The in-hospital mortality rate for patients admitted to the intensive care unit (ICU) is high, with estimates ranging from 20%–30%.1-3 Of these deaths, around a third can occur after discharge from the ICU. Some of these deaths are inevitable, with severity of illness the major determinant.1 A previous study by Goldfrad and Rowan noted that patients discharged from the ICU after-hours were more likely to die than those discharged during normal working hours.4 Furthermore, after-hours discharges had increased over a 10-year period and were more likely to be premature; the authors attributed this rise to increased pressure on ICU beds.4
These were defined by variables relating to nursing shift and hospital medical officers’ hours. There were three nursing shifts in each 24-hour period, with some overlap for handover. For our analysis, the three nursing shifts were defined as 07:00–14:59 (morning shift), 15:00–21:59 (afternoon shift) and 22:00–06:59 (night shift). Working hours for hospital medical officers (interns, junior medical officers and registrars) are generally 08:00–18:00 Monday to Friday, with a reduced number of staff between 18:00–08:00 and during weekends and on public holidays. For our analysis, out-of-hours for doctors (medical officer nights) was defined as 18:00–07:59, and medical officer weekends as Friday 18:00 to Monday 07:59. No attempt was made to account for gazetted public holidays in Melbourne each year. An analysis using the night (22:00–06:59) and early hours (00:00–04:59) discharges defined by Goldfrad and Rowan4 was also undertaken for comparison.
Severity of illness was estimated by APACHE II (Acute Physiology and Chronic Health Evaluation) score,5 in which points are accrued for abnormal clinical and blood test parameters, increasing age, defined chronic conditions, and emergency surgery, to a maximum of 71. Risk of in-hospital mortality increases with higher APACHE II scores. APACHE II scores were not calculated for short-stay admissions (< 12 hours), although they were calculated for the small number of patients under 18 years of age.
The primary outcome measure was hospital mortality after discharge from the ICU. For continuous variables, results are expressed as median (range), as none were normally distributed. Number and percentage are reported for categorical variables. Univariate analyses included the Mann–Whitney U test for continuous variables, and χ2 or Fisher’s exact test for categorical variables. Logistic regression analysis6 was used to calculate adjusted odds ratios, and stepwise logistic regression (backward and forward elimination) was used for the multivariate analysis. P < 0.05 was used to indicate statistical significance and all calculations were two-sided. Analyses were performed using Stata (version 8, StataCorp, College Station, Tex, USA).
Over the 11-year period from 1 January 1992 to 31 December 2002, there were 12 693 discharges from the ICU. By including only the last admission for multiple ICU episodes, 12 079 individual ICU admissions remained. Patient characteristics are given in Box 1.
The patient profile over the 11-year period changed between the 1992–1994 dataset and that of 2000–2002 (Box 2). Similar patient numbers were treated over the two periods (3093 versus 3176) with similar proportions of males in each group (66% versus 64%). However, the median age increased and the percentage of cardiac surgical patients decreased. The patients tended to be sicker, but ICU stay was shorter. The origin of patients also changed with more patients being admitted from the emergency department and from interhospital transfers. Medical patients made up a higher proportion of the latter group. Hospital mortality rates were not significantly different (Box 2).
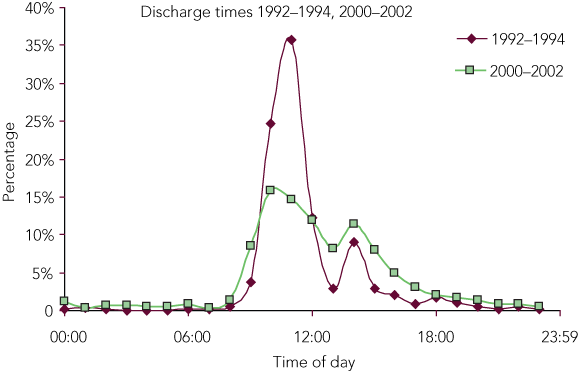
The pattern of discharge also changed between the two periods. Discharges in 2000–2002 had morning and afternoon peaks compared with a major morning peak in 1992–1994. Discharges were more likely to occur in the afternoon and overnight in 2000–2002 than in 1992–1994 (Box 3 and Box 4), during medical officer after-hours and medical officer weekend shifts.
Univariate predictors of mortality after discharge from ICU are shown in Box 5. Category of admission includes an assessment of elective or emergency requirement for ICU, renal replacement therapy and discharge times.
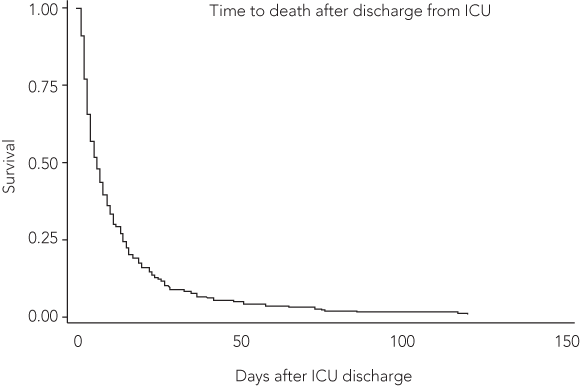
In a multiple logistic model, independent predictors were APACHE II score, origin of admission (operating room, general wards), category of admission (cardiac surgery, postoperative monitoring) and nursing shift. The inclusion of category of admission led to a change in effect for origin of admission (odds ratio, 0.22–1.47) which now represented high-risk emergency patients from the operating theatre. Nursing shift was a better predictor than models with medical officer after-hours or medical officer weekend shifts included. A Kaplan–Meier survival analysis (Box 6) showed that 25% of the observed mortality occurred within 3 days of intensive care discharge, 50% by Day 6 and 75% by Day 14.
Our study shows that, over an 11-year period at our institution, severity of illness on ICU admission increased, afternoon and evening or night discharges became more frequent, and such discharges were associated with a statistically significant increase in mortality. A quarter of these deaths occurred within the first 3 days after ICU discharge. These findings are in keeping with studies from the United Kingdom4,7 and previous Australian studies,8,9 but extend the period of risk to the afternoon shift.
There was an increase in the odds of afternoon and night discharges from the ICU in 2000–2002, and discharges were more evenly spread through the day, with morning and afternoon peaks, compared with the large morning peak in 1992–1994. This change may reflect delayed discharge due to a lack of available ward beds.10 This is more likely to occur now as hospitals have significantly downsized and attempted to maximise bed usage with day-of-admission surgery, for which postoperative beds are not guaranteed. Alternatively, the discharge pattern may be due to a demand for ICU beds in excess of supply, so that the unplanned discharge of the “least sick or least needy patient” will occur to accommodate an emergency admission.
Afternoon shift discharges were 3.87 times and night shift discharges 5.47 times more likely to occur in the 2000–2002 period. Goldfrad and Rowan4 analysed two databases (10 806 entries spanning 1988–1990 and 22 089 entries for 1995–1998) of admissions to a number of UK intensive care units. They found that night discharges (22:00–06:59) increased from 2.7% to 6.0%, proportions very similar to our own. They attributed this to increasing demand — the result of clinicians identifying more patients as being suitable for ICU, and rising rates of surgical procedures.
Night and afternoon discharges were associated with higher post-ICU mortality. Furthermore, our estimate of mortality risk for night discharge (1.63; 95% CI, 1.03–2.57) is very similar to that observed by Goldfrad and Rowan4 (1.33; 95% CI, 1.06–1.65), Beck et al7 (1.7; 95% CI, 1.28–2.25) and Duke et al9 (1.7; 95% CI, 1.03–2.9).
Several factors might explain these results. Transfer from the ICU to a ward is associated with a significant reduction in clinical observation and monitoring, with the ratio of nurses to patients varying from 1: 4 to 1: 10. There are also fewer medical staff on the wards, with the least numbers of nurses and doctors at night. Several investigators have shown that increasing severity of illness or dependency of care at time of ICU discharge adversely affects hospital mortality.1,7,11,12 Our finding that afternoon discharges are also associated with poorer outcomes suggests that longer medical and nursing observation of newly discharged patients during the day may reduce complications.
As previously noted, increased mortality of patients discharged at night may reflect premature discharge of patients with residual organ dysfunction who still require a greater level of nursing care than the wards can provide. While studies of post-ICU mortality support this hypothesis,1,11,12 we do not have enough information about the patients who died to be certain. A proportion of patients discharged at night may be those for whom continued ICU care is judged futile, or for whom palliative care has been instituted, as they are not expected to survive to hospital discharge. Such patients would be discharged before patients receiving active care, and their discharge may have skewed the mortality rates when defined by nursing shift. Limitation of treatment orders are strongly associated with mortality after ICU discharge,9,13 although, in a previous study, late discharge was still associated with increased mortality when limitation of treatment orders was taken into account.9
Thus, it seems likely that hospital “systems”, such as nursing and medical staff numbers, play an important role in the outcome of patients with more severe illness (eg, those discharged from ICU). Changing hospital systems might reverse this negative association, and there is some evidence to support this hypothesis. More intensive nursing is one strategy. Beck et al were able to show reduced mortality for ICU patients discharged to a high dependency unit compared with those sent to general wards.7 Critical Care Outreach Teams, as implemented in the UK, have been shown, in a number of studies, to reduce mortality, ICU admissions, and adverse events.14-16 The role of medical emergency teams in Australia17,18 needs to be defined; otherwise, health institutions will be pressured to increase ICU beds to meet increased demand and the need to hold patients for longer periods.
1 Characteristics of patients (n = 12 079) admitted to the intensive care unit (ICU), 1992–2002
Emergency department or interhospital transfer; operating or recovery room; ward |
|||||||||||||||
General medicine; special medicine; general surgery; special surgery |
|||||||||||||||
2 Comparison of patients admitted to the intensive care unit (ICU), 1992–1994 and 2000–2002
Emergency department or interhospital transfer; operating or recovery room; ward |
|||||||||||||||
General medicine; special medicine; general surgery; special surgery |
|||||||||||||||
3 Percentages and odds ratios (95% CIs) for discharge (alive) from the intensive care unit in the two periods, 1992–1994 and 2000–2002
Received 27 July 2005, accepted 14 February 2006
- Antony E Tobin1
- John D Santamaria2
- Intensive Care Unit, St Vincent's Hospital, Melbourne, VIC.
None identified.
- 1. Moreno R, Miranda DR, Matos R, Fevereiro T. Mortality after discharge from intensive care: the impact of organ system failure and nursing workload use at discharge. Intensive Care Med 2001; 27: 999-1004.
- 2. Uusaro A, Kari A, Ruokonen E. The effects of ICU admission and discharge times on mortality in Finland. Intensive Care Med 2003; 29: 2144-2148.
- 3. Wallis CB, Davies HT, Shearer AJ. Why do patients die on general wards after discharge from intensive care units? Anaesthesia 1997; 52: 9-14.
- 4. Goldfrad C, Rowan K. Consequences of discharges from intensive care at night. Lancet 2000; 355: 1138-1142.
- 5. Knaus WA, Draper EA, Wagner DP, Zimmerman JE. APACHE II: a severity of disease classification system. Crit Care Med 1985; 13: 818-829.
- 6. Hosmer DW, Lemeshow S. Applied logistic regression. John Wiley & Sons, 1989: 305.
- 7. Beck DH, McQuillan P, Smith GB. Waiting for the break of dawn? The effects of discharge time, discharge TISS scores and discharge facility on hospital mortality after intensive care. Intensive Care Med 2002; 28: 1287-1293.
- 8. Bristow PJ. Discharges out of hours are associated with increased mortality [abstract]. Anaesth Intensive Care 2003; 31: 222.
- 9. Duke GJ, Green JV, Briedis JH. Night-shift discharge from intensive care unit increases the mortality-risk of ICU survivors. Anaesth Intensive Care 2004; 32: 697-701.
- 10. Williams T, Leslie G. Delayed discharges from an adult intensive care unit. Aust Health Rev 2004; 28: 87-96.
- 11. Azoulay E, Adrie C, De Lassence A, et al. Determinants of postintensive care unit mortality: a prospective multicenter study. Crit Care Med 2003; 31: 428-432.
- 12. Smith L, Orts CM, O’Neil I, et al. TISS and mortality after discharge from intensive care. Intensive Care Med 1999; 25: 1061-1065.
- 13. Iapichino G, Morabito A, Mistraletti G, et al. Determinants of post-intensive care mortality in high-level treated critically ill patients. Intensive Care Med 2003; 29: 1751-1756.
- 14. Goldhill DR. Preventing surgical deaths: critical care and intensive care outreach services in the postoperative period. Br J Anaesth 2005; 95: 88-94.
- 15. Department of Health (UK). Comprehensive Critical Care: a review of adult critical care services. Available at: http://www.dh.gov.uk/PublicationsAndStatistics/Publications/PublicationsPolicyAndGuidance (accessed Feb 2006).
- 16. Intensive Care Society. Guidelines for the introduction of outreach services. Available at: http://www.ics.ac.uk/downloads/icsstandards-outreach.pdf (accessed Feb 2006).
- 17. Hillman K, Chen J, Cretikos M, et al. Introduction of the medical emergency team (MET) system: a cluster-randomised controlled trial. Lancet 2005; 365: 2091-2097.
- 18. Lee A, Bishop G, Hillman KM, Daffurn K. The Medical Emergency Team. Anaesth Intensive Care 1995; 23: 183-186.





Abstract
Objective: To investigate the change in pattern of discharge of patients from an intensive care unit (ICU) to hospital wards and to determine the impact of discharge time on subsequent hospital mortality.
Design and participants: A retrospective cohort study of 10 903 patients discharged alive from a single ICU between 1 January 1992 and 31 December 2002.
Main outcome measure: In-hospital mortality.
Results: Of the 10 903 patients discharged alive from the ICU, 486 (4.5%) died in hospital wards. When discharge times were categorised according to nursing shift (morning, 07:00–14:59; afternoon, 15:00–21:59; and night, 22:00–06:59), patients were more likely to be discharged on an afternoon shift (odds ratio, 3.63; 95% CI, 3.05–4.30) or night shift (4.52; 95% CI, 3.15–6.64) in 2000–2002 compared with 1992–1994. In a multiple logistic model, hospital mortality after discharge from the ICU was increased by higher APACHE II score (1.14; 95% CI, 1.12–1.16); admission to ICU from the operating room (1.47; 95% CI, 1.11–1.95) and from the general ward (1.75; 95% CI, 1.37–2.23); and discharge during the afternoon (1.36; 95% CI, 1.08–1.70) and night shifts (1.63; 95% CI, 1.03–2.57).
Conclusion: Over an 11-year period, more patients are being discharged from the ICU in the afternoon and night suggesting increasing pressure on ICU beds. Patients discharged on these shifts have an increased risk of death.