and inconsistencies. Consistency between laboratory results and final diagnoses was checked during data entry and while data were being analysed. Inconsistencies were discussed with GPs and corrected. Common diagnoses were grouped together and ranked according to frequency, and associations were analysed with Stata, version 8 (StataCorp, College Station, Tex, USA). The significance of the difference in proportions was tested by means of P values calculated on χ2 distributions.
Ethical approval for this project was obtained from the Victorian Government Department of Human Services Human Research Ethics Committee. As a relatively small group of patients were involved in this study, we felt that information on HIV status was potentially identifiable; therefore these results have not been included.
Between 1 September and 30 November 2005, data were collected from 258 patient files. Box 1 summarises characteristics of the population.
The most common laboratory tests performed were full blood examinations, vitamin D levels and schistosomiasis serology (Box 2). Vitamin D deficiency was the condition most frequently detected by laboratory investigations, followed by anaemia, iron deficiency, gastrointestinal parasite infection, and schistosomiasis (Box 3). There were variations in the prevalence of conditions between regions of birth. Compared with refugees born in eastern Africa, refugees from western Africa were unlikely to have vitamin D deficiency, while schistosomiasis and hepatitis B infection were more prevalent.
Only a third of patients were screened for latent tuberculosis (TB), but of those, 25% (24/96) had a positive Mantoux test result (> 9 mm induration) or positive Quanti-FERON-TB Gold test (Cellestis International, Melbourne, Vic) result. In patients aged 35 years and under, 84/228 (37%) were tested and 18/84 (21%) were positive and eligible for prophylactic treatment. Among these, two were subsequently found to have clinical symptoms and chest x-ray findings consistent with active TB (pleural effusion and middle lobe opacity) and were referred for further management.
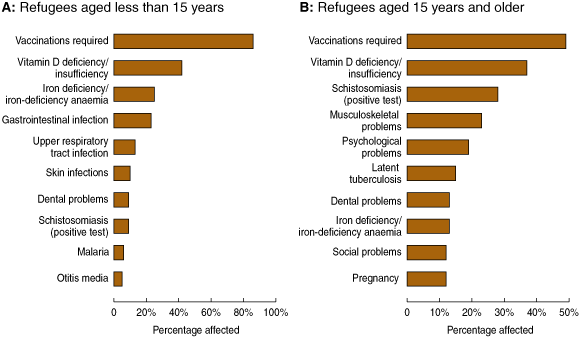
Inadequate vaccination was the most common problem identified by GPs (Box 4). Serological testing indicated that a high proportion of refugee patients, particularly children, were not immune to common vaccine-preventable diseases (Box 5). Other common problems in children included vitamin D deficiency/insufficiency, gastrointestinal infections and iron-deficiency anaemia, upper respiratory tract infections, skin infections (including tinea, scabies and folliculitis), and dental disease (Box 4A). Vitamin D deficiency/insufficiency was also common among adult patients. More common in adults than children were musculoskeletal problems and psychological problems, including depression, anxiety and post-traumatic stress disorder. Social issues were encountered in 12% of adults and included housing problems, parenting difficulties and financial problems (Box 4B).
Our study shows that recently arrived refugees from African countries presenting to GP clinics in Melbourne have previously unrecognised health conditions, including inadequate immunity to vaccine-preventable diseases, latent tuberculosis, gastrointestinal infections, schistosomiasis, and nutritional deficiencies.
Our study does not reflect the true prevalence of disease in this population, but rather highlights some of the health problems that exist. The study has several limitations. Specific GPs were invited to participate because they were known to have an interest in refugee health and had included comprehensive health assessments in their routine practice. This convenience sampling and retrospective data analysis allowed rapid assessment of health problems, but may have led to biases that mean that our results are not applicable to all African refugees in Melbourne.
For example, most people in our study were born in Sudan or Liberia, and the health priorities that were highlighted reflect the health issues in these groups. While diseases such as iron-deficiency anaemia and gastrointestinal infections were common in all groups, there were differences according to country of origin in the prevalence of other diseases such as vitamin D deficiency. However, of the 3818 humanitarian entrants to Victoria between 1 July 2004 and 30 June 2005, 60.1% were from Sudan, and the second most common African country of origin was Liberia (5.7%).1 Most refugees arriving in Victoria were young (52% of those arriving in 2000–2005 were aged under 25 years),1 which is similar to our study group.
The accuracy of the health problems we report was dependent on the accuracy of GP diagnoses. Conditions that are well known in refugee populations are more likely to be investigated and treated while others, such as Helicobacter pylori infection,12 may be under-diagnosed. The fact that most patients in our study attended for health assessment and did not have an initial presenting complaint may have introduced another bias. Refugees with jobs, and those without children, for example, may have been less likely to attend. Not all patients had comprehensive health assessments despite the obvious commitment and motivation of the GPs concerned. The reasons for this require further investigation and need to be considered by those issuing health assessment guidelines.
There was a high rate of inadequate immunity to vaccine-preventable diseases. For example, immunity to measles was 56% in 1–14-year-olds in the study population, compared with 90% in Australian children aged 1–18 years.13 The low level of immunity to tetanus in both adults and children further emphasises the need for adult diphtheria–tetanus vaccines, supplies of which have been reduced since the introduction of the conjugated adult pertussis–diphtheria–tetanus vaccine for adolescents.14
Malaria is likely to cause the most immediate threat to the health of African refugee patients, particularly children, although there were only a few cases in this series. Plasmodium falciparum malaria is common in many parts of Africa, and can be fatal. Testing of newly arrived refugees is a priority, especially if patients are symptomatic.
A quarter of the refugees who were screened with a Mantoux or QuantiFERON-TB Gold test had a positive result, although this rate was much lower than the 50% reported in a Minnesota study of sub-Saharan African refugees.5 Possible explanations for this difference include variations in patient age, previous exposure to vaccination, and country of origin or transit. The significant number of refugees with apparent latent TB emphasises the importance of testing in this population, as it is considered that about 10% may go on to develop active TB.15 Prophylactic treatment with isoniazid will reduce the chance of active disease by 2.5 times,16 and treatment is recommended in Victoria17 and overseas18 for those under 35 years of age with a positive screening test result.
Many of the study patients were not tested for TB, possibly because Mantoux testing was not available in the laboratories that were close to GP clinics, or because of poor compliance with follow-up appointments at 48–72 hours to read the test. The Quanti-FERON-TB Gold test requires only one blood test and current evidence suggests that it may be a good alternative to the Mantoux test.15 While its use in children, immunocompromised individuals, those with extrapulmonary disease or those from endemic areas has not been established, in other adults, it has been found to be more specific and as sensitive as the Mantoux test.15 However, the Medicare rebate for the QuantiFERON-TB Gold test is currently only available to immunocompromised patients.19 Improving the rate of TB testing will require that such tests be made more affordable to immigrant groups.
As schistosomiasis and strongyloidiasis can be persistent and cause possible serious complications if they are left untreated,20,21 most clinicians offer treatment to seropositive patients. Treatment for schistosomiasis is relatively simple, requiring two to three doses of praziquantel,20 and treatment in the general practice setting is possible. However, this may be impeded by the cost of praziquantel ($52.54), which is not listed under the Pharmaceutical Benefits Schedule (PBS).22 Similarly, two doses of ivermectin are recommended for strongyloidiasis,23 but special approval is required to prescribe this medication under the PBS unless the patient is immunocompromised.22
As has been previously suggested,24 nutritional deficiencies were common in the refugees. The use of ferritin levels as a marker of iron deficiency may have led to an underestimate because ferritin levels may be elevated in chronic inflammatory conditions. Iron deficiency in this setting is likely to be the result of a combination of factors, including poor nutrition and parasitic infections.24 Possible risk factors for the high levels of vitamin D deficiency observed include dark skin, lack of sunlight in refugee accommodation in some countries, and decreased sun exposure after settling in Melbourne. Screening and treatment are import-ant as deficiency may lead to rickets in children and osteomalacia in adults if left untreated.25 Studies have also linked deficiencies with other conditions such as tuberculosis and musculoskeletal pain.25,26
Psychological problems such as depression, anxiety and post-traumatic stress disorder are common in refugees as a result of past experiences of violence.12 Standard questionnaires were not used to screen for psychological problems in our study population. Diagnoses were based on GP opinions recorded in patient notes, and the finding that 19% of adult refugee patients were thought to have mental health issues may not accurately reflect the true prevalence of psychological problems; the high physical health care needs of this population are likely to have impeded exploration of psychological issues during early contact with health care providers.
Australia’s willingness to accept refugees shows enormous humanity and bodes well for the Australian motto of the “fair go”. However, African refugees settling here have considerable health care needs which must be met to ensure their healthy and productive futures in Australia. While our study has limitations, it provides a useful indication of the common health problems in this population. GPs need to be aware of these health problems and need support in conducting comprehensive health assessments during the early settlement period. These assessments may be assisted by the use of health assessment guidelines that are avail-able from many sources (eg, http://www.internationalhealth.unimelb.edu.au/). The removal of logistic and cultural barriers to refugees accessing appropriate health care and important investigations and treatment options, such as praziquantel for schistosomiasis and ivermectin for strongyloidiasis, will assist this process.
1 Characteristics of 258 newly arrived African refugees seen in general practice clinics in Melbourne, 2005
Characteristic |
No. |
||||||||||||||
Age |
|
||||||||||||||
0–14 years |
128 (50%) |
||||||||||||||
15–62 years |
130 (50%) |
||||||||||||||
Sex |
|
||||||||||||||
Female |
144 (56%) |
||||||||||||||
Male |
114 (44%) |
||||||||||||||
Country of birth |
|
||||||||||||||
Eastern Africa* |
170 (66%) |
||||||||||||||
Western Africa† |
65 (25%) |
||||||||||||||
Central Africa‡ |
23 (9%) |
||||||||||||||
Clinic attended |
|
||||||||||||||
Community health centre A |
170 (66%) |
||||||||||||||
Community health centre B |
57 (22%) |
||||||||||||||
Private practice A |
25 (10%) |
||||||||||||||
Private practice B |
6 (2%) |
||||||||||||||
Reason for first presentation |
|
||||||||||||||
Health screening |
192 (74%) |
||||||||||||||
Health problem |
66 (26%) |
||||||||||||||
* Sudan, 146; Ethiopia, 8; Kenya, 6; Somalia, 6; Egypt, 4. Transit countries included Egypt, Kenya, South Africa, Uganda and Sudan. † Liberia, 45; Ivory Coast, 10; Guinea, 7; Sierra Leone, 3. Transit countries included Guinea, Ivory Coast, Sierra Leone and Ghana. ‡ Tanzania,11; Burundi, 9; Rwanda, 3. Transit countries included Rwanda and Tanzania. |
|||||||||||||||
2 The most frequent laboratory tests performed by general practitioners seeing newly arrived African refugees in Melbourne, 2005
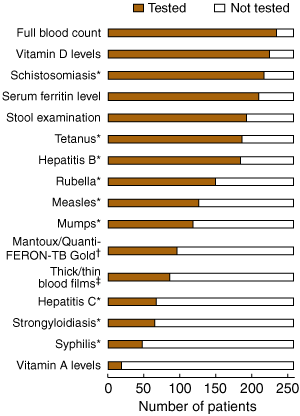
* Serological tests. † For tuberculosis. ‡ For malaria.
3 Proportion of refugee patients with positive laboratory test results by region of birth in Africa (number testing positive/total number tested)
|
Region of birth in Africa |
||||||||||||||
|
Western |
Central |
Eastern |
All |
|||||||||||
Total patients |
65 |
23 |
170 |
258 |
|||||||||||
Condition |
|
|
|
|
|||||||||||
Malaria (positive immunochromatographic test or thick/thin blood films) |
4/43 (9%) |
4/19 (21%) |
1/23 (4%) |
9/86 (10%) |
|||||||||||
Tuberculosis (Mantoux > 9 mm, or positive QuantiFERON-TB Gold test result) |
4/28 (14%) |
0/1 (0) |
20/67 (30%) |
24/96 (25%) |
|||||||||||
Vitamin D deficiency (25OH-Vit D < 37 nmol/L)* |
0/44 (0) |
2/22 (9%) |
63/158 (40%) |
65/225 (29%) |
|||||||||||
Vitamin A deficiency (serum retinol < 0.7 µmol/L) |
0/11 (0) |
None tested |
0/8 (0) |
0/19 (0) |
|||||||||||
Anaemia (based on haemoglobin reference ranges according to age and sex) |
11/58 (19%) |
4/23 (17%) |
30/154 (19%) |
45/235 (19%)§ |
|||||||||||
Iron deficiency (based on ferritin reference ranges according to age and sex) |
8/43 (19%) |
2/21 (10%) |
33/136 (24%) |
43/210 (20%)¶ |
|||||||||||
Gastrointestinal infection (pathogenic organism isolated from stools) |
5/36 (14%) |
2/17 (12%) |
23/140 (16%) |
30/193 (16%)** |
|||||||||||
Schistosomiasis (IgG > 1:32)*† |
16/51 (31%) |
5/21 (23%) |
6/145 (4%) |
27/217 (12%) |
|||||||||||
Strongyloidiasis (optical density > 0.5) |
4/39 (10%) |
1/6 (17%) |
1/21 (5%) |
6/66 (9%) |
|||||||||||
Hepatitis B carrier (surface-antigen positive)* |
9/47 (19%) |
0/19 (0) |
6/118 (5%) |
15/184 (8%) |
|||||||||||
Hepatitis C (IgG positive)‡ |
— |
— |
— |
1/68 (1%) |
|||||||||||
Syphilis (reactive Treponema enzyme immunoassay) |
2/23 (9%) |
0/1 (0) |
2/24 (8%) |
4/48 (8%) |
|||||||||||
* Significant differences (P < 0.05) found between regions of birth. † The Dade Behring Cellognost-Schistosomiasis H test detects both Schistosoma haematobium and S. mansoni. ‡ Regional differences are not described to preserve confidentiality. § 11/235 patients (5%) had haemoglobin levels < 100 g/L. ¶ 19/210 patients (9%) had ferritin levels < 12 μg/L. ** 23 Giardia lamblia, 4 Hymenolepis spp., 1 Shigella flexneri, 1 hookworm, and 1 Cryptosporidium spp. |
|||||||||||||||
4 The 10 most common problems identified by general practitioners in newly arrived African refugee patients in Melbourne, 2005
5 Newly arrived African refugee patients who were tested and immune (positive IgG) to vaccine-preventable diseases
|
Age |
||||||||||||||
|
1–14 years |
15 years and over |
|||||||||||||
Disease |
Total tested |
Immune |
Total tested |
Immune |
|||||||||||
Measles |
64 |
36 (56%) |
61 |
58 (95%) |
|||||||||||
Mumps |
61 |
37 (60%) |
56 |
47 (84%) |
|||||||||||
Rubella |
69 |
54 (78%) |
79 |
76 (96%) |
|||||||||||
Tetanus |
96 |
50 (52%) |
89 |
42 (47%) |
|||||||||||
Hepatitis B* |
87 |
23 (26%) |
96 |
58 (60%) |
|||||||||||
* Immune patients were hepatitis B surface antibody (sAb) positive, surface antigen (sAg) negative and core antibody (cAb) negative; sAb positive, sAg negative, and cAb positive; or sAb negative, sAg negative, and cAb positive. |
|||||||||||||||
- Albert C D Tiong1
- Mahomed S Patel1
- Joanne Gardiner2
- Rowena Ryan2
- Karen S Linton3
- Kate A Walker3,4
- John Scopel5
- Beverley-Ann Biggs4,6
- 1 National Centre for Epidemiology and Population Health, Australian National University, Canberra, ACT.
- 2 Darebin Community Health Centre, Melbourne, VIC.
- 3 Western Region Health Centre, Melbourne, VIC.
- 4 Department of General Practice, The University of Melbourne, Melbourne, VIC.
- 5 The Clinic Footscray, Melbourne, VIC.
- 6 Department of Medicine and the Centre for Clinical Research Excellence in Infectious Diseases, and the Victorian Infectious Diseases Service, University Of Melbourne, Royal Melbourne Hospital, Melbourne, VIC.
This research was funded by the Victorian Government Department of Human Services Victoria. We thank Dr Rosemary Lester for providing departmental support, Dr Jim Black and Dr Chris Lemoh for providing advice on study design, and Dr Trevor Adcock for data collection.
None identified.
- 1. Refugee health and wellbeing action plan. Melbourne: Policy Projects Branch, Victorian Government Department of Human Services, 2005. http://www.dhs.vic.gov.au/multicultural/html/refugee_action.htm (accessed Nov 2006).
- 2. Harris M, Zwar N. Refugee health. Aust Fam Physician 2005; 34: 825-829.
- 3. Murray SB, Skull SA. Hurdles to health: immigrant and refugee health care in Australia. Aust Health Rev 2005; 29: 25-29.
- 4. Medicare Benefits Schedule (items 714 and 716). Canberra: Australian Government Department of Health and Ageing, 2006: 109. http://www.health.gov.au/internet/wcms/publishing.nsf/Content/mbsonline-downloads (accessed Nov 2006).
- 5. Lifson AR, Thai D, O’Fallon A, et al. Prevalence of tuberculosis, hepatitis B virus, and intestinal parasitic infections among refugees to Minnesota. Public Health Rep 2002; 117: 69-77.
- 6. Caruana S, Kelly H, Ngeow J, et al. Undiagnosed and potentially lethal parasite infections among immigrants and refugees in Australia. J Travel Med 2006; 13: 233-239.
- 7. Rice JE, Skull SA, Pearce C, et al. Screening for intestinal parasites in recently arrived children from East Africa. J Paediatr Child Health 2003; 39: 456-459.
- 8. Victorian Foundation for Survivors of Torture on behalf of the Western Melbourne Division of General Practice. Caring for refugee patients in general practice. Melbourne: Victorian Foundation for Survivors of Torture, 2002.
- 9. Zweck N, Spencer E, Anstey N, et al. Guidelines for screening and management of infectious diseases in refugees from sub-Saharan Africa — based on a single initial blood sample after arrival in the Northern Territory. Northern Territory Dis Control Bull 2004; 11: 13-17.
- 10. ICPC-2 International classification of primary care. 2nd ed. Prepared by the International Classification Committee of WONCA. Oxford: Oxford University Press, 1998.
- 11. Okkes I, Jamoulle M, Lamberts H, et al. ICPC-2-E: the electronic version of ICPC-2. Differences from the printed version and the consequences. Fam Pract 2000; 17: 101-107.
- 12. Burnett A, Peel M. Health needs of asylum seekers and refugees. BMJ 2001; 322: 544-547.
- 13. Gilbert GL, Escott RG, Gidding HF, et al. Impact of the Australian Measles Control Campaign on immunity to measles and rubella. Epidemiol Infect 2001; 127: 297-303.
- 14. Phillips C, Patel P. The switch to new conjugated vaccines may compromise immunisation coverage for refugees [letter]. Med J Aust 2006; 184: 473. <MJA full text>
- 15. Pai M, Riley LW, Colford JM Jr. Interferon-gamma assays in the immunodiagnosis of tuberculosis: a systematic review. Lancet Infect Dis 2004; 4: 761-776.
- 16. Smieja MJ, Marchetti CA, Cook DJ, et al. Isoniazid for preventing tuberculosis in non-HIV infected persons. Cochrane Database Syst Rev 2000; (2): CD001363.
- 17. Management, control and prevention of tuberculosis: guidelines for health care providers (2002–2005). Melbourne: Rural and Regional Health and Aged Care Services Division, Victorian Government Department of Human Services, 2002. http://www.health.vic.gov.au/ideas/diseases/tb_mgmt_guide.htm#intro (accessed Jun 2006).
- 18. American Thoracic Society, Centers for Disease Control and Prevention. Targeted tuberculin testing and treatment of latent tuberculosis infection. Am J Respir Crit Care Med 2000; 161 (4 Pt 2): S221-S247.
- 19. Medicare benefits schedule (item 69471). Canberra: Australian Government Department of Health and Ageing, 2006: 642. http://www.health.gov.au/internet/wcms/publishing.nsf/Content/mbsonline-downloads (accessed Nov 2006).
- 20. Mahmoud A. Trematodes (schistosomiasis) and other flukes. In: Mandell GL, Bennett J, Dolin R, editors. Principles and practice of infectious diseases. 5th ed. Philadelphia: Churchill Livingstone, 2000: 2950-2953.
- 21. Mahmoud A. introduction to helminth infections. In: Mandell GL, Bennett J, Dolin R, editors. Principles and practice of infectious diseases. 5th ed. Philadelphia: Churchill Livingstone, 2000: 2948-2943.
- 22. MIMS annual 2005. Sydney: Medimedia Australia, 2005.
- 23. Speare R, Durrheim DN. Strongyloides serology — useful for diagnosis and management of strongyloidiasis in rural Indigenous populations, but important gaps in knowledge remain. Rural Remote Health 2004; 4: 264.
- 24. Davidson N, Skull S, Chaney G, et al. Comprehensive health assessment for newly arrived refugee children in Australia. J Paediatr Child Health 2004; 40: 562-568.
- 25. Shaw NJ, Pal BR. Vitamin D deficiency in UK Asian families: activating a new concern. Arch Dis Child 2002; 86: 147-149.
- 26. Plotnikoff GA, Quigley JM. Prevalence of severe hypovitaminosis D in patients with persistent, nonspecific musculoskeletal pain. Mayo Clin Proc 2003; 78: 1463-1470.





Abstract
Objective: To identify the most common health issues diagnosed by general practitioners in newly arrived African refugees.
Design: Descriptive study based on a purposive sample of six GPs to collate data from medical records of patients from African countries who had attended their clinics for the first time between 1 January and 30 June 2005.
Setting: Two community health centres and two private general practices in metropolitan Melbourne.
Participants: African refugee patients who arrived in Australia after 1 June 2004 and were seen by the six participating GPs between 1 January and 30 June 2005.
Main outcome measures: Demographic characteristics, laboratory test results and final diagnoses.
Results: Data were collected from 258 patient files. Most patients were from Sudan (57%) or Liberia (17%). Half were aged under 15 years. The most common health problems identified were inadequate vaccinations, nutritional deficiencies (vitamin D and iron), infectious diseases (gastrointestinal infections, schistosomiasis, and latent tuberculosis) and dental disease. Musculoskeletal, psychological and social problems were common in adults. 37% of patients were tested for latent tuberculosis, and 25% of these tested positive.
Conclusions: African refugees require comprehensive health assessments for undiagnosed and untreated health problems. While most of the common diseases identified are non-communicable, if left untreated they will affect the long-term health and productivity of new settlers.