After the use of adjuvanted diphtheria, tetanus and pertussis vaccines was established in the 1940s, one of the pioneers in this area published a detailed outline of a vaccination practice that minimised severe local adverse effects.1 This involved deep injection into the lateral gluteal muscle mass with a 25 gauge, 1/2 inch or 5/8 inch needle, terminating each dose with 0.1 mL of air, the latter to prevent vaccine draining back along the injection tract into the subcutaneous tissue. Subsequently, vaccination techniques have been modified in response to reports of vaccination experience in the medical literature.
The gluteal site of injection was abandoned in favour of the anterolateral thigh because of the risk of sciatic nerve injury with viscous agents like penicillin.2 However, vaccine-induced sciatic nerve injury has never been reported, despite international canvassing for cases by MacDonald and Marcuse.3
Angling of the needle to the long axis of the thigh at muscle entry, as in techniques recommended by United States4 and Australian5 vaccine advisory groups, is related to a report by Talbert et al of gangrene of the foot in an infant after intramuscular injection of penicillin, with the needle entering the thigh muscle at 90° to the long axis of the femur.6 No neurovascular adverse reactions have been reported after vaccination with needle entry at this angle in the thigh.
The recommendation for the use of 23 gauge, 25 mm long needles for anterolateral thigh intramuscular injection derives from a study by Hick et al, who measured subcutaneous layer thickness in 4-month-old infants, concluding that a 16 mm long needle introduced at an angle of 45° would penetrate muscle in only 5/24 subjects (21%).7 Unfortunately, the results of this very small study have not been reproduced. Two other similar studies in 408 and 589 infants indicated that intramuscular injection would be routinely achieved using a 16 mm long needle and the technique advocated by the World Health Organization.10
Clinical trial support for the use of the 23 gauge, 25 mm long needles has been drawn from a small study by Diggle and Deeks11 that had significant methodological weaknesses.
Vaccinations were given according to the Australian Childhood Immunisation Schedule5 with diphtheria–tetanus–acellular pertussis–hepatitis B vaccine (InfanrixHepB, [GlaxoSmithKline]) (children aged 2, 4 and 6 months) and diphtheria–tetanus–acellular pertussis vaccine (Infanrix, [GlaxoSmith-Kline]) (children aged 18 months). Haemophilus influenzae type b conjugate vaccine (Pedvax, [Merck Sharp & Dohme]) was given concurrently with the same technique as the acellular pertussis vaccine into the contralateral thigh of children aged 2 and 4 months. Oral polio (Sabin) vaccine (two drops) was given to children aged 2, 4 and 6 months.
The three intramuscular injection techniques used were:
Australian — the needle was inserted at the junction of the upper and middle thirds of the vastus lateralis with the needle angled at 45°–60° to the skin and pointing down towards the knee.5
World Health Organization — the needle was inserted into the anterolateral thigh at an angle of 90° to the long axis of the femur with the skin compressed between the index finger and the thumb.10
United States — the needle was inserted into the upper lateral quadrant of the thigh at an angle of 45° to the long axis of the femur and posteriorly at an angle of 45° to the table top, with the baby supine. The thigh muscle was bunched at the injection site to increase muscle mass and to minimise the chance of striking bone.4
The study outcome measures — local adverse reactions (bruising and redness/swelling), systemic adverse reactions (irritability, perceived fever, persistent crying/screaming, drowsiness, vomiting/poor feeding) and parental acceptance — were assessed 24 hours after injection by the practice nurse, as in other pertussis vaccine reactogenicity studies.12,13
A previously validated research instrument14 was used to objectively assess local reactions — bruising and redness/swelling — on a visual analogue scale (VAS), where 0 = no reaction and 5 = whole leg involved; and subjectively (parent) reported irritability, perceived fever, persistent crying/screaming, drowsiness, vomiting/poor feeding on a VAS, where 0 = no reaction and 5 = very severe reaction. Likewise, parental rating of vaccination outcome was scored 0 = very happy and 5 = very unhappy.
The sample size was based on the anticipated proportion of patients with redness/swelling after vaccination. It was expected that 37.6% of patients would experience redness/swelling with the WHO technique,14 compared with 20% with each of the US and Australian techniques. With an α level of 5%, adjusted for multiple comparisons and power of 80%, it was calculated that 360 participants would be required, 120 in each of the three groups. Thus, 125 participants per group were recruited as, from our previous study,14 a “drop out” rate of less than 4% was expected.
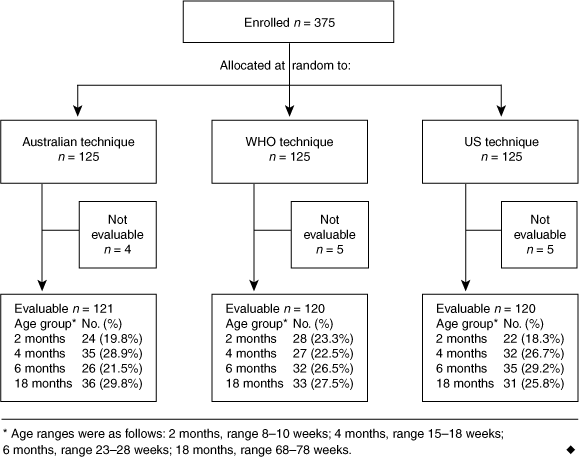
A total of 375 consecutive children were enrolled in the study, all satisfying the inclusion criteria at presentation. The reason for unavailability of all 14 children who could not be evaluated 24 hours after vaccination was parental non-compliance rather than adverse effects. This was ascertained in follow-up contact by the practice nurse. The study groups were similar in terms of numbers per age group for the three techniques (Box 1).
No statistically significant interaction was found between technique and age in any of the logistic regression models, so this interaction factor was removed from the analysis model. Where the age factor was not significant, it was also removed from the analysis model. Age was significant in the analysis of redness/swelling, bruising, irritability and persistent crying, and was kept in these models. In Box 2, the P values for these parameters represent the significance of the test after adjusting for age.
The WHO technique resulted in significantly fewer patients with the systemic adverse reaction variable “irritability” (30.0%) compared with the Australian technique (45.5%) and the US technique (49.2%) (P = 0.0039). There was a significant difference between the groups overall for bruising with acellular pertussis vaccine (P = 0.0418) after controlling for age. The difference was due to 6.7% bruising for the US technique compared with 0.8% for the WHO technique (P = 0.0356), but this was not statistically significant at the α = 0.025 level after adjusting for multiple comparisons (Box 2).
Ascertaining the best technique for paediatric vaccination is mandated by increasing concern about vaccine-induced adverse reactions15 in the context of a decreasing incidence of vaccine-preventable diseases. This concern is highlighted in a recent Australian study of children with incomplete vaccination, in which it was found that 70% of those who disagreed with or were concerned about immunisation had concerns about adverse reactions.16
The conclusion on the needle length aspect of vaccination practice by Diggle and Deeks11 was weakened by the use of needles with different gauges (23 gauge/25 mm v 25 gauge/16 mm). Similarly, the inability to control needle length and gauge as potential variables in our study may have weakened the conclusions drawn regarding differences between the different techniques of injection. Our choice of needles was dictated by the recommendation of 23 gauge/25 mm long needles with the US4 and Australian5 technique and previous ultrasound studies8,9 showing that 25 mm long needles would routinely make bony contact if used with the WHO technique. Elimination of needle gauge as a possible confounding variable was not possible, as 23 gauge/16 mm long needles are not commercially available.
1 Profile for clinical trial of three vaccination techniques (Australian, World Health Organization and United States)
Received 30 March 2005, accepted 9 June 2005
- Ian F Cook1
- John Murtagh2
- 1 Discipline of General Practice, School of Medical Practice and Population Health, Faculty of Health, University of Newcastle, Callaghan, NSW.
- 2 Department of General Practice, School of Primary Health Care, Monash University, East Bentleigh, VIC.
We wish to thank all the parents/guardians who so willingly allowed their children to participate in the study. We would also like to thank the Hunter Division of General Practice for providing funding for the statistical analysis conducted by Datapharm Australia.
None identified.
- 1. Sauer LW. Immunization in early childhood. Ill Med J 1950; 97: 73-76.
- 2. Gilles FH, Matson DD. Sciatic nerve injury following misplaced gluteal injection. J Pediatr 1970; 76: 247-254.
- 3. Marcuse EK, MacDonald NE. Neurological injury after vaccination in buttocks [letter]. CMAJ 1996; 155: 374.
- 4. American Academy of Pediatrics. Report of the Committee on Infectious Diseases. ELK Grove Village, Ill: American Academy of Pediatrics 1994; 11: 19-21.
- 5. National Health and Medical Research Council. The Australian Immunisation Handbook, 8th edition. Canberra: National Health and Medical Research Council, 2003: 9.
- 6. Talbert JL, Haslam RHA, Haller JA. Gangrene of the foot following intramuscular injection in to the lateral thigh: a case report with recommendation for prevention. J Pediatr 1967; 70: 110-114.
- 7. Hick JF, Charboneau JW, Brakke DM, et al. Optimum needle length for diphtheria-tetanus-pertussis inoculation of infants. Pediatrics 1989; 84: 136-137.
- 8. Groswasser J, Kahn A, Bouche B, et al. Needle length and injection technique for efficient intramuscular vaccine delivery in infants and children evaluated through an ultrasonographic determination of subcutaneous and muscle layer thickness. Pediatrics 1997; 100: 400-403.
- 9. Cook IF, Murtagh J. Needle length required for intramuscular vaccination of infants and toddlers. An ultrasonographic study. Aust Fam Physician 2002; 31: 295-297.
- 10. World Health Organization. When and how to give vaccines. In: Immunisaton in practice. A guide for health workers who give vaccines. Geneva: WHO, 1984; 3. (PI/PHWE/84/3rev.1.)
- 11. Diggle L, Deeks J. Effect of needle length on incidence of local reactions to routine immunization in infants aged 4 months: randomized controlled trial. BMJ 2000; 34: 931-933.
- 12. Harris G, Nolan T, Hartman L. Clinical signs associated with triple antigen (DTP) vaccination in infants. J Paediatr Child Health 1995; 31: 228-232.
- 13. Freeman T, Cuff W, Duchos P, et al. A comparison of local reactions to two formulations of DPT vaccine. Can Commun Dis Rep 1996; 20: 29-32.
- 14. Cook IF, Murtagh J. Comparative reactogenicity and parental acceptability of pertussis vaccines administered into the ventrogluteal area and anterolateral thigh in children aged 2, 4, 6 and 18 months. Vaccine 2003; 21: 3330-3334.
- 15. Chen RT, Rastogi SC, Mullen JR, et al. The Vaccine Adverse Event Reporting System (VAERS). Vaccine 1994; 12: 542-550.
- 16. Lawrence GL, Hull BP, Macintyre CR, et al. Reasons for incomplete immunization among Australian children. Aust Fam Physician 2004; 33: 568-571.





Abstract
Objective: To compare the rates of adverse reactions and parental approval ratings for three different techniques for anterolateral thigh vaccination in children aged 2, 4, 6 and 18 months.
Design: Randomised, observer-blind trial.
Participants: 375 children who received pertussis-containing vaccines in a regional New South Wales town between 29 May 2001 and 30 June 2002.
Interventions: Children were randomised to receive intramuscular injection with acellular pertussis-containing and Haemophilus influenzae type b vaccines with one of three recognised injection techniques (Australian, World Health Organization or United States).
Main outcome measures: Local adverse reactions (bruising and redness/swelling), systemic adverse reactions (irritability, perceived fever, persistent crying/screaming, drowsiness, vomiting/poor feeding) and parental acceptance were assessed 24 hours after injection.
Results: 361 children (96%) were evaluated 24 hours after vaccination. The WHO technique resulted in significantly fewer children, than with the other two techniques, with the systemic adverse reaction variable “irritability” (P = 0.0039). There was a significant difference between the technique groups overall for the local adverse reaction “bruising” with acellular pertussis-containing vaccines (P = 0.0418), due to a lower reaction rate in the WHO group compared with the US group (P = 0.0356).
Conclusion: The WHO technique appears to be the optimal technique for anterolateral thigh injection in children — it ensures that the injection is intramuscular, results in fewer adverse reactions, and is the easiest technique to perform as it does not require angling of the needle to the long axis of the femur.