Anaemia (generally defined as haemoglobin concentration [Hb] below 120 g/L) is a common problem in patients with cancer. Its causes are multifactorial,1,2 and its incidence depends on factors such as type of malignancy, disease duration and treatment regimen.1 Anaemia has numerous physical effects which can greatly affect quality of life in patients with cancer. However, it is difficult to accurately determine its incidence and frequency from the literature because of its widely varying definitions.3
The Australian Cancer Anaemia Survey (ACAS) was initiated to create a “snapshot” database documenting prevalence, incidence, frequency and management of anaemia in the adult cancer population attending oncology departments in Australia, and to identify patient and treatment characteristics associated with anaemia. ACAS was a prospective multicentre survey run in parallel with the European Cancer Anaemia Survey (ECAS), conducted in 24 European countries.4
The study was a prospective, multicentre survey intended to track anaemia and its management. Consenting participants were followed up for 6 months after enrolment, at approximately monthly intervals.
All data were recorded by a research nurse and were managed centrally by an independent company, The Epsilon Group (Charlottesville, Va, USA). This company also performed all statistical analyses of the data.
Data recorded at enrolment were: demographic details, disease status, previous treatment for cancer, treatment for anaemia in the 30 days before enrolment, weight, World Health Organization (WHO) performance status (a global assessment of ability for self care and ambulation),5 and results from a full blood examination. Data recorded at subsequent visits were: treatment status, treatment regimen, blood parameters, transfusions, erythropoietic agent and iron therapy administered, performance status and weight.
The timing of data collection depended on the cancer treatment being administered. Patients about to receive treatment provided enrolment data before the treatment began. Those receiving chemotherapy, either alone or simultaneously with radiotherapy, provided enrolment data at the end of a treatment cycle, and further data at the end of each subsequent treatment cycle, for a maximum of six cycles. Those receiving radiotherapy alone provided enrolment data 3–6 weeks after radiotherapy began, on its completion, and at each subsequent visit. Patients not currently receiving treatment provided data at each follow-up visit (maximum once per month).
For analysis of anaemia frequency by cancer treatment, patients were categorised as: chemotherapy only; radiotherapy only; concomitant chemoradiotherapy (chemotherapy and radiotherapy administered simultaneously); combination therapy (both chemotherapy and radiotherapy administered, but not simultaneously); or follow-up (patient had completed cancer treatment).
To ensure a valid sample across tumour types, sampling was stratified by tumour type. Statistical analyses were performed using the Statistical Package for the Social Sciences (SPSS), version 12.01.6 Descriptive statistics were used to explore patient characteristics and baseline haemoglobin concentrations. Differences between groups were examined using t tests (for proportions) or Mann–Whitney tests (for continuous variables). Logistic regression was used to model the incidence of anaemia.
The survey was approved by the ethics committee at each institution.
A total of 727 patients were enrolled. As no records were kept of patients who declined to participate, the participation rate is unknown. Of the 727 enrolled, 694 had evaluable data (33 had inconsistent coding of diagnoses). Their characteristics are shown in Box 1.
The prevalence of anaemia at the time of enrolment (defined as patients with a documented Hb level < 120g/L) was 35% overall (199/562). Prevalence varied by tumour type, disease status and treatment type (Box 2). It was highest for patients with gynaecological tumours (65%) and for those with persistent and recurrent disease (44%).
Most (78%; 156/199) patients with anaemia had an Hb level of 100–119 g/L. Prevalence of moderate–severe anaemia (Hb < 100g/L) was 7.7% (43/562) overall and was highest among patients with leukaemia (20%), gynaecological (18%) and urogenital (17%) cancers, and among those receiving concurrent chemotherapy and radiotherapy (15%).
The frequency of anaemia (defined as patients who had anaemia at enrolment or developed anaemia during the study period) is shown in Box 3. Frequency was 57% overall (323/566), and 60% (249/418) and 50% (74/148) in patients with solid and haematological malignancies, respectively (“other” tumour types were excluded). For individual tumour types, frequency varied from 49% for lymphoma/myeloma to 85% for urogenital cancer.
Frequency of moderate–severe anaemia (Hb < 100g/L) was 19% overall (110/588), and was highest in urogenital cancer (45%).
The frequency of anaemia was compared between cancer treatment groups for the 594 patients with at least one data point after enrolment (Box 3B). The group receiving chemotherapy plus radiotherapy, either concomitantly (8 patients) or in combination (78 patients), was more likely to have anaemia (73%; 63/86) than the group receiving chemotherapy alone (58%; 263/455; P = 0.004; 95% CI, 5%–25%). Patients who received chemotherapy alone were more likely to have anaemia than those in follow-up after treatment (58% [263/455] v 29% [14/48]; P < 0.001; 95% CI, 15%–43%).
The incidence of anaemia was defined as the percentage of patients who were not anaemic at enrolment (311 patients) but developed anaemia during the survey (Box 2). Overall incidence was 37% (114/311).
Of the 236 patients who received chemotherapy and did not have anaemia at enrolment, 191 were evaluable at the second data point (about 2 months from enrolment): 44 (23%) had developed anaemia, and three (2%) had moderate–severe anaemia. Analysis of these data by tumour type showed that incidence of anaemia in patients with breast cancer (23%; 13/57) was significantly lower than in those with lung cancer (67%; 6/9; P = 0.03; 95% CI, 5%–83%), but not different from incidence in patients with lymphoma/myeloma (27%; 10/37; P = 0.65) or colorectal malignancies (14%; 8/58; P = 0.22).
Incidence of anaemia throughout the survey was higher among patients who received chemotherapy (36%; 85/236) than among the follow-up group (not receiving treatment) (17%; 5/30; P = 0.01; 95% CI, 4%–34%).
A logistic regression model was used to identify the factors influencing the development of anaemia in chemotherapy patients. Variables analysed were low baseline Hb level (120–134 g/L for men, 120–129 g/L for women), age, sex, tumour type, disease status, WHO performance score and use of platinum-based chemotherapy. For anaemia, low baseline Hb level (adjusted odds ratio [OR], 5.4; 95% CI, 2.7–10.9; P < 0.001) and platinum-based chemotherapy (adjusted OR, 4.8; 95% CI, 2.1–11.4; P < 0.001) were found to be independent predictors. For Hb level < 100 g/L, only use of platinum-based chemotherapy was found to be an independent predictor (adjusted OR, 9.7; 95% CI, 2.7–35.7; P = 0.001).
Records as to whether anaemia was treated during the ACAS were available for 573 patients (421 with a solid tumour and 152 with a haematological malignancy). Among those with anaemia, 64% (208/323) were not specifically treated for anaemia during ACAS — 67% (167/249) of those with solid tumours and 55% (41/74) of those with haematological malignancies.
The frequency of anaemia treatment during the ACAS differed between tumour types (Box 4). The proportion of patients with breast cancer not treated for their anaemia was 81% (73/90), significantly greater than the proportion of patients with lung cancer (41%; 13/32; difference = 40%; 95% CI, 20%–60%; P < 0.001) and gynaecological cancer (52%; 15/29; difference = 29%; 95% CI = 8%–50%; P < 0.01).
Of the 176 patients with anaemia at enrolment, 41% received at least one therapeutic intervention for anaemia during the survey period: 36% received a transfusion; 5%, iron; and 2%, erythropoietic agents. Even among the 39 with moderate–severe anaemia at enrolment, 28% received no anaemia treatment during the survey.
The mean Hb “trigger” level for initiating transfusion as treatment for anaemia was 95 g/L (SD, 19) and differed between tumour types (Box 5). The mean trigger Hb was significantly higher for colorectal tumours than for lung tumours (P = 0.02) and lymphoma/myeloma (P = 0.003), but did not differ between patients receiving chemotherapy and those receiving either combination or concomitant chemotherapy and radiotherapy.
Our survey found that anaemia (Hb < 120g/L) was common among adults with cancer, with 57% of patients either having anaemia at enrolment or developing it during the following 6 months and 19% either having or developing moderate–severe anaemia (Hb < 100g/L). Patients who received radiotherapy as well as chemotherapy were more likely to have anaemia (73%) than those who received chemotherapy alone (58%) or those in the follow-up category not receiving cancer treatment (29%). Despite the high frequency of anaemia, 64% (208/323) of anaemic patients received no specific treatment for this during the survey.
The frequency of anaemia found in our survey is consistent with previous reports of cancer-related anaemia, irrespective of whether patients were receiving treatment.1,3 It is also comparable to the frequency of anaemia observed in ECAS.4
The major limitation of our survey is selection bias, as only selected patients from hospital oncology departments were included. It is difficult to assess how well the ACAS reflects the population with cancer in the community. Indeed, there were some differences in the profile of tumour types between the ACAS and incidence data for the total Australian population with cancer.7 However, this comparison is limited. It would have been optimum to compare prevalence rather than incidence data, but no accurate data exist on Australian cancer prevalence according to disease type. Secondly, this kind of comparison does not take into account differences in disease stage, as patients with more advanced cancers are more likely to be treated in a hospital oncology department. Interestingly, comparison of the ACAS and the European Cancer Anaemia Survey showed that patient profiles in the two settings were similar (Box 1).
In the ACAS, patients with gynaecological, urogenital and lung malignancies had the greatest risk of developing anaemia. These cancers are often treated with platinum-based chemotherapy,1 and indeed this therapy and “low-normal” baseline Hb level were independently predictive of developing anaemia. Of note in the ACAS, the rate of anaemia for myeloma/lymphoma was lower (49%) than reported in the ECAS (73%), even after accounting for proportional differences of lymphoma to myeloma within this subgroup.4 Other studies also report up to a 70% incidence of anaemia in patients with lymphoma/myeloma receiving treatment.8,9 It is difficult to explain our different finding, as age, sex and anaemia treatment before enrolment were comparable in the two surveys.
Cancer treatment may play a major role in the development of anaemia.1,10 This is illustrated in the ACAS, as 23% of the patients who were not anaemic at enrolment and received chemotherapy developed anaemia by the second data point (about 2 months after enrolment). In addition, the incidence of anaemia was higher in patients receiving both chemotherapy and radiotherapy compared with chemotherapy alone, but we note that the chemotherapy group was considerably larger than the combination group, which may have biased results.
Despite the high frequency of anaemia, most patients with anaemia were not specifically treated for it. The rates of treatment for anaemia were similar between the ACAS and ECAS surveys, for both solid malignancies (33% and 39%, respectively) and haematological malignancies (45% and 47%, respectively).4 When treatment was given during the ACAS, red cell transfusion was the most common therapy, given to 36% of patients with anaemia; 2% of treatments involved an erythropoietic agent. In comparison, in the ECAS, transfusion was given to 17% and erythropoietic agents to 15%.4 The mean Hb trigger level for initiating transfusion in the ACAS was similar to the trigger level in the ECAS (97 g/L).
Our survey was not able to assess whether the treatment of anaemia was appropriate. Although the current National Health and Medical Research Council guidelines for transfusion practice do not recommend transfusion routinely above a Hb level of 70 g/L,11 these guidelines are not specifically for patients with cancer.
Exogenous erythropoietic agents were used rarely. These agents are recommended to treat mild to moderate anaemia in cancer patients by both the American Society of Hematology/American Society of Clinical Oncology and the European Organization for Research and Treatment of Cancer.7,12 Further, the Cochrane Review on this subject concludes that such agents improve haematological response in patients with baseline Hb level < 100 g/L. However, debate continues on the impact of erythropoietic agents on quality of life.13 This debate, along with the fact that these agents are not currently reimbursed by the Pharmaceutical Benefits Scheme in Australia for treatment of cancer-related anaemia, probably contributed to their low usage during ACAS.
1 Population characteristics compared between the Australian Cancer Anaemia Survey, European Cancer Anaemia Survey and Australian cancer population*
|
Australian Cancer Anaemia Survey (n = 694)† |
European Cancer Anaemia Survey4 |
Australian incidence‡7 |
||||||||||||
Mean age in years (range) |
59.7 (19–90) |
59.0 (18–96) |
66 (M), 64 (F) |
||||||||||||
Female sex |
369 (61.2%) |
7014 (56.4%) |
40 578 (45.9%) |
||||||||||||
Disease status |
|
|
|
||||||||||||
New diagnosis, no cancer treatment |
127 (18.6%) |
4433 (32.8%) |
- |
||||||||||||
New diagnosis, cancer treatment |
238 (34.2%) |
2966 (22.0%) |
- |
||||||||||||
Persistent or recurrent |
258 (37.9%) |
4684 (34.7%) |
- |
||||||||||||
Remission |
63 (9.3%) |
1417 (10.5%) |
- |
||||||||||||
Treatment status |
|
|
- |
||||||||||||
No treatment |
185 (30.1%) |
7211 (53.7%) |
- |
||||||||||||
Chemotherapy alone |
396 (64.5%) |
5265 (39.2%) |
- |
||||||||||||
Chemotherapy + radiotherapy |
26 (4.2%) |
362 (2.7%) |
- |
||||||||||||
Radiotherapy alone |
7 (1.1%) |
578 (4.3%) |
- |
||||||||||||
Tumour type |
|
|
- |
||||||||||||
Breast |
186 (26.8%) |
2923 (21.8%) |
11 886 (13.4%) |
||||||||||||
Colorectal |
168 (24.4%) |
2245 (16.7%) |
12 844 (14.5%) |
||||||||||||
Lymphoma/myeloma |
152 (22.1%) |
2179 (16.2%) |
5416 (6.1%) |
||||||||||||
Lung |
47 (6.8%) |
1900 (14.1%) |
8275 (9.4%) |
||||||||||||
Gynaecological |
38 (5.5%) |
1564 (11.6%) |
2832 (3.2%) |
||||||||||||
Leukaemia |
38 (5.5%) |
601 (4.5%) |
2516 (2.8%) |
||||||||||||
Urogenital |
21 (3.1%) |
810 (6.0%) |
16 603 (18.8%) |
||||||||||||
Head and neck |
7 (1.0%) |
625 (4.7%) |
3398 (3.8%) |
||||||||||||
Other |
31 (4.5%) |
587 (4.4%) |
24 751 (28%) |
||||||||||||
WHO performance status§ |
|
|
|
||||||||||||
0 |
254 (37.8%) |
5248 (35.5%) |
- |
||||||||||||
1 |
320 (47.6%) |
6418 (43.4%) |
- |
||||||||||||
2 |
74 (11.0%) |
2439 (16.5%) |
- |
||||||||||||
3 |
23 (3.4%) |
582 (3.9%) |
- |
||||||||||||
4 |
1 (0.1%) |
86 (0.6%) |
- |
||||||||||||
* Numbers and percentages unless otherwise indicated. †Data were missing for: sex, 91; disease status, 13; treatment status, 80; tumour type, 6; and performance status, 22. ‡Australian incidence (not prevalence) data for 2001.7 §Global assessment of ability for self care and ambulation, where 0 = fully active; 1 = restricted in physically strenuous activity but able to carry out light work or activities; 2 = ambulatory and capable of self care but unable to work; 3 = capable of only limited self care, confined to bed or chair > 50% of time; 4 = completely disabled, totally confined to bed or chair.5 M = male. F = female. |
|||||||||||||||
2 Prevalence of anaemia at enrolment and incidence of anaemia developing during the study*
|
Prevalence at enrolment |
Incidence during study† |
|||||||||||||
Overall |
199/562 (35%) |
114/311 (37%)‡ |
|||||||||||||
By tumour type |
|
|
|||||||||||||
Breast |
50/149 (34%) |
29/86 (34%) |
|||||||||||||
Colorectal |
44/143 (31%) |
24/89 (27%) |
|||||||||||||
Lymphoma and myeloma |
37/122 (31%) |
22/71 (31%) |
|||||||||||||
Lung |
13/33 (39%) |
13/19 (68%) |
|||||||||||||
Gynaecological |
22/34 (65%) |
6/11 (55%) |
|||||||||||||
Leukaemia |
14/30 (47%) |
1/8 (13%) |
|||||||||||||
Urogenital |
9/18 (50%) |
7/9 (78%) |
|||||||||||||
Head and neck |
2/6 (33%) |
3/4 (75%) |
|||||||||||||
Other |
6/22 (27%) |
9/14 (64%) |
|||||||||||||
By disease status |
|
|
|||||||||||||
Newly diagnosed, untreated |
25/76 (33%) |
19/44 (43%) |
|||||||||||||
Newly diagnosed, treated |
65/213 (31%) |
46/132 (35%) |
|||||||||||||
Persistent/recurrent disease |
93/210 (44%) |
42/99 (42%) |
|||||||||||||
Remission |
13/54 (24%) |
6/34 (18%) |
|||||||||||||
By cancer treatment status |
|
|
|||||||||||||
No treatment§ |
15/61 (25%) |
5/30 (17%) |
|||||||||||||
Receiving treatment |
184/501 (37%) |
111/284 (39%) |
|||||||||||||
Chemotherapy |
147/413 (36%) |
86/236 (36%) |
|||||||||||||
Combination therapy¶ |
31/71 (44%) |
21/40 (53%) |
|||||||||||||
Concomitant therapy** |
4/13 (31%) |
3/6 (50%) |
|||||||||||||
Radiotherapy |
2/4 (50%) |
1/2 (50%) |
|||||||||||||
* Anaemia was defined as haemoglobin level < 120 g/L. †Percentage of non-anaemic patients at enrolment who developed anaemia during the survey. ‡Population of patients with at least one data point after enrolment. §About to begin treatment (prevalence) or in follow-up (incidence). ¶Combination therapy = patient received both chemotherapy and radiotherapy, but not simultaneously. ** Concomitant therapy = chemotherapy and radiotherapy delivered simultaneously. |
|||||||||||||||
3 Frequency of anaemia during ACAS
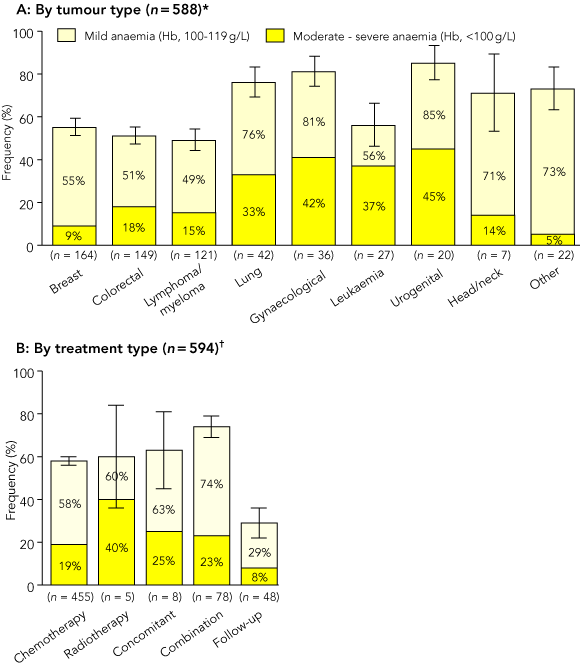
Error bars indicate the standard error of the mean for total frequency. * Data were missing for 14 patients. †Data were missing for 8 patients. Concomitant therapy = chemotherapy and radiotherapy delivered simultaneously. Combination therapy = both chemotherapy and radiotherapy administered, but not simultaneously.
4 Frequency of anaemia treatment during ACAS by tumour type for patients who had anaemia at enrolment or developed it during the survey (n = 339)*
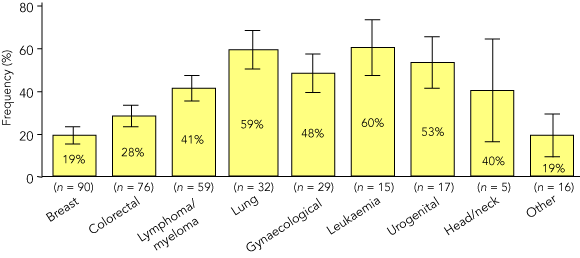
* Data were missing for 4 patients. Error bars indicate standard error of the mean for total frequency.
5 “Trigger” haemoglobin level for initiating treatment of anaemia
|
“Trigger” haemoglobin level (g/L) |
||||||||||||||
Tumour type |
Mean |
Median |
SD |
||||||||||||
Gynaecological (n = 15) |
96 |
95 |
22 |
||||||||||||
Urogenital (n = 9) |
98 |
88 |
27 |
||||||||||||
Leukaemia (n = 10) |
101 |
95 |
25 |
||||||||||||
Lung (n = 19) |
95 |
89 |
19 |
||||||||||||
Breast (n = 19) |
102 |
96 |
19 |
||||||||||||
Head/neck (n = 1) |
87 |
87 |
– |
||||||||||||
Colorectal (n = 28) |
113 |
105 |
28 |
||||||||||||
Lymphoma/ myeloma (n = 26) |
93 |
85 |
19 |
||||||||||||
Other (n = 4) |
128 |
126 |
19 |
||||||||||||
Received 20 September 2004, accepted 17 March 2005
- Tara Seshadri1
- H Miles Prince2
- David R Bell3
- Paul B Coughlin4
- Philip P B James5
- Gary E Richardson6
- Boris Chern7
- Peter Briggs8
- John Norman9
- Ian N Olver10
- Chris Karapetis11
- John Stewart12
- 1 Peter MacCallum Centre, Melbourne, VIC.
- 2 Royal North Shore Hospital, Sydney, NSW.
- 3 Box Hill Hospital, Box Hill, VIC.
- 4 Maroondah Hospital, Ringwood East, VIC.
- 5 Cabrini Hospital, Melbourne, VIC.
- 6 Redcliffe Hospital, Brisbane, QLD.
- 7 Monash Medical Centre, Melbourne, VIC.
- 8 Queen Elizabeth Hospital, Adelaide, SA.
- 9 Royal Adelaide Hospital, Adelaide, SA.
- 10 Flinders Medical Centre, Adelaide, SA.
- 11 Mater Hospital, Newcastle, NSW.
Statistical advice and data analysis were provided by the Epsilon Group. We would like to acknowledge the contribution of all those involved in the ACAS. Principal Investigators were: Geoffrey Beadle (Wesley Hospital, QLD); David Bell (Royal North Shore Hospital, NSW); Richard Bell (Geelong Hospital VIC); Peter Briggs (Monash Medical Centre, VIC); Robert Brodribb (Toowoomba Base Hospital, QLD); Michael Byrne (Sir Charles Gairdner Hospital, WA); Boris Chern (Redcliffe Hospital, QLD); Paul Coughlin (Box Hill Hospital, VIC); David Dalley (St Vincents Hospital, NSW); Narelle Driver (Westmead Private Hospital, NSW); Vinod Ganju (Frankston Hospital, VIC); Philip James (Maroondah Hospital, VIC); Chris Karapetis (Flinders Medical Centre, SA); Robert Lindeman (Prince of Wales Hospital, NSW); Alison Love (Nambour General Hospital, QLD); James Morton (St Andrews Hospital, Toowoomba, QLD); John Norman (Queen Elizabeth Hospital, SA); Ian Olver (Royal Adelaide Hospital, SA); Miles Prince (Peter MacCallum Centre, VIC); Gary Richardson (Cabrini Hospital, VIC); M Slancar (Gold Coast Hospital, QLD); John Sullivan (Freemasons Hospital, VIC); John Stewart (Mater Hospital, Newcastle, NSW); Kerry Taylor (Mater Public Hospital, QLD).
This study was supported by financial assistance from Janssen-Cilag (Australia). Gabrielle Allen, a medical writer from Janssen-Cilag, provided assistance with data analysis and editorial comments on drafts of the report.
- 1. Groopman JE, Itri LM. Chemotherapy-induced anemia in adults: incidence and treatment. J Natl Cancer Inst 1999; 91: 1616-1634.
- 2. Smith RE, Tchekmedyian S. Practitioners’ practical model for managing cancer-related anemia. Oncology (Huntingt) 2002; 16 Suppl 10: 55-63.
- 3. Knight K, Wade S, Balducci L. Prevalence and outcomes of anemia in cancer: a systematic review of the literature. Am J Med 2004; 116 Suppl 7A: 11S-26S.
- 4. Ludwig H, Barrett-Lee P, Birgegard G, et al. The European Cancer Anaemia Survey (ECAS): a large, multinational, prospective survey defining the prevalence, incidence, and treatment of anaemia in cancer patients. Eur J Cancer 2004; 40: 2293-2306.
- 5. World Health Organization. WHO handbook for reporting results of cancer treatment. Geneva: WHO, 1979. (WHO offset publication no. 48).
- 6. Statistical Package for the Social Sciences (SPSS), version 12.01 for Windows. Chicago, Ill: SPSS Inc.
- 7. Australian Institute of Health and Welfare, Australasian Association of Cancer Registries. Cancer in Australia 2001. Cancer series no. 28. Canberra: AIHW. (Catalogue No. CAN 23.) Available at: http://www.aihw.gov.au/publications/index.cfm/title/10083 (accessed Sep 2004).
- 8. San Miguel JF, Garcia-Sanz R, Gonzalez M, et al. A new staging system for multiple myeloma based on the number of S-phase plasma cells. Blood 1995; 85: 448-455.
- 9. Garton JP, Gertz MA, Witzig TE, et al. Epoetin alfa for the treatment of the anemia of multiple myeloma. A prospective, randomized, placebo-controlled, double-blind trial. Arch Intern Med 1995; 155: 2069-2074.
- 10. Dicato M. Anemia in cancer: some pathophysiological aspects. Oncologist 2003; 8 Suppl 1: 19-21.
- 11. National Health and Medical Research Council. Clinical practice guidelines. Appropriate use of red blood cells. Canberra: NHMRC, 2001. Available at: http://www.health.gov.au/nhmrc/publications/pdf/cp81.pdf (accessed Sep 2004).
- 12. Rizzo JD, Lichtin AE, Woolf SH, et al. Use of epoetin in patients with cancer: evidence-based clinical practice guidelines of the American Society of Clinical Oncology and the American Society of Hematology. Blood 2002; 100: 2303-2320.
- 13. Bohlius J, Langensiepen S, Schwarzer G, et al. Erythropoietin for patients with malignant disease. Cochrane Database Syst Rev 2004; 3: CD003407.





Abstract
Objective: To evaluate the frequency and management of anaemia in Australian adults with solid and haematological malignancies.
Design: 6-month observational, prospective, multicentre study.
Participants: 694 patients recruited from outpatient oncology clinics in 24 hospitals in five Australian states between 9 April 2001 and 31 July 2001.
Main outcome measures: Frequency of anaemia (haemoglobin [Hb] level < 120 g/L) at enrolment and over ensuing 6 months, by tumour type, disease status and cancer treatment; anaemia treatment and “trigger” Hb level for this treatment.
Results: Participants had median age 60 years, and 61% were women. Prevalence of anaemia at enrolment was 35% (199/562), with 78% of these 199 having mild anaemia (Hb, 100–119 g/L). Frequency of anaemia (either present at enrolment or developing during the study) was 57% overall (323/566), and varied with tumour type, from 49% (lymphoma/myeloma) to 85% (urogenital cancer). Patients who received radiotherapy either in combination or concomitant with chemotherapy were more likely to have anaemia (73%) than those receiving chemotherapy alone (58%) (P = 0.004). Of all chemotherapy patients not anaemic at enrolment, 23% developed anaemia by the second monthly follow-up. Independent predictors for anaemia in chemotherapy patients were low baseline Hb level (odds ratio [OR], 5.4; 95% CI, 2.7–10.9) and use of platinum chemotherapeutic agents (OR, 4.8; 95% CI, 2.1–11.4) (P < 0.001). Anaemia was treated in 41% of patients with anaemia at enrolment — by transfusion (36%), iron (5%) and erythropoietic agents (2%). Frequency of anaemia treatment varied between tumour types, from 19% (breast cancer) to 60% (leukaemia). The mean “trigger Hb” for initiating transfusion was 95 g/L.
Conclusions: Anaemia is prevalent among Australian patients with cancer managed in hospital oncology units. Its management varies between tumour types. Many patients do not receive treatment for their anaemia.