An osteoid osteoma is a small, benign bone tumour that occurs in young people. Ninety per cent of cases arise before 30 years of age, with a male predominance. The proximal femur and tibia are the most common sites,1 although occurrence has been reported in most bones. It is characteristically solitary and cortically based, although periosteal and medullary lesions sometimes occur.
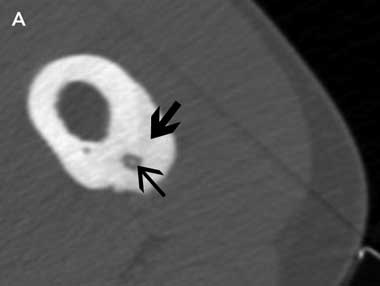
Radiographically, the lesion consists of a small, well rounded radiolucency, the nidus, which is surrounded by dense, reactive new bone. The nidus is often obscured on radiographs by reactive bone. Bone-targeted computed tomography (CT) best delineates the bony architecture. The nidus is characteristically less than 10 mm in maximum diameter and lesions greater than 15–20 mm are usually described as osteoblastomas.2 Technetium-99M HDP bone scintigraphy typically reveals intense focal radiotracer uptake in the lesion. Dynamic contrast CT3 and magnetic resonance imaging help differentiate between osteoid osteoma and infection.
Percutaneous radiofrequency ablation (RFA) for osteoid osteoma was first reported by Rosenthal et al in 1992,4 and has been practised in a number of centres around the world.5-9 Because of the significantly reduced morbidity reported, we adopted this technique in 2000. Here we present our experience to date.
Preoperative evaluation by clinical history and radiological investigation was performed and reviewed in the service’s weekly multidisciplinary clinical conference by surgeons, radiologists and pathologists. In all cases, a nidus with surrounding radiodense bone was demonstrated on a CT scan, and technetium-99M HDP bone scintigraphy showed increased tracer uptake within the lesion. Dynamic contrast imaging was routinely employed to characterise the nidus.3
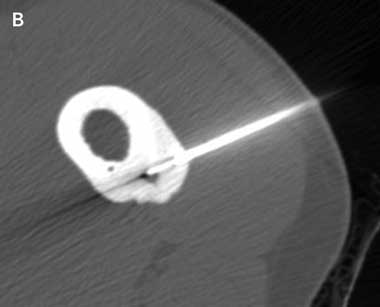
An insulated SMK C-15 electrode (Radionics, Burlington, Mass, USA) was inserted through the Bonopty cannula into the lesion and its position confirmed by CT imaging. Thermal coagulation to 90°C for 6 minutes was achieved using a radiofrequency generator (Radionics RFG-3B, Burlington, Mass, USA) as described by Rosenthal et al4 (Box 1A and Box 1B). If the nidus was larger than 10 mm in maximum diameter, the electrode was repositioned within the nidus and further burns were performed to cover the entire nidus. As the needle entered the nidus and ablation was commenced, elevation of the patients’ resting pulse rate by 10–20 beats/minute was commonly observed. At completion of the procedure an adhesive dressing was applied.
Percutaneous radiofrequency ablation of an osteoid osteoma is a relatively new treatment. Our overall success rate of 96% after multiple treatments compares favourably with the few available reports in the medical literature.5-9
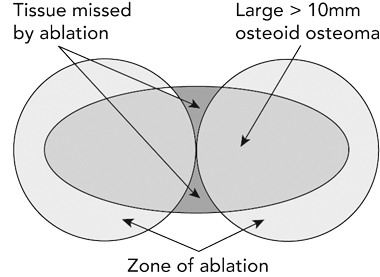
Our recurrence rate of 35% is higher than those in other reports, which range from 5% to 24%.5-9 We observed that recurrence was significantly higher in lesions ≥ 10 mm and five of eight of our patients in this group experienced recurrence. RFA burns an area of about 10 mm in diameter,10 and although lesions greater than 10 mm were burnt in multiple locations, the burn pattern is one of overlapping spheres; it is likely that, in some areas of tumour outside those spheres, ablation was not adequate (Box 2). Woertler et al suggested, on the basis of their patient series, that the larger lesions were more likely to recur.11 Vanderschueren et al reported that multiple needle passes reduced the risk of recurrence and recommended that multiple passes be used in all lesions ≥ 10 mm in maximal diameter.12 We agree with these comments, and with the assertion that recurrences and residual pain are due to viable remnant tissue, rather than a new lesion in situ.6
RFA offers several advantages over other treatments. Previously, surgical excision has been the treatment of choice.13 Precise intra-operative localisation of the nidus is often difficult and en-bloc excision often requires a long incision, extensive dissection and, frequently, internal fixation as a prophylaxis against fracture through the operative site. Intra-articular femoral head lesions may require dislocation of the hip, with attendant risks to the vascular supply. Hospital stay usually exceeds 4 days,5 whereas our patients were discharged within 12 hours. Lower-limb lesions treated by surgical resection often require prolonged periods of protected weight-bearing. Our protocol allowed immediate weight-bearing without restriction.
Failure to locate the lesion at open excision is a well recognised complication,13 but with precise CT-guided localisation of the nidus during RFA, incorrect targeting is much less likely to occur.
Another treatment is CT-controlled laser photocoagulation, which has been reported to give good results, but it requires expensive equipment14 and complicated logistics. CT-guided percutaneous drilling using a large-bore drill has also been described.15 This requires protected weight-bearing after operation. One series reported a complication rate of 24%, including two patients with femoral neck fractures and one with osteomyelitis.15 We have not encountered any postoperative complications to date, and this result concurs with those of other studies.5-9
Histological confirmation of the diagnosis was attempted on all our patients and yielded a firm diagnosis in 41% of patients, a rate higher than obtained by Vanderschueren et al (36%),7 but less than with wide-bore drilling (74%).15 The chief differential diagnosis is subacute osteomyelitis, although we have also found that stress reactions with cyst formation produce similar radiological features. We concur with McGrath et al3 that these lesions can be distinguished from osteoid osteoma preoperatively by performing dynamic contrast CT imaging. As 23 of our 24 patients had a successful outcome, we conclude that failure to provide a tissue diagnosis in every case is not a drawback significant enough to change our practice.
- Richard A Kjar1
- Gerard J Powell1
- Stephen M Schilcht1
- Peter J Smith1
- John Slavin1,2
- Peter F M Choong1,2
- 1 St Vincent's Hospital, Melbourne, VIC.
- 2 University of Melbourne, Melbourne, VIC.
None identified.
- 1. Vigorita VJ. Orthopaedic pathology. Philadelphia: Lippincott Williams and Wilkins, 1999.
- 2. Fechner RE, Mills SE. Tumors of the bones and joints. Washington, DC: Armed Forces Institute of Pathology, 1993.
- 3. McGrath BE, Bush CH, Nelson TE, Scarborough MT. Evaluation of suspected osteoid osteoma. Clin Orthop Relat Res 1996; 327: 247-252.
- 4. Rosenthal DI, Alexander A, Rosenberg AE, Springfield D. Ablation of osteoid osteomas with a percutaneously placed electrode: a new procedure. Radiology 1992; 183: 29-33.
- 5. Rosenthal DI, Hornicek FJ, Wolfe MW, et al. Percutaneous radiofrequency coagulation of osteoid osteoma compared with operative treatment. J Bone Joint Surg Am 1998; 80: 815-821.
- 6. Lindner NJ, Ozaki T, Roedl R, et al. Percutaneous radiofrequency ablation in osteoid osteoma. J Bone Joint Surg Br 2001; 83: 391-396.
- 7. Vanderschueren GM, Taminiau AHM, Obermann WR, Bloem JL. Osteoid osteoma: clinical results with thermocoagulation. Radiology 2002; 224: 82-86.
- 8. Barei DP, Moreau G, Scarborough MT, Neel MD. Percutaneous radiofrequency ablation of osteoid osteoma. Clin Orthop Relat Res 2000; 373: 115-124.
- 9. Ghanem I, Collet LM, Kharrat K, et al. Percutaneous radiofrequency coagulation of osteoid osteoma in children and adolescents. J Pediatr Orthop B 2003; 12: 244-252.
- 10. Tillotson CL, Rosenberg AE, Rosenthal DI. Controlled thermal injury of bone. Report of a percutaneous technique using radiofrequency electrode and generator. Invest Radiol 1989; 24: 888-892.
- 11. Woertler K, Vestring T, Boettner F, et al. Osteoid osteoma: CT-guided percutaneous radiofrequency ablation and follow-up in 47 patients. J Vasc Interv Radiol 2001; 12: 717-722.
- 12. Vanderschueren GM, Taminiau AHM, Obermann WR, et al. Osteoid osteoma: factors for increased risk of unsuccessful thermal coagulation. Radiology 2004; 233: 757-762.
- 13. Sim FH, Dahlin CD, Beabout JW. Osteoid-osteoma: diagnostic problems. J Bone Joint Surg Am 1975; 57: 154-159.
- 14. Gangi A, Dietmann JL, Gasser B, et al. Interstitial laser photocoagulation of osteoid osteomas with use of CT guidance. Radiology 1997; 203: 843-848.
- 15. Sans N, Galy-Fourcade D, Assoun J, et al. Osteoid osteoma: CT-guided percutaneous resection and follow-up in 38 patients. Radiology 1999; 212: 687-692.





Abstract
Objective: To review our experience with percutaneous radiofrequency ablation (RFA) for osteoid osteoma.
Participants, design and setting: Retrospective review of 24 patients with osteoid osteoma treated with percutaneous RFA at St Vincent’s Hospital, Melbourne, from August 2000 to February 2005.
Main outcome measures: Initial response to treatment, return of symptoms, time to recurrence, complications and histopathological correlation.
Results: In 23 of 24 patients, there was immediate relief of symptoms. One-third of patients had a return of symptoms. The 24 patients underwent a total of 32 RFA procedures. Of the eight patients who had a recurrence, five had an initial lesion ≥ 10 mm in maximum diameter. Twenty-three of the 24 patients were pain-free at the end of the study period. Patients were followed up for a median of 26 months. There were no long-term complications.
Conclusions: Percutaneous RFA is a safe and efficacious treatment for osteoid osteoma with a low morbidity rate. Despite recurrence after treatment, re-treatment is simple and effective.