Osteoarthritis (OA) is a chronic, degenerative disorder of multifactorial aetiology, characterised by loss of articular cartilage and periarticular bone remodelling. OA causes joint pain, typically worse with weight bearing and activity, and stiffness after inactivity. There is no cure, and gradual, although slow, progression is most common. Almost 1.2 million Australians have symptoms of OA, and 13% are classified as disabled or handicapped.1 As well as affecting over half of people aged over 75 years, OA is a significant problem for 10% of adults still in the workforce. Overall, OA is the leading cause of musculoskeletal pain, disability and handicap in Australia.1
Goals of managing OA include controlling pain, maintaining and improving the range of movement and stability of affected joints, and limiting functional impairment. 2,3 These goals should be achieved with minimal toxicity. Joint arthroplasty is indicated by end-stage joint failure with intractable pain, but most patients will be managed without surgery. Management must be individualised and patient-centred, and usually involves multiple strategies. Most morbidity is associated with OA of the large weight-bearing joints (the knee and hip). Here, we provide a pragmatic outline of the medical management of OA of these joints. A treatment algorithm is outlined in Box 1.
Education of patients with OA can reduce their pain and improve their quality of life.4 The aim is to provide patients with an understanding of the disease process, its prognosis and the rationale and implications of managing their condition. Patients can be educated during the consultation with a doctor, through consumer groups or by being provided with written material. Anecdotally, patients often seek information from the Internet. Some quality sources of information for patients are outlined in Box 2.
The Arthritis Self-Management Program is a community-oriented, peer-led program in which patients receive education and gain skills for self-managing the consequences of arthritis. The Arthritis Foundation of Australia coordinates the running of these courses, which are led by trained volunteers and held in community halls. Randomised trials have shown that participants have reduced joint pain, fewer arthritis-related visits to doctors, increased physical activity and improved quality of life.5 A systematic review of published trials of all types of education programs for self-management of arthritis confirms clinically small, but significant, reductions in pain and disability.6
Obesity is a risk factor for the development of OA, and is associated with radiological progression of the disease, and disability.7 When people walk, three to six times their body weight is transferred across the knee joint; any excess weight should be multiplied by this factor to estimate the excess force across the knee joint of overweight people. The Framingham study showed that modest weight loss reduced the risk of developing symptomatic knee OA in women.8 Small studies of overweight patients with knee OA have shown that modest weight loss (< 5 kg) has significant short-term9 and long-term10 reduction in symptoms of OA.
In managing OA, weight reduction should be a key goal. Exercise plays a role, but pain and disability can make it difficult for patients to exercise sufficiently to lose weight. Weight loss can be achieved with regular sessions with a dietitian who can provide instruction on reducing caloric intake and the use of food diaries, and cognitive-behavioural modification to change dietary habits.10
Inactivity due to pain leads to reduced muscle bulk around the osteoarthritic joint, and joint instability. The aim of exercise is to reduce pain and disability by strengthening muscle, improving joint stability, increasing the range of movement and improving aerobic fitness. Other, theoretical benefits include better self-esteem, weight reduction and improved general health.
Systematic reviews of short-term exercise programs show a small to moderate reduction of pain and disability, with similar benefits seen regardless of the type of exercise.11 Many such programs have involved intensive supervision and sophisticated equipment, with their sustainability over time unknown. A simple, largely unsupervised, home-based exercise program has been shown to reduce knee pain and disability, with its effects sustained over two years.12 Although hydrotherapy is widely used, no trials have shown any advantage over land-based exercise. Anecdotally, patients enjoy hydrotherapy and it may be a gentle start in encouraging exercise.
Osteoarthritis at different sites requires different approaches. Range-of-motion exercises may exacerbate pain in OA of the hip, and extension exercises can worsen the pain in patellofemoral OA. Thus, an individual exercise program should be formulated with the patient in consultation with a doctor, physiotherapist or qualified fitness instructor (Box 3).
Although there is no evidence available from well-designed trials to support the efficacy of walking sticks in OA, they are widely recommended. 2,3 Patients should be encouraged to wear shock-absorbing footwear with good mediolateral support, adequate arch support and calcaneal cushion. Short-term studies have shown biomechanical aids are effective in reducing OA-related pain. Lateral heel wedges may reduce pain related to OA of the medial tibiofemoral compartment,14 and applying adhesive tape to the patella can provide relief in patellofemoral OA.15 In patients with significant varus deformity, use of a tube-like knee support made of neoprene or an unloader brace both reduced pain over 6 months, with the brace being slightly more effective.16 Both may be uncomfortable to wear and the brace is expensive. Physiotherapy and occupational therapy assessment are recommended if there is functional limitation secondary to OA.
Pharmacological management should be considered an adjunct to non-pharmacological measures. Drug therapy should be individualised after a careful assessment of symptom severity, comorbid conditions, concomitant therapy, side effects, cost of therapy and patient preferences.
Paracetamol: It is widely accepted that paracetamol is the oral analgesic of first choice and, if successful, should be taken long term. 2,3 Although some patients prefer non-steroidal anti-inflammatory drugs (NSAIDs), paracetamol should be used as initial therapy based on relative cost and safety. It should be taken in divided doses, at regular intervals, with the total daily dose not exceeding 4 g. Although it is one of the safest analgesics, paracetamol can be associated with clinically important adverse events. Paracetamol may prolong the half-life of warfarin, so patients taking warfarin must have their INR (international normalised ratio) monitored closely and their warfarin dose adjusted if necessary.17 Paracetamol should be used with caution in patients who have liver disease and those with a history of excessive alcohol consumption.
NSAIDs and cyclo-oxygenase-2-specific inhibitors: NSAIDs should be considered only for patients who do not obtain adequate pain relief with paracetamol. Clinicians now have a choice between conventional NSAIDs and cyclo-oxygenase-2-specific (COX-2) inhibitors. COX-2 inhibitors have similar analgesic effects to those of non-selective NSAIDs, with a gastrointestinal (GI) side-effect profile and incidence of endoscopic ulceration similar to placebo. In two large trials, patients taking COX-2 inhibitors who did not take aspirin appeared to have a lower incidence of ulcer complications than those taking conventional NSAIDs. 18,19 The methods of these studies have been widely criticised, but meta-analysis supports these findings.20 Both NSAIDs and COX-2 inhibitors may cause acute deterioration in renal function, fluid retention and hypertension. The newer COX-2 inhibitors are considerably more expensive than NSAIDs, and uncertainty remains about potential increased risk of cardiovascular events.21 The VIGOR trial unexpectedly showed a fourfold increase in the rate of myocardial infarction in patients using rofecoxib compared with those using naproxen,19 and a retrospective cohort study found that doses of rofecoxib of more than 25 mg per day were associated with an excess risk of coronary events.22 This effect has not been seen with conventional NSAIDs or celecoxib. Rofecoxib is, in fact, the only commercially available, highly selective COX-2 inhibitor; celecoxib has a similar biochemical profile to diclofenac, and both meloxicam and celecoxib are more COX-2-selective than diclofenac.23 Inhibition of leukocyte COX-2 at a site of inflammation without inhibition of platelet COX-1 may lead to a prothrombotic state. Cardiovascular risk factors should be considered and patients should be counselled before prescribing rofecoxib (Box 4).
The choice between NSAIDs and COX-2 inhibitors should be made after carefully assessing the risk of serious upper-GI complications and discussing with patients the risk of serious thrombotic cardiovascular events (Boxes 4 and 5). Patients with no risk factors should use conventional NSAIDs, commencing at a low dose, with dose titration against effect. NSAIDs should be used on an as-required basis, although this often means continuous use. All NSAIDs have similar efficacy, so those with the lowest risk profile for upper-GI haemorrhage (ibuprofen and diclofenac) are recommended.2 COX-2 inhibitors are recommended for patients with any GI risk factors. 2,21 Rofecoxib should be avoided in patients with known risk factors for cardiovascular disease. All patients prescribed NSAIDs and COX-2 inhibitors should be counselled about the symptoms of upper-GI haemorrhage and monitored for new or severe upper-GI symptoms. NSAIDs should never be used in combination, except with low-dose aspirin for cardioprotection.
For patients with any risk factors for deterioration in renal function (Box 5), NSAIDs and COX-2 inhibitors should only be prescribed after very careful consideration of all other options. Plasma sodium, potassium and creatinine levels, blood pressure and the presence of oedema should be checked at baseline and regular intervals.
Glucosamine and chondroitin: Glucosamine sulfate (GS) and chondroitin sulfate (CS) are derivatives of glycosaminoglycans found in articular cartilage, and are available without prescription from pharmacies and supermarkets. A meta-analysis of 15 small, double-blind, randomised controlled trials concluded that these compounds had a small analgesic effect, but this may have been exaggerated by issues of quality and publication bias.26 Subsequently, two well-designed studies of oral GS confirmed a 20%–25% reduction in pain in patients with mild to moderate primary knee OA. 27,28 Patients with more severe disease and higher pain scores do not seem to benefit.29 GS is contraindicated in seafood allergy, but is otherwise well tolerated and causes no major side effects. GS should be used at a dose of 1500 mg per day as a divided dose for at least 3 months to determine whether it is therapeutic in any given patient. Topical application of GS and CS may be effective in reducing pain from knee OA.30 Some studies also suggest GS may slow radiological progression of OA. 27,28 However, the assessment of the radiological progression in published studies has been criticised, and further studies are required.
Opioids: The combination of codeine and paracetamol provides better analgesia than paracetamol alone. However, nausea, vomiting, dizziness and constipation lead to discontinuation of this combination in up to a third of patients.31 Some patients with intractable pain, who are unsuitable for arthroplasty, may require stronger opiate analgesia. Tramadol is a centrally acting synthetic opioid which inhibits the reuptake of serotonin and noradrenaline. It is generally well tolerated, but is contraindicated in seizure disorders, as it lowers the seizure threshold, and in combination with selective serotonin reuptake inhibitors because of the risk of serotoninergic syndrome.
Viscosupplementation: Hyaluronan is a component of synovial fluid, responsible for its viscoelasticity. In OA, catalytic enzymes reduce the concentration and molecular weight of the hylan polymers. Thus, viscosupplementation with synthetic long-chain hyaluronan preparations has been developed as intra-articular therapy for OA of the knee. The preparation available in Australia is Hylan G-F 20. Given as a weekly intra-articular injection for 3 weeks, Hylan G-F 20 decreases pain compared with placebo over 6 months, with efficacy comparable to continuous oral NSAID therapy.34 Adverse effects are uncommon, but include acute joint pain with effusion, which requires aspiration to exclude sepsis. This local reaction occurs after about 2%–3% of injections in a first course, but after up to 21% of injections in patients who have received more than one course of treatment.35 Hylan G-F 20 costs $440 per course, and this must be met entirely by the patient. This precludes widespread application of viscosupplementation in clinical practice.
Glucocorticoids: Clinical studies suggest intra-articular glucocorticoid injection affords some patients a modest and short-lived reduction in pain.32 In clinical practice, some patients have a dramatic and sustained response. Unfortunately, no clinical features reliably predict which patients might respond.32 Iatrogenic infection is rare if aseptic technique is used. Common side effects include flushing (40%), worsening hyperglycaemia and post-injection flare (thought to be inflammation in response to glucocorticoid crystals). The effect of repeated injections is unknown and it is recommended that a single joint not be injected more than three times a year. A recent study of the use of intra-articular steroids concluded that, although frequently repeated injections (four times per annum) were not particularly effective for reducing pain, they were safe.33 However, this study was not designed or powered to exclude harm related to repeated injections, and current recommendations, although not evidence-based, are unchanged.
Topical treatment is appropriate for patients as an adjunct to simple analgesia, monotherapy for a single symptomatic joint, or for patients who cannot tolerate systemic therapy. Topical capsaicin has a modest analgesic effect.36 A local burning sensation is common, but decreases with continued use. Patients must avoid inadvertently transferring the capsaicin to eyes or mucous membranes. A systematic review of topical NSAIDs concluded that 65% of patients allocated to active treatment had a good response, compared with 30% of patients using placebo.37 Although there is some systemic absorption of topical NSAIDs, there is no excess risk of upper-GI bleeding.
Patients with persistent pain and progressive limitation of daily activities despite medical management may be referred to an orthopaedic surgeon for assessment. To be suitable candidates for surgery, patients must be medically fit and able to participate in a rehabilitation program postoperatively. Total joint arthroplasty relieves pain and improves function over at least 10 years.38 Total joint arthroplasties do deteriorate over time, and may require revision. Revision arthroplasty is a more complicated procedure, so arthroplasty may be best postponed in younger patients with OA. The exact role of knee arthroscopy remains controversial and further well designed studies are required.39
For optimal results, management of OA requires multiple therapies and an individualised approach. Patients need to be involved in formulating and executing the management plan. As the disease progresses, or as comorbid conditions develop, management may need to be revised. The doctor’s role is to coordinate non-pharmacological approaches, supervise pharmacological management with the aim of minimising toxicity, and empower patients to manage their chronic condition.
1: Management algorithm for osteoarthritis of the knee and hip
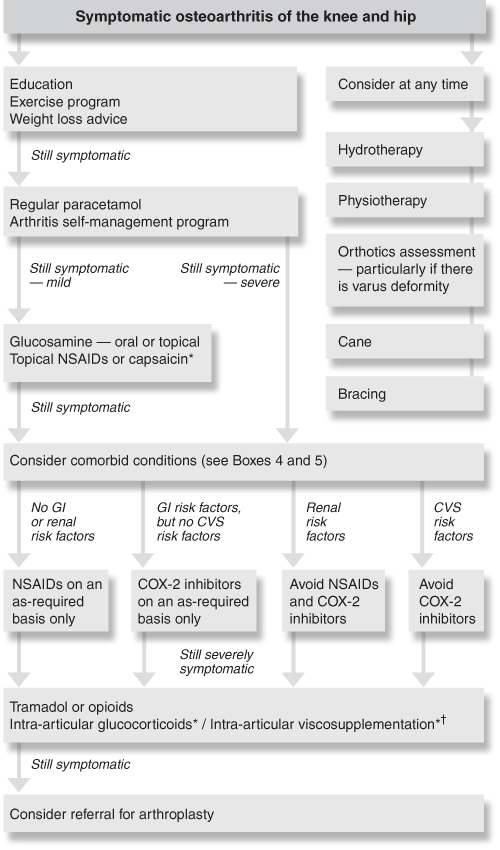
* Knee only. † With hyaluronan (Hylan G-F 20 [Synvisc]). NSAIDs = non-steroidal anti-inflammatory drugs. COX-2 = cyclo-oxygenase-2. GI = gastrointestinal. CVS = cardiovascular system.
2: Sources of patient education material
Books
Lorig K, Fries J. The arthritis helpbook — a tested self-management program for coping with arthritis and fibromyalgia. 5th ed. Cambridge: Perseus Books, 2000.
Websites
Australian Arthritis Foundation <www.arthritisfoundation.com.au>
Australian Rheumatology Association <www.rheumatology.org.au>
Arthritis USA <www.arthritis.org>
American College of Rheumatology <www.rheumatology.org>
Arthritis UK <www.arthritiscare.org.uk>
3: Creating an exercise program for lower-limb osteoarthritis*
The program should be individualised after considering:
Severity of pain
Joint stability
Patient’s resources (time, money, facilities, equipment)
Patient’s interests
The program should include:
Warm-up — 5 minutes. Range-of-movement exercises.
Flexibility exercises — daily stretching and range-of-movement exercises.
Strengthening exercises — (a) Isometric exercises (static muscle contraction that does not move a joint or alter muscle length) up to twice daily during acute inflammatory periods; and (b) Isotonic exercises (resistance training exercises, often with weights), maximum two days per week.
Endurance/fitness exercises — such as walking, swimming, dancing, aquarobics, cycling, 3–4 times per week.
The intensity, duration, and frequency of exercise should be specified and graded to allow for progression.
Tactics to improve compliance
Set specific, realistic, positive goals which should be written down
Start slowly and build up gradually
Keep a diary
Plan to exercise at a time when least tired and sore
Exercise with a friend
* Adapted from the American Geriatrics Society Panel on Exercise and Osteoarthritis, 2001.13
4: Rofecoxib and cardiovascular risk
An apparent increased risk of myocardial infarct in a long-term safety study of rofecoxib in patients with rheumatoid arthritis19 was also seen at higher dose (50 mg daily) in a general prescribing study.22
This risk is hypothesised to be especially high in patients with conventional cardiovascular risk factors (smoking, hypertension, diabetes mellitus, hyperlipidaemia and family history of premature cardiovascular disease in a first-degree relative).
Patients prescribed rofecoxib should be assessed for cardiovascular risk factors and be informed about the possibility of increased risk of cardiovascular events.
Rofecoxib should never be prescribed at doses greater than 25 mg daily.
5: Risk factors for complications when taking non-steroidal anti-inflammatory drugs
Risk of upper gastrointestinal tract complications24
Age > 65 years
Comorbid medical conditions
Use of oral glucocorticoids
History of peptic ulcer disease
History of upper-gastrointestinal haemorrhage
Risk of renal complications25
Age > 65 years
Raised serum creatinine level
Hypertension
Congestive heart failure
Use of angiotensin-converting enzyme inhibitors
Use of diuretics
- 1. Access Economics. The prevalence, cost and disease burden of arthritis in Australia — 2001. Melbourne: Arthritis Foundation of Australia, 2001.
- 2. Altman RD, Hochberg MC, Moskowitz RW, Schnitzer TJ. Recommendations for the medical management of osteoarthritis of the hip and knee. Arthritis Rheum 2000; 43: 1905-1915.
- 3. Pendelton A, Arden N, Dougados M, et al. EULAR recommendations for the management of knee osteoarthritis: report of a task force of the Standing Committee for International Clinical Studies Including Therapeutic Trials (ESCISIT). Ann Rheum Dis 2000; 59: 936-944.
- 4. Lorig KR, Manzonson PD, Holman HR. Evidence suggesting health education for self-management in patients with chronic arthritis has sustained health benefits while reducing health care costs. Arthritis Rheum 1993; 36: 439-446.
- 5. Superio-Cabuslay E, Ward MM, Lorig KR. Patient education interventions in osteoarthritis and rheumatoid arthritis: a meta-analytic comparison with nonsteroidal anti-inflammatory drug treatment. Arthritis Care Res 1996; 9: 292-301.
- 6. Warsi A, LaValley MP, Wang PS, et al. Arthritis self-management education programs. A meta-analysis of the effect on pain and disability. Arthritis Rheum 2003; 48: 2207-2213.
- 7. Felson DT, Lawrence RC, Dieppe PA, et al. Osteoarthritis: new insights. Part 1: the disease and its risk factors. Ann Intern Med 2000; 133: 635-646.
- 8. Felson DT, Zhang Y, Anthony JM, et al. Weight loss reduces the risk for symptomatic knee osteoarthritis in women. The Framingham Study. Ann Intern Med 1992; 116: 535-539.
- 9. Toda Y, Toda T, Takemura S, et al. Change in body fat, but not body weight or metabolic correlates of obesity, is related to symptomatic relief of obese patients with knee osteoarthritis after a weight control program. J Rheumatol 1998; 25: 2181-2186.
- 10. Messier SP, Loeser RF, Mitchell MN, et al. Exercise and weight loss in obese older adults with knee osteoarthritis: a preliminary study. J Am Geriatric Soc 2000; 48: 1062-1072.
- 11. Van Baar M, Assendeft WJJ, Dekker J, et al. Effectiveness of exercise therapy in patients with osteoarthritis of the hip or knee. A systematic review of randomised clinical trials. Arthritis Rheum 1999; 42: 1361-1369.
- 12. Thomas KS, Muir KR, Doherty M, et al. Home based exercise programme for knee pain and knee osteoarthritis: randomised controlled trial. BMJ 2002; 325: 752-756.
- 13. American Geriatrics Society Panel on Exercise and Osteoarthritis. Exercise prescription for older adults with osteoarthritis pain: consensus practice recommendations. A supplement to the AGS Clinical Practice Guidelines on the management of chronic pain in older adults. J Am Geriatr Soc 2001; 49: 808-823.
- 14. Keating EM, Faris PM, Ritter MA, Kane J. Use of lateral heel and sole wedges in treatment of medical osteoarthritis for the knee. Orthop Rev 1993; 22: 921-924.
- 15. Hinman RS, Bennell KL, Crossley KM, McConnell J. Immediate effects of adhesive tape on pain and disability in individuals with knee osteoarthritis. Rheumatology 2003; 42: 865-869.
- 16. Kirkley A, Webster-Bogaert S, Litchfield R, et al. The effect of bracing on varus gonarthrosis. J Bone Joint Surgery Am 1999; 81: 539-548.
- 17. Hyiek EM, Heiman H, Skates SJ, et al. Acetominophen and other risk factors for excessive warfarin anticoagulation. JAMA 1998; 279: 657-662.
- 18. Silverstein FE, Faich G, Goldstein JL, et al. Gastrointestinal toxicity with celecoxib vs nonsteroidal anti-inflammatory drugs for osteoarthritis and rheumatoid arthritis. The CLASS study: a randomised controlled trial. JAMA 2000; 284: 1247-1255.
- 19. Bombardier C, Laine L, Reicin A, et al. Comparison of upper gastrointestinal toxicity of rofecoxib and naproxen in patients with rheumatoid arthritis. VIGOR Study Group. N Engl J Med 2000; 343: 1520-1528.
- 20. Juni P, Rutjes AWS, Dieppe PA. Are selective COX-2 inhibitors superior to traditional non-steroidal anti-inflammatory drugs? BMJ 2002; 324: 1287-1288.
- 21. The Australian COX-2-Specific Inhibitor (CSI) Prescribing Group. Considerations for the safe prescribing and use of the COX-2-specific inhibitors. Med J Aust 2002; 176: 328-331. <MJA full text>
- 22. Wayne AR, Stein CM, Daughtery JR, et al. COX-2 selective non-steroidal anti-inflammatory drugs and risk of serious coronary heart disease. Lancet 2002; 360: 1071-1073.
- 23. Patrignani P, Panara MR, Greco A, et al. Biochemical and pharmacological characterization of the cyclooxygenase activity of human blood prostaglandin endoperoxide synthases. J Pharmacol Exp Ther 1994; 271: 1705-1712.
- 24. Gabriel SE, Jaakkimainen L, Bomberdier C. Risk for serious gastrointestinal complications related to the use of nonsteroidal anti-inflammatory drugs: a meta-analysis. Ann Intern Med 1991; 115: 787-796.
- 25. Garella S, Matarese RA. Renal effects of prostaglandins and clinical adverse effects of nonsteroidal anti-inflammatory agents. Medicine (Baltimore) 1984; 63: 165-181.
- 26. McAlindon TE, LaValley MP, Gulin JP, Felson DY. Glucosamine and chondroitin for treatment of osteoarthritis. A systematic quality assessment and meta-analysis. JAMA 2000; 283: 1469-1475.
- 27. Reginster JY, Deroisy R, Rovati LC, et al. Long-term effects of glucosamine sulfate on osteoarthritis progression: a randomised, placebo-controlled clinical trial. Lancet 2001; 357: 251-256.
- 28. Pavelka K, Gatterova J, Olejarova M, et al. Glucosamine sulfate use delays progression of knee osteoarthritis. A 3-year, randomized, placebo-controlled, double-blind study. Arch Intern Med 2002; 162: 2113-2123.
- 29. Hughes R, Carr A. A randomised, double-blind, placebo-controlled trial of glucosamine sulphate as an analgesic in osteoarthritis of the knee. Rheumatology 2002; 41: 279-284.
- 30. Cohen M, Wolfe R, Mai T, Lewis D. A randomized, double-blind, placebo-controlled trial of a topical cream containing glucosamine sulfate, chondroitin sulfate, and camphor for osteoarthritis of the knee. J Rheumatol 2003; 30: 523-528.
- 31. Kjaersgaard-Anderson P, Nafei A, Skov O, et al. Codeine plus paracetamol versus paracetamol in longer-term treatment of chronic pain due to osteoarthritis of the hip: a randomized, double-blind, multicentre study. Pain 1990; 43: 309-318.
- 32. Creamer P. Intra-articular corticosteroid treatment in osteoarthritis. Curr Opin Rheum 1999; 11: 417-421.
- 33. Raynauld JP, Buckland-Wright C, et al. Safety and efficacy of long-term intraarticular steroid injections in osteoarthritis of the knee: a randomized, double-blind, placebo-controlled trial. Arthritis Rheum 2003; 48: 370-377.
- 34. Adams ME, Atkinson MH, Lussier AJ, et al. The role of viscosupplementation with hylan G-F 20 (Synvisc TM) in the treatment of osteoarthritis of the knee. A Canadian multicenter trial comparing hylan G-F alone, hylan G-F 20 with non-steroidal anti-inflammatory drugs (NSAIDs) and NSAIDs alone. Osteoarthrits Cartilage 1995; 3: 213-225.
- 35. Leopold SS, Warme WJ, Pettis PD, Shott S. Increased frequency of acute local reaction to intra-articular Hylan GF-20 (Synvisc) in patients receiving more than one course of treatment. J Bone Joint Surgery (Am) 2002; 84A: 1619-1623.
- 36. Deal CL, Schnitzer TJ, Lipstein E, et al. Treatment of arthritis with topical capsaicin: a double blind trial. Clin Therapeutics 1991; 13: 383-395.
- 37. Moore RA, Tramèr MR, Carroll D, et al. Quantitative systematic review of topically applied non-steroidal anti-inflammatory drugs. BMJ 1998; 316: 333-338.
- 38. Harris W, Sledge C. Total hip and knee replacement. N Engl J Med 1990; 323: 725-731.
- 39. Chapman AB, Feller JA. Therapeutic arthroscopy for knee osteoarthritis: time to reconsider? Med J Aust 2003; 179: 179-180. <MJA full text>





Abstract
Osteoarthritis is a common, chronic condition which requires an individualised management plan involving multiple kinds of treatment.
Exercise programs and the Arthritis Self-Management Program reduce pain and disability associated with osteoarthritis.
Paracetamol is the most appropriate first-line analgesic.
Non-steroidal anti-inflammatory drugs may be used as second-line analgesia on an as-needed basis (including continuous use), but must be used with caution. Cyclo-oxygenase-2-specific inhibitors are used if there are risk factors for upper-gastrointestinal complications, but only after considering cardiovascular risk.
Glucosamine sulfate is a safe and effective over-the-counter treatment.
Intra-articular therapies are used when others have failed.