Clinical record
A 33-year-old woman presented to the emergency department with a 10-day history of vesicular rash, 2 days of cough and fever, and 12 hours of dyspnoea, malaise and facial swelling. Her three children were recovering uneventfully from chickenpox. She had no past history of varicella infection or vaccination. She was a non-smoker, had no pre-existing medical conditions and was not known to be pregnant at the time of presentation.
On presentation, the patient was in respiratory distress, with a respiratory rate of 36 per minute, pulse of 140 bpm, and oxygen saturation of 72% in room air. Subcutaneous emphysema was noted over the upper chest wall. Multiple healing and healed vesicles were present. The provisional clinical diagnosis was pneumonia, in the presence of resolving varicella. Results of blood tests on admission are shown in Box 1. Blood film demonstrated neutrophilia with toxic changes. Chest x-ray revealed pneumomediastinum and subcutaneous emphysema, while computed tomography showed pneumomediastinum and consolidation in the left upper lobe with cavitation (Box 2A). Electrocardiography revealed sinus tachycardia with paroxysmal atrial fibrillation.
The patient was admitted to the intensive care unit with acute respiratory failure. Antimicrobial therapy was begun with ceftriaxone, flucloxacillin and aciclovir based on the clinical diagnosis of pneumonia with cavitation in a patient with active varicella. Blood cultures revealed gram-positive cocci in chains after 24 hours of incubation; intravenous clindamycin was added to the antibiotic regimen. The next morning cultures of blood and bronchoalveolar fluid confirmed the presence of Streptococcus pyogenes (group A streptococcus).
Uncontrolled sepsis progressed within hours to multiorgan failure, precipitated by an overwhelming systemic inflammatory response syndrome. Clinical features were consistent with streptococcal toxic shock syndrome, with the primary streptococcal infection being pneumonia.
Intensive care management included haemofiltration for acute renal failure; inotropic support for septic shock; invasive lung ventilation for respiratory failure (acute respiratory distress syndrome); activated protein C for the systemic inflammatory response syndrome; and infusion of intravenous gamma globulin for toxic shock syndrome, and fresh frozen plasma and platelets for coagulation dysfunction and thrombocytopenia.
Despite maximal support, the patient died 44 hours after presentation. An autopsy confined to the chest and abdomen showed interstitial emphysema involving the mediastinum, pericardium and left upper lobe of the lung, and extensive necrotising left bronchopneumonia with abscess formation, but no signs of pneumothorax (Box 2B). Numerous gram-positive cocci consistent with streptococci were evident on microscopy of lung sections. Varicella zoster virus DNA was detected by polymerase chain reaction in one of three tissue samples tested, but no viral cytopathic changes were seen, and no virus was detected by culture.
Varicella (chickenpox) is a highly infectious disease caused by varicella zoster virus. Monthly notifications in Australia fluctuate from 40 to 180, with three to four deaths yearly.1
Varicella zoster pneumonia with interstitial pneumonitis and respiratory distress syndrome is a well recognised complication which occurs more frequently in adults than children (particularly smokers, pregnant women and those who are immunocompromised). Despite the presence of varicella zoster virus DNA, there was no histological evidence that varicella pneumonia predisposed to the development of secondary group A streptococcal pneumonia in this case.
In children, varicella is a well recognised predisposing condition for invasive group A streptococcal infection, preceding 15% of recorded cases of this infection in a recent Canadian study.2 The most common manifestation of invasive group A streptococcal infection is necrotising fasciitis, a serious but rare infection of the deeper layers of skin and fatty subcutaneous tissues. This is one of the fastest-spreading infections known, consuming tissue at a rate of up to 3 cm per hour.3 Pneumonia is a less common manifestation but has a higher fatality rate (38% versus 12% for necrotising fasciitis), with a reported median survival of 2 days.4
In adults, varicella is less common as a predisposing condition for invasive group A streptococcal infection, but cases have been reported of varicella gangrenosum with limb-necrotising fasciitis and toxic shock syndrome5 and of septic arthritis6 secondary to group A streptococcal infection after chickenpox. The speed of the patient’s demise and the presence of streptococcal bacteraemia and streptococci in postmortem lung tissue supports S. pyogenes as the pathogen causing death in this patient. Pneumomediastinum has many causes, particularly barotrauma and other forms of trauma, but is rare secondary to pneumonia and pulmonary abscess. We believe that this is the first case of group A streptococcal pneumonia with mediastinal gas leakage recorded in Australia. Pneumomediastinum occurring ultimately as a complication of varicella has similarly not been reported previously in this country.
Primary vaccination of children is now recommended,7 but is not federally funded. Vaccination of adolescents and adults should also be strongly considered, as these age groups are more at risk of severe or lethal complications, including overwhelming secondary infection.
Once invasive group A streptococcal infection is clinically suspected, it can be confirmed bacteriologically by culture of blood or affected tissue. Early medical treatment with intravenous penicillin and clindamycin is indicated. When infection is associated with streptococcal toxic shock syndrome, normal human immunoglobulin reduces organ failure and possibly mortality.8 Aggressive surgical debridement is critical for invasive fasciitis or myositis. However, in our patient, surgical debridement of affected pulmonary and mediastinal tissue was not an option. The prognosis was clearly extremely poor at presentation. The illness could have been prevented by varicella vaccination.
1 Results of blood tests in a patient with complications from varicella
Test |
Result |
Normal range |
|||||||||||||
On admission |
|
|
|||||||||||||
Serum sodium (mmol/L) |
121 |
135–145 |
|||||||||||||
Serum potassium (mmol/L) |
3.4 |
3.2–4.5 |
|||||||||||||
Serum chloride (mmol/L) |
79 |
100–110 |
|||||||||||||
Urea (mmol/L) |
47.7 |
3.0–8.0 |
|||||||||||||
Creatinine (mmol/L) |
0.48 |
0.05–0.10 |
|||||||||||||
Albumin (g/L) |
21 |
33–47 |
|||||||||||||
Globulins (g/L) |
50 |
25–45 |
|||||||||||||
Bilirubin (μmol/L) |
31 |
< 20 |
|||||||||||||
Alkaline phosphatase (U/L) |
144 |
30–120 |
|||||||||||||
γ-Glutamyl transferase (U/L) |
308 |
< 50 |
|||||||||||||
Aspartate aminotransferase (U/L) |
89 |
< 40 |
|||||||||||||
Lactate dehydrogenase (U/L) |
425 |
110–250 |
|||||||||||||
Prothrombin time (s) |
19 |
8–14 |
|||||||||||||
Fibrinogen (g/L) |
9.9 |
1.5–4.0 |
|||||||||||||
Haemoglobin (g/L) |
127 |
110–165 |
|||||||||||||
White blood cell count (cells/L) |
17.3 x 109 |
3.5–11.0 x 109 |
|||||||||||||
Neutrophils (cells/L) |
16.2 x 109 |
2.0–8.0 x 109 |
|||||||||||||
Platelets (cells/L) |
113 x 109 |
140–400 x 109 |
|||||||||||||
6 h after admission* |
|||||||||||||||
pH |
7.01 |
7.35–7.45 |
|||||||||||||
pco2 (mmHg) |
54 |
35–45 |
|||||||||||||
po2 (mmHg) |
87 |
75–100 |
|||||||||||||
* After intubation and ventilation. |
|||||||||||||||
2 Appearance of the lungs in a patient with group A streptococcal pneumonia
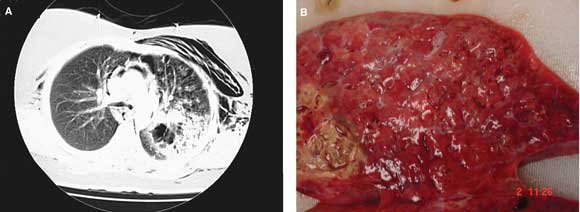
A: Computed tomography of the chest on admission showed subcutaneous emphysema, pneumomediastinum and consolidation in the left upper lobe with cavitation. B: Postmortem examination showed extensive necrotising left bronchopneumonia with abscess formation.
- 1. Chant KG, Sullivan EA, Burgess MA, et al. Varicella–zoster virus infection in Australia. Aust N Z J Public Health 1998; 22: 413-418.
- 2. Laupland KB, Davies HD, Low DE, et al. Invasive group A streptococcal disease in children and association with varicella-zoster virus infection. Ontario Group A Streptococcal Study Group. Pediatrics 2000; 105: E60.
- 3. Green RJ, Dafoe DC, Raffin TA. Necrotizing fasciitis. Chest 1996; 110: 219-229.
- 4. Muller MP, Low DE, Green KA, et al. Ontario Group A Streptococcal Study. Clinical and epidemiologic features of group A streptococcal pneumonia in Ontario, Canada. Arch Intern Med 2003; 163: 467-472.
- 5. Gonzalez-Ruiz A, Ridgway GL, Cohen SL, et al. Varicella gangrenosum with toxic shock-like syndrome due to group A streptococcal infection in an adult. Clin Infect Dis 1995; 20: 1058-1060.
- 6. Glass KL, Dillard TA, Dennis GJ, et al. Pyogenic arthritis complicating varicella infection. South Med J 1994; 87: 857-859.
- 7. National Health and Medical Research Council. Australian immunisation handbook. 8th ed. Canberra: NHMRC, 2003.
- 8. Darenberg J, Ihendyane N, Sjolin J, et al. Intravenous immunoglobulin G therapy in streptococcal toxic shock syndrome: a European randomized, double-blind, placebo-controlled trial. Clin Infect Dis 2003; 37: 333-340.





Acknowledgement: We thank Queensland Health Pathology and Scientific Services for microbiological identification and typing, and acknowledge the assistance of Dr Kathryn Urankar for performing the autopsy and photographing the lung.