Acute generalised exanthematous pustulosis (AGEP) is a clinical reaction pattern that is induced, in over 90% of cases, by systemic drugs (most frequently antibacterial drugs). This is the first reported case of AGEP caused by the herbal remedy Ginkgo biloba.
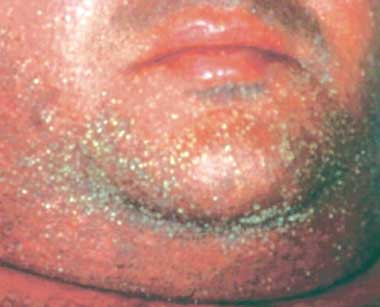
A 45-year-old man developed a symmetrical maculopapular eruption on his limbs. Within 2 days, the rash generalised to involve the face. Disseminated non-follicular small pustules on erythematous skin were predominant (Box 1). The palms, soles and mucous membranes were spared. His body temperature was 38.8°C.
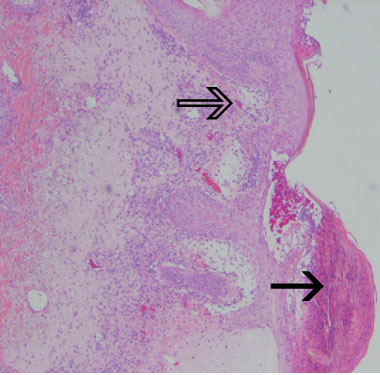
Blood tests returned the following results: C-reactive protein 135 mg/L (reference range [RR], < 12 mg/L); white blood cells 17.2 × 109/L (RR, 4.0–11.0 × 109/L); neutrophils 15.8 × 109/L (RR, 2.0–7.5 × 109/L); and eosinophils 0.79 × 109/L (RR, 0.04–0.40 × 109/L). A skin biopsy showed neutrophil-containing spongiotic pustules in the epidermis and a mixed cellular infiltrate with oedema in the papillary dermis, consistent with acute generalised exanthematous pustulosis (Box 2).
Ginkgo fruits and seeds have been used in traditional Chinese medicine for millennia, mostly to treat asthma and chilblains. The flavonoids and terpene lactones from the ginkgo leaf, which today is used for medicinal purposes, are associated with diverse pharmacological actions. In clinical practice, ginkgo is mostly used to treat memory impairment,1,2 dementia,3 tinnitus4 and intermittent claudication.5 In some European countries, ginkgo is registered for these indications; in the United States, it is marketed as a dietary supplement. In-vitro and in-vivo studies suggest that ginkgo has antioedemic, antihypoxic, antioxidant, metabolic, antiplatelet, haemorrhagic, microcirculatory and free-radical-scavenging actions.6,7
Reported adverse skin reactions to Ginkgo biloba include contact dermatitis and diffuse morbilliform eruption. The primary allergens from the ginkgo leaf appear to be ginkgolic acids. Ginkgolic acid molecules have similar side-chains to the catechols of the poison ivy group and are suspected of displaying some cross-allergenicity.8 Allergic contact dermatitis to the Rhus antigen present in the ginkgo fruit seeds has been extensively documented.8,9 This raises the question of whether or not ginkgolic acids could evoke a systemic reaction if taken orally. Systemic contact dermatitis can develop from oral challenge after contact sensitisation with other allergens.10
The criteria for diagnosis of AGEP are defined by the EuroSCAR score.11 Features of the condition include acute onset of small, sterile, non-follicular pustules on erythematous skin beginning in intertriginous areas or on the face; systemic signs, with fever over 38°C (in a typical course, an acute rash appears within 1–2 days and resolves by skin desquamation within 4–10 days after discontinuation of the causative agent); and neutrophilia, in the setting of a consistent histological pattern.11 The patient described here had a classic drug-induced AGEP with typical morphology, course and histology and a validation score of 12 points (the range for definite AGEP is 8–12).11
- 1. Kleijnen J, Knipschild P. Ginkgo biloba for cerebral insufficiency. Br J Clin Pharmacol 1992; 34: 352-358.
- 2. Hopfenmüller W. [Evidence for a therapeutic effect of Ginkgo biloba special extract. Meta-analysis of 11 clinical studies in patients with cerebrovascular insufficiency in old age] [German]. Arzneimittelforschung 1994; 44: 1005-1013.
- 3. Ernst E, Pittler MH. Ginkgo biloba for dementia: a systematic review of double-blind placebo-controlled trials. Clin Drug Invest 1999; 17: 301-308.
- 4. Ernst E, Stevinson C. Ginkgo biloba for tinnitus: a review. Clin Otolaryngol 1999; 24: 164-167.
- 5. Pittler MH, Ernst E. Ginkgo biloba extract for the treatment of intermittent claudication: a meta-analysis of randomized trials. Am J Med 2000; 108: 276-281.
- 6. Cupp MJ, editor. Toxicology and clinical pharmacology of herbal products. Totowa, NJ: Humana Press, 2000.
- 7. DeFeudis F. Ginkgo biloba extract: pharmacological activities and clinical applications. Amsterdam: Elsevier, 1991.
- 8. Tomb RR, Foussereau J, Sell Y. Mini-epidemic of contact dermatitis from ginkgo tree fruit (Ginkgo biloba L). Contact Dermatitis 1998; 19: 281-283.
- 9. Lepoittevin JP, Benezra C, Asakawa Y. Allergic contact dermatitis to Ginkgo biloba L: relationship with urushiol. Arch Dermatol Res 1989; 281: 227-230.
- 10. Maibach H. Oral substitution in patients sensitized by transdermal clonidine treatment. Contact Dermatitis 1987; 16: 1-8.
- 11. Sidoroff A, Halevy S, Bavinck JN. Acute generalized exanthematous pustulosis (AGEP): a clinical reaction pattern. J Cutan Pathol 2001; 28: 113-119.





I would like to thank Dr Gael Phillips and Dr Anne Stewart for their assistance with the manuscript.
None identified.