A recent review and meta-analysis by Buchwald et al1 summarises succinctly the impact of bariatric (obesity) surgery on morbid obesity (Box 1) and its related comorbidities. The percentage excess weight loss in patients who have had bariatric surgery is reported to be 50%–70%, and “cure” or significant amelioration of diabetes, hypertension, hyperlipidaemia and obstructive sleep apnoea is experienced by over 80% of patients, on average, for each condition. The meta-analysis confirms that bariatric surgery is a safe and effective intervention, with positive effects persisting for years or decades. This leads us to consider the current difficulties in providing obesity surgery for Australian patients.
Obesity is now epidemic in the Western world, due to a complex range of environmental and genetic factors.3 The AusDiab survey showed a combined prevalence of overweight and obesity of about 60% in Australian adults.4 It would be fair to assume, therefore, that overweight and obesity are now more prevalent risk factors for disease than smoking. Extrapolating from overseas data, the yearly number of deaths in Australia attributable to obesity is in the order of 12 000–17 000.5,6 Over the past 20 years, the prevalence of both obesity and diabetes in Australia has doubled,4 and the upward trend is projected to continue. Unfortunately, poor results of non-surgical intervention mean that there is rarely an exit for patients entering the obese cohort.
Treatment strategies for obesity have been exhaustively evaluated, both at a primary-care level and as intensive medical therapies instituted for high-risk patients. Primary-care interventions have either been ineffective or of insufficient duration to assess long-term results.7-9 The National Health and Medical Research Council (NHMRC), after an extensive evaluation of available therapies,10 concluded that obesity is associated with significant morbidity and mortality and that medical treatments for obesity generally result in weight loss of less than 10 kg of variable duration. While a sustained 3–5 kg weight loss may be acceptable for an “overweight” or “Class I” obese patient (Box 1), recommending non-surgical therapy to morbidly obese patients needing more significant weight loss is unsupported by evidence. Surgery is documented as the only consistently effective therapeutic intervention for the morbidly obese.1,10,11
The fact that surgery is not widely advocated by clinicians managing severely obese patients may, in part, be explained by the chequered history of some procedures that have been introduced with enthusiasm, rapidly disseminated, then later abandoned because of either dangerous side effects (eg, jejuno-ileal bypass) or ineffective weight loss (eg, gastroplasty). The number of patients happy to undergo often untested procedures with unknown long-term consequences is testament to the desperation faced by those afflicted with obesity.
Current bariatric surgical techniques have evolved to produce highly safe and effective treatments for obesity and are now considered mainstream. Perhaps no other type of surgical procedure has been as extensively scrutinised. Multiple studies have shown that bariatric surgery leads to long-term weight loss with low morbidity and mortality. Indeed, obesity surgery has become one of the most frequently performed major surgical procedures in the United States (with over 120 000 projected cases for 2004), and is the second most frequent upper gastrointestinal surgical procedure (after cholecystectomy) performed in Australia (unpublished Health Insurance Commission data).
Bariatric surgery has been evaluated by the NHMRC,10 the UK National Institute for Clinical Excellence (NICE)11,12 and the US National Institutes of Health (NIH).13 These three agencies have explicitly recommended that surgery be made available to selected morbidly obese patients (Box 2). On the basis of a cost-effectiveness analysis of gastric bypass surgery, the NICE has recommended that National Health Service trusts actively promote bariatric surgery. Similar analyses in the United States are prompting the US government to consider more widespread payment, through Medicare, to allow surgery in uninsured patients.
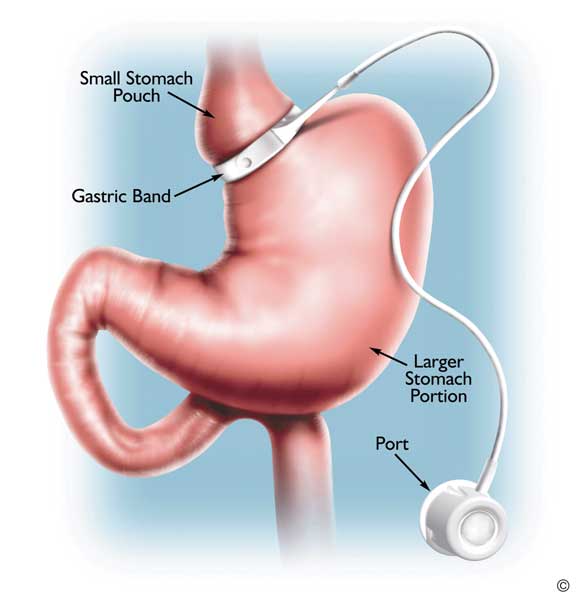
Worldwide, about 65% of bariatric surgical procedures performed each year are variations of the gastric bypass (Box 3), with the laparoscopic band (Box 4) being the second most common procedure. The latter procedure is the most common performed outside the United States.14
In Australia, we have an unusual situation in which Medicare pays for bariatric surgery and postsurgical care for privately insured patients, while non-insured patients are denied the same services in public hospitals. As the prevalence of obesity is significantly greater in lower socioeconomic classes, there are obviously a significant number of obese people excluded from treatment that has been recommended by the NHMRC.10 A surgical approach to obesity treatment is also supported by the Australian Safety and Efficacy Register of New Interventional Procedures — Surgical (ASERNIP-S)15 and the Medical Services Advisory Committee (MSAC).16 The recommendations of the ASERNIP-S and the MSAC were to continue provision of funding for gastric banding in particular, but bariatric surgery in general was also supported.
The reason bariatric surgery was allowed to proceed in the UK National Health Service was a cost analysis showing that gastric bypass was cheaper than other interventions on a quality-adjusted-life-year (QALY) basis.
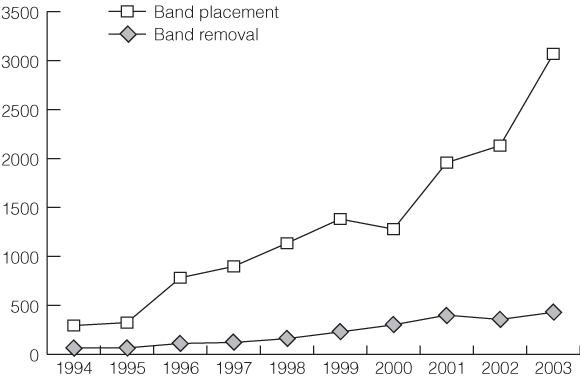
Gastric bypass surgery is the only intervention, to date, that has been shown to be cost-effective for treating severe obesity.17,18 It results in lower morbidity, mortality and cost in operated compared with non-operated patients.19,20 Although laparoscopic gastric banding did not appear as cost-effective as gastric bypass when evaluated by the NICE,11,12 modelling analysis suggests it could be cost-effective.21 This seems likely, given the good weight control and comorbidity resolution observed in Australian studies of gastric banding.22 The MSAC report16 costed gastric banding at just over $9000 and gastric bypass at just over $8000 per procedure — figures that accord fairly well with our own estimates (unpublished data), except that our bypass costs were $1000–$2000 higher than banding costs because they included hospital and intensive care unit/high-dependency unit stays. These figures do not take into account the apparent high rate of band removal/replacement,23-25 also reported in Health Insurance Commission data, that would need to be included in cost calculations (Box 5).
Patients with diabetes are of particular interest as a potential target population for bariatric surgery. All studies to date have shown a cure rate of at least 80% for type 2 diabetes after gastric bypass surgery.27-30 There are currently over 900 000 Australians with type 2 diabetes, with the number projected to rise to over 1.2 million by 2010.31 The yearly cost of managing each patient with diabetes averages $10 900 (ranging from $9095 to $15 850, depending on the presence of complications).32 This means that a patient with diabetes having bariatric surgery in a public hospital is likely to have the procedure pay for itself within a year. Existing federal–state funding arrangements are not conducive to promoting obesity surgery as a cost-saving measure. While most of the costs of managing people with diabetes and serious obesity-related comorbidities are borne by the federal government, state-government-funded hospitals bear the costs of surgery. It is unlikely that state governments, without benefiting from the overall savings, would be swayed by cost-effectiveness arguments. The current inequities may, therefore, continue.
Currently, Australia is far behind many Western countries in developing strategies to reduce the future burden of obesity and treat people who are severely afflicted. We have had no open-forum discussions between stakeholders and government of the kind that produced the recent UK House of Commons report on obesity.33 We have no primary-care equivalent of the UK Counterweight Project,8 and no effective treatments available to those who can not afford either drug therapy or surgery. Surgery for obesity is regarded by many people, including clinicians, to be akin to cosmetic surgery, a perception that is likely to persist while it remains solely in the domain of the private system. Although managing obesity is going to be a problem of major proportions, the longer we wait, the more difficult it will be to find solutions that suit the Australian population. The first step will be to acknowledge the severity of the problem and to offer treatment for the morbidly obese based on best available evidence. To do otherwise is to either ignore the evidence or simply discriminate against the obese.
1 Obesity definitions*2
|
BMI (kg/m2) |
Obesity class |
|||||||||||||
Underweight |
< 18.5 |
|
|||||||||||||
Normal |
18.5–24.9 |
|
|||||||||||||
Overweight |
25.0–29.9 |
|
|||||||||||||
Obesity |
30.0–34.9 |
I |
|||||||||||||
Severe |
35.0–39.9 |
II |
|||||||||||||
Very severe |
40.0+ |
III |
|||||||||||||
BMI = body mass index. * The term “morbid obesity” refers to BMI > 40 kg/m2, or BMI 35–39.9 kg/m2 with medical comorbidities. |
|||||||||||||||
2 Selection criteria for bariatric surgery13
Body weight
BMI > 40 kg/m2; or
BMI 35–39.9 kg/m2 with medical comorbidities
No endocrine cause of obesity
Resistant obesity
Obesity present > 5 years
Multiple failed non-surgical attempts to lose weight
Psychological profile
No alcohol or drug use
No (or controlled) psychiatric conditions
Understanding of the surgery involved and commitment to follow-up
3 Gastric bypass
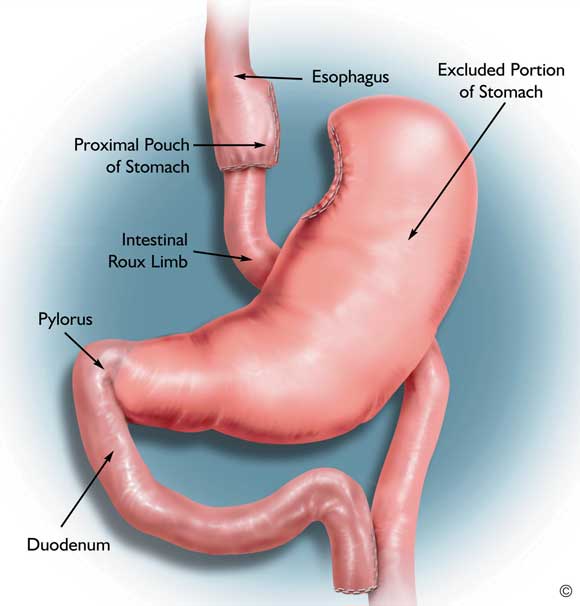
A small part of the upper stomach is separated from the main part and connected to the small bowel. This bypasses most of the stomach, duodenum and proximal jejunum. (Diagram courtesy of Johnson and Johnson Medical.)
- Michael L Talbot1
- John O Jorgensen2
- Ken W Loi3
- Department of Surgery, University of New South Wales at St George Hospital, Kogarah, NSW.
Drs Talbot and Jorgensen received travel assistance from Johnson and Johnson Medical to attend an obesity surgery meeting in 2004. All three authors are bariatric surgeons with public hospital appointments.
- 1. Buchwald H, Avidor Y, Braunwald E, et al. Bariatric surgery: a systematic review and meta-analysis. JAMA 2004; 292: 1724-1737.
- 2. World Health Organization. Obesity: preventing and managing the global epidemic. Report of a WHO consultation. World Health Organ Tech Rep Ser 2000; 894: i–xii, 1–253.
- 3. Swinburn B, Egger G. The runaway weight gain train: too many accelerators, not enough brakes. BMJ 2004; 329: 736-739.
- 4. Cameron AJ, Welborn TA, Zimmet PZ, et al. Overweight and obesity in Australia: the 1999–2000 Australian Diabetes, Obesity and Lifestyle Study (AusDiab). Med J Aust 2003; 178: 427–432. <MJA full text>
- 5. Allison DB, Fontaine R, Manson JE, et al. Annual deaths attributable to obesity in the United States. JAMA 1999; 282: 1530-1538.
- 6. Banegas JR, Lopez-Garca E, Gutierrez-Fisac JL, et al. A simple estimate of mortality attributable to excess weight in the European Union. Eur J Clin Nutr 2003; 57: 201–208.
- 7. Moore H, Summerbell CD, Greenwood DC, et al. Improving management of obesity in primary care: cluster randomised trial. BMJ 2003; 327: 1085-1089.
- 8. The Counterweight Project Team. A new evidence-based model for weight management in primary care: the Counterweight Programme. J Hum Nutr Diet 2004; 17: 191-208.
- 9. James J, Thomas P, Cavan D, Kerr D. Preventing childhood obesity by reducing consumption of carbonated drinks: cluster randomised controlled trial. BMJ 2004; 328: 1237.
- 10. National Health and Medical Research Council. Clinical practice guidelines for the management of overweight and obesity in adults. Canberra: NHMRC, September 2003. Available at: www.health.gov.au/internet/wcms/publishing.nsf/Content/obesityguidelines-guidelines-adults.htm/$FILE/adults.pdf (accessed Feb 2005).
- 11. Clegg A, Sidhu MK, Colquitt J, et al. Clinical and cost effectiveness of surgery for people with morbid obesity. Southampton: National Institute for Clinical Excellence, 2001. Available at: www.nice.org.uk/pdf/AssessmentReport-Surgeryforobesity.pdf (accessed Mar 2005).
- 12. NHS National Institute for Clinical Excellence. Guidance on the use of surgery to aid weight reduction for people with morbid obesity. Technology Appraisal Guidance No. 46. London: NICE, July 2002. Available at: www.nice.org.uk/pdf/Fullguidance-PDF-morbid.pdf (accessed Feb 2005).
- 13. US National Heart, Lung, and Blood Institute. Clinical guidelines on the identification, evaluation, and treatment of overweight and obesity in adults. Bethesda, MD: NHLBI, 1998. Available at: www.nhlbi.nih.gov/guidelines/obesity/ob_home.htm (accessed Feb 2005).
- 14. Buchwald H. 50 years of bariatric surgery. Obes Surg 2004; 14: 898.
- 15. Chapman A, Game P, O’Brien P, et al. Systematic review of laparoscopic adjustable gastric banding in the treatment of obesity. Update and re-appraisal. Adelaide: Australian Safety and Efficacy Register of New Interventional Procedures — Surgical, Royal Australasian College of Surgeons, June 2002. Available at: www.surgeons.org/asernip-s/systematic_review/LAGBreviewUp0602.pdf (accessed Feb 2005).
- 16. Medical Services Advisory Committee, Australian Department of Health and Ageing. Laparoscopic adjustable gastric banding for morbid obesity. MSAC reference 14. Assessment report. Canberra: DHA, 2003. Available at: www.msac.gov.au/pdfs/reports/msacref14.pdf (accessed Feb 2005).
- 17. Gallagher SF, Banasiak M, Gonzalvo JP, et al. The impact of bariatric surgery on the Veterans Administration healthcare system: a cost analysis. Obes Surg 2003; 13: 245-248.
- 18. Craig BM, Teng DS. Cost-effectiveness of gastric bypass for severe obesity. Am J Med 2002; 113: 491-498.
- 19. Christou NV, Sampalis JS, Liberman M, et al. Surgery decreases long-term mortality, morbidity, and health care use in morbidly obese patients. Ann Surg 2004; 240: 416–424.
- 20. Sampalis JS, Liberman M, Auger S, Christou NV. The impact of weight reduction surgery on health-care costs in morbidly obese patients. Obes Surg 2004; 14: 939-947.
- 21. Clegg A, Colquitt J, Sidhu M, et al. Clinical and cost effectiveness of surgery for morbid obesity: a systematic review and economic evaluation. Int J Obes 2003; 27: 1167-1177.
- 22. Dixon JB, O’Brien PE. Changes in comorbidities and improvements in quality of life after LAP-BAND placement. Am J Surg 2002; 184(6B): 51S-54S.
- 23. Biertho L, Steffen R, Branson R, et al. Management of failed adjustable gastric banding. Surgery 2005; 137: 33-41.
- 24. Tweddle EA, Woods S, Blamey S. Laparoscopic gastric banding: safe and modestly successful. ANZ J Surg 2004; 74: 191-194.
- 25. Camerini G, Adami G, Marinari GM, et al. Thirteen years of follow-up in patients with adjustable silicone gastric banding for obesity: weight loss and constant rate of late specific complications. Obes Surg 2004; 14: 1343-1348.
- 26. O’Brien P, Brown W, Dixon J. Revisional surgery for morbid obesity – conversion to the Lap-Band system. Obes Surg 2000; 10: 557-563.
- 27. Hickey MS, Pories WJ, MacDonald KG, et al. A new paradigm for type 2 diabetes mellitus: could it be a disease of the foregut? Ann Surg 1998; 227: 637-644.
- 28. Schauer PR, Burguera B, Ikramuddin S, et al. Effect of laparoscopic Roux-en-Y gastric bypass on type 2 diabetes mellitus. Ann Surg 2003; 238: 467-485.
- 29. MacDonald KG, Long SD, Swanson MS, et al. The gastric bypass operation reduces the progression and mortality of non-insulin dependent diabetes mellitus. J Gastrointest Surg 1997; 1: 213–220.
- 30. Scopinaro N, Adami GF, Marinari GM, et al. Biliary pancreatic diversion. World J Surg 1998; 22: 936-946.
- 31. McCarty DJ, Zimmet P, Dalton A, et al. The rise and rise of diabetes in Australia, 1996: a review of statistics, trends and costs. Canberra: Department of Health and Family Services, 1996.
- 32. Colagiuri S, Colagiuri R, Conway B, et al. DiabCo$t Australia: assessing the burden of type 2 diabetes in Australia. Canberra: Diabetes Australia, 2003.
- 33. House of Commons Health Committee. Obesity. Third report of session 2003–04. London: Stationery Office, 2004. Available at: www.publications.parliament.uk/pa/cm200304/cmselect/cmhealth/23/23.pdf (accessed Feb 2005).





Abstract
Morbid obesity (defined as having a body mass index [BMI] > 40 kg/m2, or BMI > 35 kg/m2 with obesity-related comorbidities) is a medical disorder associated with increased morbidity and mortality.
Management guidelines published by the National Health and Medical Research Council and by similar US and UK bodies have recommended surgery as the most effective treatment available for selected patients with morbid obesity.
A recent meta-analysis of obesity surgery has documented its safety and effectiveness in resolving some of the major medical comorbidities that occur in obese patients.
To date, no intervention other than surgery has proven either effective or cost-effective in treating severe obesity and its associated medical conditions.
Targeting patients with metabolic complications of obesity (eg, type 2 diabetes) could lead to substantial cost savings for the public health system.
Currently, Medicare pays for privately insured patients to undergo obesity surgery, while uninsured patients are denied access to surgery in public hospitals. This raises significant equity issues that should be addressed.