The health sector is characterised by complexity, a plurality of players (national and state governments, public and private providers, professions, and consumer groups), rapid change in its environment (eg, new technology and new global diseases), and a recent proliferation of regulatory agencies and regulatory strategies. We argue that “responsive” regulation — the use by regulators of mechanisms that are responsive to the context, conduct and culture of those being regulated — is a promising strategy for improving the quality and safety of patient care. These ideas were elaborated in our discussion paper The governance of health safety and quality, which presents contemporary research on regulatory thinking, including examples from other high-risk industries.1 While definitions of regulation abound, we use the term to mean governance in the broad sense of steering the flow of events, rather than the narrow sense of enforced compliance with rules.2
In the “new regulatory state”, with its increasing privatisation, regulation by government and its agents has become more rather than less common, but the crucial difference from the old “command and control” view is that governments increasingly “steer not row” and are seeking flexible, participatory and devolved forms of regulation. These changes are possible given the changed nature of organisations in the information age, where power flows through networks that are more fluid and complex than older structures of governance.3 This perspective is particularly appropriate in the health sector with its multiple players and where the state generally has left the regulation of health care performance to the medical profession. However, external pressures have been mounting over the past decade for governance systems to insist on more accountability and transparency from health care providers.4 Until recently, most mechanisms intended to improve performance have relied on internal rather than external monitoring; for example, hospitals were left to monitor their own surgical mortality rates. The normative assumption has been that techniques for improving performance rely on voluntary adoption by a professional or an organisation, whether incident reporting or clinical protocols. Walshe argues that the problem with this “soft” approach is that it treats a health provider as if it exists in isolation from its environment, oblivious to the institutional, social and economic pressures that drive organisational willingness to contemplate internal reforms.5 Moreover, the medical profession has traditionally resisted what it sees as encroachment on professional autonomy,6 although, in the United Kingdom at least, it has been claimed that the old system of self-regulation by doctors is dead.7
The public expects a high standard from 21st century health care given the dramatic advances in technology and in medical and surgical knowledge. However, greater scrutiny of performance, and more media interest in its failings, has shaken public confidence. Ongoing medical “scandals” in many countries, including Australia8 — most recently at Bundaberg Hospital9 — mean that the public is no longer confident about leaving safety and quality to doctors and health departments. Realisation has dawned that health care is a risky business, as shown by the Quality in Australian Health Care Study, where re-analysis of data revealed that over 10% of patients experience some type of adverse event in hospital.10 Estimates and consensus from other countries suggest that a large proportion of errors are preventable.11,12
The challenge for safety and quality is to design safer systems and inculcate a culture of safety, while the challenge for governance is to ensure that these systems and practices are actually applied. The argument of responsive regulation is that regulators are more likely to succeed if they use strategies that are responsive to the culture of those being regulated.13 For example, bringing about change in hospitals must take into account professional and organisational cultures,14 as shown in the research on “magnet” hospitals (so termed because of their success in attracting and retaining nursing staff).15 Similarly, designing safer systems must take into account human factors.16
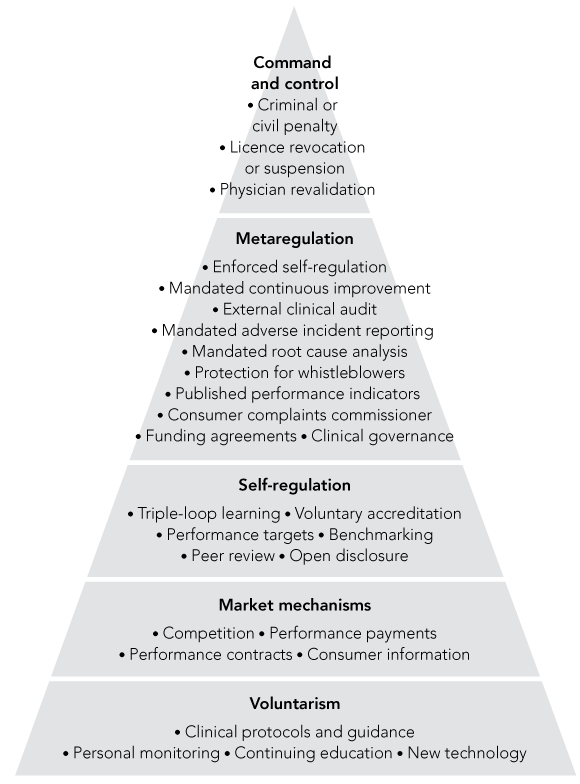
The regulator begins at the base of the pyramid with persuasion;
A single regulatory mechanism is seldom sufficient as the weaknesses of one mechanism must be complemented by the strengths of another; and
There must be the capacity for escalation if persuasion fails.
Box 1 depicts some examples of regulatory mechanisms from the health sector that range upwards in a pyramid from persuasion to punishment — although some mechanisms, such as accreditation, might be voluntary or mandatory depending on the context. Thus, a policy maker or hospital manager could use the pyramid to think responsively in choosing among a range of regulatory options.
The idea of responsive regulation arose out of the actual experiences of private firms. Some argue that business people understand only the “bottom line” profit and therefore must be consistently punished for lawbreaking, while others assert that business people are responsible citizens who can be persuaded. There is truth in both positions, depending on the context. But neither consistent punishment nor consistent persuasion has proven effective in the long term. Consistent punishment is too costly, and consistent cajoling of the incorrigibly unethical or incompetent is naïve. For example, a study of 410 chief executive officers of small health care organisations found that while expected severity of sanctions did not predict compliance, regulators who were consistently “tolerant and understanding” increased non-compliance.17 Although there is no body of evidence that responsive regulation will work in more complex organisations such as hospitals — that remains to be tested — there is a body of evidence that responsive regulation works in settings such as mines and nursing homes, and also in more complex domains, such as the nuclear industry.18
Despite the emergence of new regulatory bodies, commentators in several countries, including the United States19 and Australia,20 are frustrated by an apparent lack of progress. This is despite the large body of widely disseminated research and recommendations over the past 5 years by the Australian Council for Safety and Quality in Health Care, and the host of initiatives underway.21 For example, there are no national published measures of adverse events, despite the beginnings of state-based monitoring of sentinel events. Without some form of standardised reporting, there is no way to benchmark performance and to systematically trace progress.20
A second gap is in innovation through systematic learning, as undertaken in other risky enterprises. For example, this philosophy underwrote the dramatic improvements in the safety of air travel in the 20th century. The aviation industry has systematised error reporting and staff training to produce a safety culture that offers lessons for medicine.22 The challenge for health care is to shift from a blame culture to a learning culture, in order to learn from adverse events.
A metaregulator needs power to enforce self-regulation when an issue requires both the problem-solving creativity of self-regulation, and the assurance that minimum standards are met. This concept underlies the framework of clinical governance introduced into the UK National Health Service (NHS) in the late 1990s.23 The Healthcare Commission, as the metaregulator, now regularly reviews the progress of NHS health providers in implementing quality improvement systems and achieving better outcomes.
Another example of enforced self-regulation is an external requirement that every hospital must implement an infection control plan and plot infection outcomes over time. Innovative hospital administrators are permitted to design their own standards and programs, as long as they make a good case that their standards are better than the default standards.19 Dynamism can be built into enforced self-regulation to prod reluctant managers by requiring continuous improvement. This means that, each year, managers are required to do something to make infection control better than last year. The tough part about enforced self-regulation is that, unlike voluntary self-regulation, it is enforceable. Hence, if a hospital fails to meet one of the privately written but publicly ratified rules, the organisation and its managers can be sanctioned for that failure. This enforced self-regulation strategy attempts to secure the creativity, flexibility, and cost-effectiveness of moving away from command and control, while simultaneously retaining public enforcement capability.
One of the hopes for well crafted meta-risk management is that it will foster creative approaches to continuous improvement. An example is the nursing home industry in the US, where homes were required to identify their greatest health quality problems, choose one to improve each year, design an intervention, and measure the targeted outcome before and after the intervention. Because the level of restraint of elderly people in nursing homes was such an obvious problem (40% of residents nationally were physically restrained), many nursing homes were pushed to target this issue for their quality improvement study. Between 1990 and 2005, the proportion of residents physically restrained dropped from 40% to 4%.24 This was accomplished not by a “command and control” rule, but by the industry’s own management creativity in finding better ways to manage patients without tying them into a chair or bed.
The more complex and dynamic the care process (as in a hospital versus a nursing home), the more relevant a policy of devolution of management decisions closer to the complexities of managing staff and patients. In addition, a regulatory strategy that harnesses management creativity to deliver continuous improvement in quality allows leaders to raise the standards of the whole sector.25
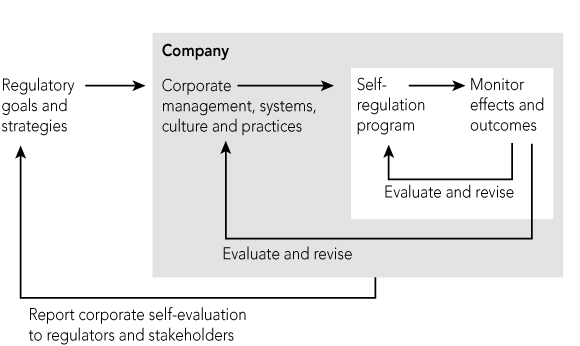
Doctors are no longer expected to rely solely on their own clinical judgement or to apply received wisdom from their teachers (handed down perhaps decades ago), but instead to practise evidence-based medicine, or at least to consult guidelines and clinical protocols as to contemporary best practice. Triple-loop learning (Box 2) takes voluntary regulation some steps further.26
The first loop occurs when self-regulatory innovators monitor their effectiveness at improving an outcome. An example is a personal learning model using portable digital technology to enable reflective practice.27,28 The second loop occurs when policy learning is monitored by clinicians and senior managers who, in response, change their management systems, culture and practices. An example is the research collaborations between health providers that support the creativity of professionals in solving their own quality problems.29 The third loop occurs when a regulator (such as an accreditation agency or a health department) learns from monitoring the organisation’s double-loop learning and revises its regulatory goals for the whole field.
The slow translation of evidence into practice by doctors30 has prompted the establishment of dissemination bodies, such as the National Institute of Clinical Studies, which hope to improve the take-up in clinical practice of the best available evidence — in other words, to promote triple-loop learning. Until now, the movement from single to double to triple loops of learning has not been systematic.
- Judith Healy1
- John Braithwaite2
- Regulatory Institutions Network, Research School of Social Sciences, Australian National University, Canberra, ACT.
This article is based partly on our discussion paper,1 funded by the Australian Council for Safety and Quality in Health Care. We thank the reviewers for their comments on an earlier version of this article.
None identified.
- 1. Braithwaite J, Healy J, Dwan K. The governance of health safety and quality. Canberra: Commonwealth of Australia, 2005.
- 2. Black J. Critical reflections on regulation. Aust J Legal Philos 2002; 27: 1-36.
- 3. Castells M. The rise of the network society. Melbourne: Blackwell Publishing, 2000.
- 4. McKee M, Healy J, editors. Hospitals in a changing Europe. European Observatory on Health Care Systems Series. Buckingham, UK: Open University Press, 2002.
- 5. Walshe K. Regulating healthcare: a prescription for improvement? Maidenhead, UK: Open University Press, 2003.
- 6. Friedson E. Professionalism reborn: theory, prophecy and policy. Chicago: University of Chicago Press, 1994.
- 7. Smith R. Managing the clinical performance of doctors. A coherent response to an intractable problem [editorial]. BMJ 1999; 319: 1314-1315.
- 8. Faunce TA, Bolsin SN. Three Australian whistleblowing sagas: lessons for internal and external regulation. Med J Aust 2004; 181: 44-47. <MJA full text>
- 9. Van Der Weyden M. The Bundaberg Hospital scandal: the need for reform in Queensland and beyond [editorial]. Med J Aust 2005; 183: 284-285. <MJA full text>
- 10. Runciman WB, Webb RK, Helps SC, et al. A comparison of iatrogenic injury studies in Australia and the USA. II: Reviewer behaviour and quality of care. Int J Qual Health Care 2000; 12: 379-388.
- 11. Institute of Medicine. To err is human: building a safer health system. Washington, DC: National Academy Press, 2000.
- 12. Department of Health. An organisation with a memory. Report of an expert group on learning from adverse events in the NHS. London: The Stationery Office, 2000.
- 13. Braithwaite J. Restorative justice and responsive regulation. Oxford: Oxford University Press, 2002.
- 14. Braithwaite J, Westbrook MT, Iedema R, et al. A tale of two hospitals: assessing cultural landscapes and compositions. Soc Sci Med 2005; 60: 1149-1162.
- 15. Aiken L, Sloane D. Hospital organization and culture. In: McKee M, Healy J, editors. Hospitals in a changing Europe. Buckingham, UK: Open University Press, 2002: 265-278.
- 16. Reason J. Managing the risks of organizational accidents. Aldershot, UK: Ashgate Publishing, 1997.
- 17. Braithwaite J, Makkai T. Testing an expected utility model of corporate deterrence. Law Soc Rev 1991; 25: 7-40.
- 18. Braithwaite J. Restorative justice and responsive regulation. Oxford: Oxford University Press, 2002: 17-71.
- 19. Leape LL, Berwick DM. Five years after “To err is human”: what have we learned? JAMA 2005; 293: 2384-2390.
- 20. Wilson R McL, Van Der Weyden MB. The safety of Australian healthcare: 10 years after QAHCS [editorial]. Med J Aust 2005; 182: 260-261. <MJA full text>
- 21. Australian Council for Safety and Quality in Health Care. Achieving safety and quality improvements in health care: sixth report to the Australian Health Ministers’ Conference. Canberra: Commonwealth of Australia, 2005.
- 22. Helmreich RL, Merritt AC. Culture at work in aviation and medicine: national, organizational and professional influences. Aldershot, UK: Ashgate Publishing, 1998.
- 23. Scally G, Donaldson LL. The NHS’s 50th anniversary. Clinical governance and the drive for quality improvement in the new NHS in England. BMJ 1998; 317: 61-65.
- 24. Centers for Medicare and Medicaid Services. Nursing home data compendium 2005. Baltimore, USA: Centers for Medicare and Medicaid Services, Department of Health and Human Services, 2005.
- 25. Gunningham N, Sinclair D. Leaders and laggards: next-generation environmental regulation. Sheffield, UK: Greenleaf Publishing, 2002.
- 26. Parker C. The open corporation: effective self-regulation and democracy. Cambridge: Cambridge University Press, 2002.
- 27. Bolsin S, Faunce T, Colson M. Using portable digital technology for clinical care and critical incidents: a new model. Aust Health Rev 2005; 29: 297-305.
- 28. Bolsin S, Patrick A, Colson M, et al. New technology to enable personal monitoring and incident reporting can transform professional culture: the potential to favourably impact the future of health care. J Eval Clin Pract 2005; 11: 499-506.
- 29. Australian Council for Safety and Quality in Health Care. Medication Safety Breakthrough Collaborative team showcase. Canberra: Commonwealth of Australia, 2005.
- 30. Freemantle N. Optimizing clinical performance. In: McKee M, Healy J, editors. Hospitals in a changing Europe. Buckingham, UK: Open University Press, 2002: 252-264.





Abstract
Self-regulation by the health professions, while improving, is no longer enough; external drivers for safer health care include governments, funders and consumers.
Enforced self-regulation is often more promising than a “command and control” strategy.
Research evidence on the responsive regulatory pyramid and its options offers lessons for health care policy makers and managers.
Start at the base of the regulatory pyramid — try persuasion first; move up the pyramid to secure compliance, and then be willing to move back down.
Use existing capacities and structures, and if possible avoid new bureaucracies of control.