To the Editor: We are concerned that the Northern Territory’s regulations on kava have not succeeded in controlling its availability in Arnhem Land (the north-eastern region of the NT). Under the Kava Management Act 1998 (NT), one wholesaler is licensed to supply kava (Piper methysticum Forst. f.) to four licensed retailers in Arnhem Land Aboriginal communities.1
“Kava Management Plans” in “Kava Licence Areas” permit retailers to supply 600–800 g per week of kava powder to each purchaser1 — more than double the known harmful consumption levels (240–440 g per week).2 Legal kava supplied will reach 26 tonnes in 2005 (worth $3.6 million), with a persistent illegal trade adding 8 tonnes, worth perhaps $2 million (Box). Two proposed additional retail licences1 will increase kava’s availability.
Kava’s social and economic effects remain an ongoing concern. The region’s community-controlled health service attributes to kava abuse an accelerated decline in participation in traditional ceremonies and mortuary rites in some localities. Kava is the psychoactive substance with greatest impact on the financial resources of communities and individuals in Arnhem Land.2
Kava’s health effects include seizures and extreme weight loss in heavy users (up to 20% of body mass), similar to that seen in anorexia nervosa.4 Extreme weight loss, evident during the 1980s, has re-emerged in the region’s kava users (M W Yunupingu, unpublished observations). Raised total and low-density lipoprotein (LDL) cholesterol levels4 add to unresolved concerns that heavy kava use may be a risk factor for cardiovascular disease and sudden cardiac deaths. Potential immunosuppressive effects are suggested by relative lymphocytopenia in heavy kava users4 and by increased risk of melioidosis.5 Raised levels of liver enzymes (alkaline phosphatase and γ-glutamyltransferase), which reverse after ceasing moderate kava use, should be monitored because of fatal hepatotoxicity documented in users of manufactured kava products available as natural therapies.6
Given the scarcity of substance misuse treatment services in the region, with no effective treatments for kava misuse, controlling supply is the only practical measure to reduce kava-related harms. Tighter controls on kava supply are urgently required while licensees implement promised demand-reduction and harm-minimisation strategies.1 We recommend that:
no further retail licences be granted until kava supply is reduced;
retail licensees supply no more than 440 g per week to individual kava consumers;
quantities permitted to be imported by the wholesaler and supplied to retailers be limited;
kava selling prices be reviewed in the light of trade-offs between higher prices to reduce demand and minimal financial drains on communities;
rigorous enforcement be continued to eliminate illegal kava dealing; and
the Kava Management Act be reviewed to facilitate these changes.
History of kava use and retail value ($ million) on the legal and black markets in Arnhem Land, Northern Territory, since 1982, extrapolated to the end of 2005*
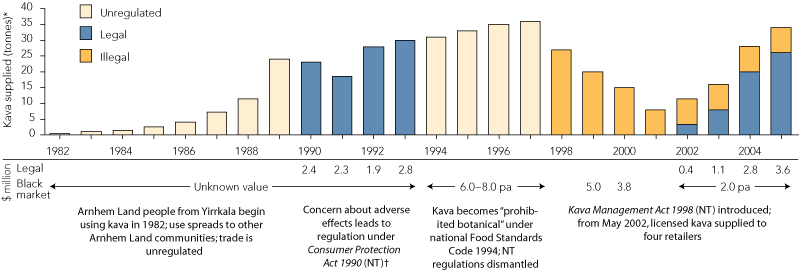
* Data on kava supplied were obtained from d'Abbs (for 19821993);3 were estimated from population surveys of kava use (for 19941997); were estimated from kava seized by Police and Licensing Inspectors (for illegal use, 19982005) (seizures were estimated to account for 14% of the illegal kava supplied, based on correlation with population surveys in 1999 and 2000); and were based on the licensed wholesalers monthly figures, extrapolated to the end of 2005 (for legal use from May 2002). Retail value was calculated from data on kava supplied and regulated values of $100 per kg (19901993), $140 per kg (20022004) and $150 per kg (2005), or a black market value of $250 per kg.2
† Approval required from Minister for communities to supply kava.
pa = per annum.
- 1. Northern Territory of Australia. Kava. Darwin: Northern Territory Treasury, Racing, Gaming and Licensing Division, 2005. Available at: http://www.nt.gov.au/ntt/licensing/kava.shtml (accessed Sep 2005).
- 2. Clough AR. Enough! or too much. What is “excessive” kava use in Arnhem Land? Drug Alcohol Rev 2003; 22: 43-51.
- 3. d’Abbs P. A review of kava control measures in the Northern Territory. Darwin: Menzies School of Health Research, 1993.
- 4. Clough AR, Jacups SP, Wang Z, et al. Health effects of kava use in an eastern Arnhem Land community. Intern Med J 2003; 33: 336-340.
- 5. Currie BJ, Fisher DA, Howard DM, et al. Endemic melioidosis in tropical northern Australia: a 10-year prospective study and review of the literature. Clin Infect Dis 2000; 31: 981-986.
- 6. Currie BJ, Clough AR. Kava hepatoxicity with Western herbal products: does it occur with traditional kava use? Med J Aust 2003; 178: 421-422. <MJA full text>





The research reported here is supported by a National Health and Medical Research Council Postdoctoral Training Fellowship.
Alan Clough has been a member of the Northern Territory Licensing Commission, the body responsible for licensing kava in the Northern Territory.