Thyroid nodules are common and of concern because of the risk of malignancy and hyperfunction. The incidence of papillary thyroid cancer appears to be increasing, both in Australia and worldwide.1 While some of this increase is due to detection of small lesions by sensitive diagnostic tests such as ultrasonography, there also seems to be an increase in larger lesions. The cause of this is unknown.
The reported prevalence of thyroid nodules varies with the method of screening. In autopsy series, up to 50% of clinically normal thyroid glands contain nodules. A lower prevalence is found by ultrasonography. For example, nodules were found in 27% of randomly selected people aged 19–50 years in an iodine-sufficient area of Finland; only 5% of these people had a clinically detectable abnormality.2 Fifty per cent of people with clinically detected solitary nodules have additional nodules when studied further by ultrasonography.3
Nodules are more common in women and in areas of iodine deficiency. Exposure to ionising radiation in childhood and adolescence increases the risk of both nodules and thyroid carcinoma.
Reports of the risk of malignancy in thyroid nodules vary. An Italian study of more than 5000 consecutive patients with “cold” thyroid nodules reported an incidence of malignancy of 5.3% in those from iodine-sufficient areas. Risk factors for malignancy included male sex and age less than 30 years or greater than 60 years.4
Thyroid nodules are usually found by the patient or a family member, or during a general physical examination, but increasingly they are incidentally discovered during neck imaging undertaken for other reasons, such as carotid duplex ultrasonography. It is important to determine whether the nodule is hyperfunctioning and whether it is malignant.
History is rarely helpful in differentiating benign and malignant nodules. An exception is a history of head and neck irradiation in childhood or teenage years. This is by far the greatest risk factor for thyroid malignancy, with a risk of carcinoma in nodules in this group of 35%–40%, in contrast to 5% in the general population.5 However, such a history is unusual in Australia, in contrast to countries such as the United States, where head and neck irradiation was used to treat enlarged tonsils, adenoids and acne in many children and adolescents. Another exception is a family history of medullary carcinoma of the thyroid, which should prompt consideration of familial medullary thyroid cancer or multiple endocrine neoplasia type 2, the latter being associated with hyperparathyroidism and phaeochromocytoma.
Physical examination is also rarely helpful in differentiating benign and malignant nodules, unless there is evidence of invasion of other structures in the neck (eg, hoarse voice) or enlarged regional lymph nodes, which suggest malignancy.
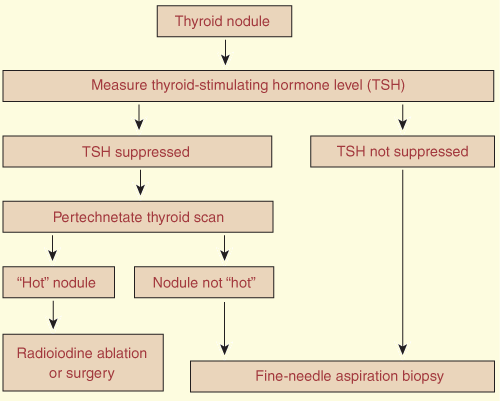
An algorithm for investigating thyroid nodules is shown in Box 1. This algorithm may be cut short in patients at high risk of cancer.
Thyroid imaging should not be routine, but should be used to answer a specific clinical question.
Thyroid scanning using pertechnetate (99MTc) (and formerly radioiodine, 131I) was traditionally used to screen thyroid nodules for malignancy. The finding of a hyperfunctioning or “hot” nodule (uptake of tracer within the nodule with suppression of uptake in the surrounding normal thyroid tissue) excludes malignancy in almost all patients. Current recommendations are that patients with TSH suppression should proceed to pertechnetate scanning to confirm that the nodule is hot. The patient can then be treated with therapeutic radioiodine or lobectomy. A non-functioning or “cold” nodule was thought to indicate increased risk of malignancy. Unfortunately, most nodules are cold, but perhaps only 5%–15% of these are malignant. Pertechnetate scans are thus not advocated for assessing a nodule in patients who are euthyroid or hypothyroid.
Thyroid ultrasonography is commonly performed. Although some ultrasonographic features, such as punctate calcification and irregular or blurred margins, suggest papillary carcinoma, routine ultrasonographic studies rarely aid clinical decision-making.6 Despite early suggestions that nodules in multinodular goitres are less likely to be malignant, more recent studies show that the risk of malignancy in a dominant nodule in this condition is similar to that in a solitary nodule.7
Computed tomography is rarely necessary, but can be useful in delineating the extent of disease in patients with known thyroid cancer. It should be remembered that use of iodinated contrast medium reduces technetium uptake and may reduce the diagnostic utility of pertechnetate scanning.
In the absence of TSH suppression, biopsy by fine-needle aspiration should be the first-line investigation for a solitary nodule or a dominant nodule in a multinodular goitre. An exception may be an “incidentaloma” (incidentally discovered small thyroid nodule) (Box 2).
The success of a biopsy depends on the adequacy of the specimen and skill of the cytopathologist. In experienced hands, the false negative rate is less than 5%, and the false positive rate less than 1%. The procedure can be done in ambulatory patients without local anaesthesia. Discomfort is usually mild. Specimens can be taken from several parts of the nodule, and slides smeared immediately. Immediate staining of some slides allows evaluation of the adequacy of material while the patient is still present and further passes if material is inadequate. There is a small risk of bleeding, which settles quickly with ice packs. Ultrasound guidance is preferred by some practitioners and is needed for impalpable lesions that warrant fine-needle aspiration biopsy.
Larger thyroid nodules are often partially cystic, containing fresh or altered blood. Aspirates often contain large numbers of macrophages, but follicular cells may be sparse. A specimen is regarded as adequate for diagnosis if it contains six to 10 clusters of thyroid cells. The more cystic the lesion, the more likely the specimen is to be non-diagnostic. A further biopsy attempt under ultrasound guidance is warranted, yielding a diagnostic sample in 63% of nodules.12 The remaining patients should not be considered to have benign disease, but even repeated aspiration may not obtain a sufficient sample in 5%–10%.13
The cytopathological report should be one of the following: insufficient for diagnosis; malignant; atypical; a follicular neoplasm; or benign. The presence of colloid and bland follicular cells suggests a benign lesion. The report should indicate adequacy of the specimen, as malignancy cannot be excluded if material is insufficient.
Papillary and often medullary carcinoma can be recognised on fine-needle aspiration biopsy (Box 3), but follicular adenomas and carcinomas have similar cytological appearance, with diagnosis of malignancy requiring demonstration of capsular or vascular invasion in paraffin sections. Follicular tumours are thus reported initially as “follicular neoplasms” and cannot be characterised as benign or malignant without histological examination. All follicular neoplasms should be excised unless there is clear evidence that the nodule is hyperfunctioning (see case report,Box 4).
Atypical cytology is found in 20% of specimens, and about a third of these lesions are found to be malignant when excised and examined histologically.
Indications for thyroid surgery thus include malignant or atypical cytology, follicular neoplasm, and failure to obtain a diagnostic sample. In the future, molecular genetic studies of aspirated material (eg, for the RET/PTC gene rearrangements often found in papillary cancer) may offer more precise diagnosis.
Thyroxine suppressive therapy may be used to treat benign nodules. The rationale is that nodule cells may remain responsive to TSH. Indeed, two meta-analyses suggest that thyroxine treatment may retard growth and sometimes shrink nodules in 25% of patients. 14,15 Suppressive therapy should not be considered until all nodules have been shown to be benign by biopsy. Given the increased risk of cardiac arrhythmia and evidence that subclinical hyperthyroidism can lead to loss of bone mass in postmenopausal women,16 TSH suppression should be used cautiously in older patients.
Well-differentiated thyroid cancers arise from follicular cells and encompass both papillary and follicular carcinomas. Papillary carcinomas are the most common thyroid cancer. They are unencapsulated, often multicentric, and bilateral in a third of cases. The most common sites of metastasis are regional lymph nodes and, less commonly, the lungs.
Follicular carcinomas represent 5%–10% of thyroid cancers. They are encapsulated, and the feature of malignancy is invasion of the capsule. Prognosis depends on the degree of invasion and differentiation, with poorly differentiated tumours having poorer outcomes. A subtype, the Hürthle-cell tumour, with eosinophilic mitochondria-rich cells, also has a poorer outcome. Follicular carcinomas can metastasise to bone, causing lytic lesions.
Well-differentiated thyroid carcinoma has an excellent prognosis, despite the presence of lymph-node metastases in 5%–20% of patients and distant metastases in 10%–15%. Ten-year survival is 80%–95%. Factors that worsen prognosis include male sex, advanced age, large tumour, poor differentiation, local invasion and distant metastases.8
Surgery: Initial management is surgical, with “total” thyroidectomy (postsurgical radioisotope scanning usually shows residual thyroid tissue) indicated for any malignancy with diameter > 1 cm. Thyroidectomy should be performed by an experienced surgeon, as this reduces complications, such as hypoparathyroidism and recurrent laryngeal nerve palsy. Although subtotal thyroidectomy has lower complication rates, thyroid remnant ablation with radioiodine is less successful when large amounts of thyroid tissue remain.
Remnant ablation: Retrospective studies indicate reduced recurrence rates and improved survival (at least in patients with follicular cancer or papillary cancer larger than 1 cm) when thyroid tissue remaining after surgery is ablated with orally administered radioiodine. This may not only destroy cancer cells in the neck and elsewhere, but also improve specificity of future radioiodine whole-body scans and serum thyroglobulin assays by removing normal thyroid cells.17 Radioiodine doses of 4000 MBq are often used for postsurgical ablation in Australia, but lower doses are often adequate. Ablation is most effective when endogenous TSH levels are maximal, usually 3–4 weeks after total thyroidectomy. Admission to a specific isolation room is required for 2–3 days, after which radioiodine levels have usually fallen to levels that permit discharge. It is usual practice to perform a whole-body scan at the time of discharge, using the therapeutic radioiodine dose as the imaging agent. This usually demonstrates uptake in the thyroid bed, salivary glands, gastrointestinal tract and bladder. Uptake in other areas indicates metastatic disease (Box 5).
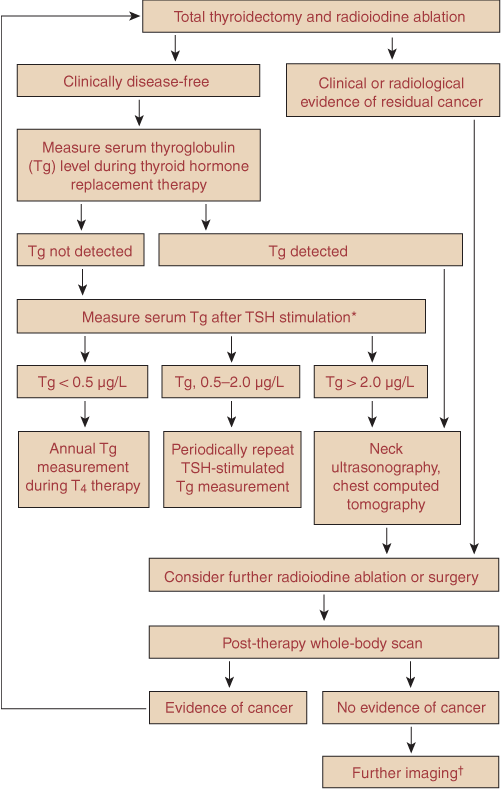
An algorithm for follow-up after treatment of differentiated thyroid cancer is shown in Box 6.
Clinical and biochemical follow-up: Clinical examination of the thyroid bed and regional lymph nodes may detect recurrence. Serum thyroglobulin levels are used to follow up patients treated by thyroidectomy and radioiodine ablation of the thyroid remnant. Tumour cells generally retain the ability to synthesise thyroglobulin in response to TSH stimulation. Unfortunately, a significantly elevated thyroglobulin level does not predict uptake of radioiodine and, conversely, tumour which concentrates radioiodine may produce insignificant amounts of thyroglobulin.18 Recombinant human TSH (rhTSH) maximally stimulates thyroglobulin secretion by residual or recurrent thyroid cancer and, when given by injection, increases the sensitivity of serum thyroglobulin measurements in detecting recurrent disease. It is as yet unclear under what conditions rhTSH, an expensive drug, will be made available in Australia.
131I whole-body scanning: Many centres repeat a whole-body scan 12 months after surgery and remnant ablation. We reserve this scanning for patients with measurable thyroglobulin levels and those with thyroglobulin autoantibodies, in whom thyroglobulin levels cannot be measured. TSH levels greater than 30 mIU/L are needed to maximise iodine uptake by residual or recurrent thyroid cancer. This is achieved by withholding thyroxine for 4 weeks, often with substitution of triiodothyronine for the first 2 weeks of the period to minimise symptoms of hypothyroidism. Alternatively, rhTSH injection allows whole body scanning without stopping thyroxine therapy. Care must be taken to ensure that no iodinated contrast medium has been administered in the 2 months before whole-body scan, as this reduces the sensitivity of the scan.
The sensitivity of scanning depends on the dose of 131I administered, but higher doses may reduce subsequent radioiodine uptake by tumour cells (“thyroid stunning”). Scans done after therapeutic doses of radioiodine (4000–6000 MBq) are the most sensitive.
Other imaging: Where there is evidence of metastatic disease (eg, raised thyroglobulin level) but no abnormal 131I uptake is detected on whole-body scan, ultrasonography or computed tomography may locate a tumour mass. Positron emission tomography using fluoro-18-deoxyglucose may detect lesions not seen on radioiodine scanning, and evidence is emerging that this is a negative prognostic indicator.19 Use of this technique has become common overseas and is increasing in Australia.
This tumour arises from the neuroendocrine C-cells of the thyroid and manufactures calcitonin, with the preoperative level correlating with the burden of disease. Fifty per cent of patients have lymph node metastases at presentation. Primary management is surgical. Thyroxine is needed as replacement rather than suppressive therapy, as the tumour cells are not under the influence of TSH. Prognosis is less favourable than for well-differentiated cancers, but, even so, survival is 86% and 78% at 5 and 10 years, respectively. Patients with distant metastases have only 36% 5-year survival.
Optimal management of metastatic disease is controversial and ranges from observation to aggressive surgery and chemoradiotherapy. Survival benefit with aggressive therapy is unclear.20
Medullary thyroid carcinoma represents just 5%–10% of thyroid carcinomas but is familial in up to 20% of cases, associated with the RET proto-oncogene. No mutation can be identified in 2% of affected families, while a germline mutation is discovered in 5%–6% of those with no family history.21 As implications for other family members are significant, most experts recommend genetic testing after appropriate counselling of all patients presenting with this carcinoma. Current guidelines advocate thyroidectomy in early childhood for known carriers of RET mutations. Patients must then be screened periodically for other features of the syndrome, such as hyperparathyroidism, pituitary disease (multiple endocrine neoplasia [MEN] type 1), and phaeochromocytoma (MEN2).
About 5% of thyroid cancers are anaplastic. In contrast to other forms of thyroid cancer, anaplastic thyroid carcinoma is one of the most aggressive tumours in humans, and death occurs at a mean of 6 months after diagnosis. An aggressive combination of chemotherapy, external beam radiotherapy and surgery is required to reduce strangulation, but the tumour is often widely metastatic and almost never curable, and therapy is aimed at palliation.22
This entity, usually a high-grade B-cell lymphoma, tends to affect middle-aged and older patients. All patients with thyroid lymphoma have a goitre, and most report rapid enlargement.23 Almost without exception there is evidence of lymphocytic infiltration of the gland in keeping with pre-existing Hashimoto’s thyroiditis, and close to half of all patients are found to be hypothyroid. Treatment is excision of the thyroid followed by combination chemotherapy. About 50% of patients are cured, while the remainder relapse, often with generalised lymphoma.
2: The “incidentaloma”
Incidental discovery of a small thyroid nodule is an increasing clinical problem. A small but significant proportion of these nodules are malignant, but there is persuasive evidence that they are rarely clinically significant. Occult thyroid cancers are commonly found at autopsy (mean, 36 cases per 1000 persons), while thyroid carcinoma has an incidence of 0.5–10 cases per 100 000 population,8 implying that most thyroid cancers do not become clinically apparent.
Recent guidelines for management of incidentally found thyroid nodules do not advise needle biopsy unless diameter is > 1 cm.9 However, many patients are concerned about the possibility of malignancy and request biopsy.
Benign findings are reassuring, as the subsequent risk of malignancy in that nodule is less than 1%. It is prudent to follow up patients with thyroid nodules that are benign on biopsy or have not undergone biopsy. In a small series of incidentally discovered lesions < 1 cm, no malignancies appeared after 5 years.10 Although nodules that enlarge are not necessarily malignant,11 most thyroid specialists would perform another biopsy.
3: Papillary thyroid carcinoma
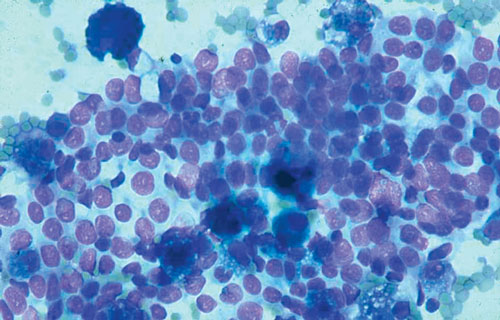
Fine-needle aspirate from a papillary thyroid carcinoma, showing cells with crowded and overlapping nuclei with inclusions (haematoxylin and eosin stain).
4: Case report — managing a thyroid nodule
 Presentation: A 38-year-old woman presented to her general practitioner after noticing a swelling on the left side of her neck which had gradually enlarged over the previous 18 months. She was otherwise well but concerned that she might have cancer. There was no relevant family history.
Presentation: A 38-year-old woman presented to her general practitioner after noticing a swelling on the left side of her neck which had gradually enlarged over the previous 18 months. She was otherwise well but concerned that she might have cancer. There was no relevant family history.
Examination: There was a 5 cm firm smooth mass on the right side of the larynx that moved with swallowing. No other abnormality was found.
Investigations: Thyroid function tests gave normal results. An ultrasound study confirmed a 48 mm solid mass in the right thyroid lobe. The remainder of the gland appeared normal. Fine-needle aspiration biopsy showed clumps of follicular cells but no colloid, consistent with a diagnosis of a follicular neoplasm.
Management: The patient was referred to a thyroid surgeon, who informed her that the nodule was probably benign but could be a follicular thyroid cancer. He recommended surgery and performed a right hemithyroidectomy. Frozen section of the 5 cm nodule showed a well-differentiated follicular lesion that was reported as an adenoma. However, examination of a paraffin section the following day showed capsular and vascular invasion by follicular cells. The diagnosis was revised to follicular cancer, and the surgeon advised removal of the left thyroid lobe, which was performed the following day.
The patient was reviewed by an endocrinologist, who recommended radioiodine ablation of remaining thyroid tissue. This was scheduled for a month later. In the interim, the patient took no thyroid hormone replacement therapy.
Plasma free thyroxine level fell to 2 pm/L (reference range [RR], 9–23 pmol/L) and plasma thyroid-stimulating hormone level rose to 94 mU/L (RR, 0.3–5 mU/L). After a negative pregnancy test, the patient was admitted to a radiation isolation room and given 4000 MBq of radioiodine.
Two days later, she underwent a whole-body gamma-camera scan, using the therapeutic radioiodine dose as the imaging agent. This showed a small amount of residual tissue in the thyroid bed and physiological uptake in salivary glands, stomach and bladder. There was no evidence of metastatic thyroid cancer.
The patient was discharged with thyroid hormone replacement therapy and returned home. To minimise exposure of her family to the remaining radioiodine, she avoided physical contact with her husband and children for the next week.
Course: The patient was reviewed every 3 months over the next year by the surgeon and endocrinologist. She remained clinically free of disease. Her thyroid function was also measured 3-monthly, and the dose of thyroxine adjusted to suppress plasma TSH levels. Serum thyroglobulin levels remained unmeasurable.
A year later, thyroxine was withdrawn for one month, and a whole-body radioiodine scan was performed. This showed physiological uptake and disappearance of the previously noted thyroid bed uptake.
Comment: The patient had a large, well-differentiated follicular thyroid cancer, which has a good prognosis. However, there is a small risk of recurrent disease, and she will require long-term follow-up.
- 1. Burgess JR. Temporal trends for thyroid carcinoma in Australia: an increasing incidence of papillary thyroid carcinoma (1982-1997). Thyroid 2002; 12: 141-149.
- 2. Brander A, Viikinkoski P, Nickels J, Kivisaari L. Thyroid gland: US screening in a random adult population. Radiology 1991; 181: 683-687.
- 3. Tan GH, Gharib H, Reading CC. Solitary thyroid nodule. Comparison between palpation and ultrasonography. Arch Intern Med 1995; 155: 2418-2423.
- 4. Belfiore A, La Rosa GL, La Porta GA, et al. Cancer risk in patients with cold thyroid nodules: relevance of iodine intake, sex, age, and multinodularity. Am J Med 1992; 93: 363-369.
- 5. Sarne D, Schneider AB. External radiation and thyroid neoplasia. Endocrinol Metab Clin North Am 1996; 25: 181-195.
- 6. Belfiore A, La Rosa GL. Fine-needle aspiration biopsy of the thyroid. Endocrinol Metab Clin North Am 2001; 30: 361-400.
- 7. Tollin SR, Mery GM, Jelveh N, et al. The use of fine-needle aspiration biopsy under ultrasound guidance to assess the risk of malignancy in patients with a multinodular goiter. Thyroid 2000; 10: 235-241.
- 8. Schlumberger MJ, Torlantano M. Papillary and follicular thyroid carcinoma. Baillieres Best Pract Res Clin Endocrinol Metab 2000; 14: 601-613.
- 9. Burguera B, Gharib H. Thyroid incidentalomas. Prevalence, diagnosis, significance, and management. Endocrinol Metab Clin North Am 2000; 29: 187-203.
- 10. Brander AE, Viikinkoski VP, Nickels JI, Kivisaari LM. Importance of thyroid abnormalities detected at US screening: a 5-year follow-up. Radiology 2000; 215: 801-806.
- 11. Alexander EK, Hurwitz S, Heering JP, et al. Natural history of benign solid and cystic thyroid nodules. Ann Intern Med 2003; 138: 315-318.
- 12. Alexander EK, Heering JP, Benson CB, et al. Assessment of nondiagnostic ultrasound-guided fine needle aspirations of thyroid nodules. J Clin Endocrinol Metab 2002; 87: 4924-4927.
- 13. Gharib H. Fine-needle aspiration biopsy of thyroid nodules: advantages, limitations, and effect. Mayo Clin Proc 1994; 69: 44-49.
- 14. Castro MR, Caraballo PJ, Morris JC. Effectiveness of thyroid hormone suppressive therapy in benign solitary thyroid nodules: a meta-analysis. J Clin Endocrinol Metab 2002; 87: 4154-4159.
- 15. Zelmanovitz F, Genro S, Gross JL. Suppressive therapy with levothyroxine for solitary thyroid nodules: a double-blind controlled clinical study and cumulative meta-analyses. J Clin Endocrinol Metab 1998; 83: 3881-3885.
- 16. Cooper DS. Clinical review 66: thyroxine suppression therapy for benign nodular disease. J Clin Endocrinol Metab 1995; 80: 331-334.
- 17. Mazzaferri EL. An overview of the management of papillary and follicular thyroid carcinoma. Thyroid 1999; 9: 421-427.
- 18. Torrens JI, Burch HB. Serum thyroglobulin measurement. Utility in clinical practice. Endocrinol Metab Clin North Am 2001; 30: 429-467.
- 19. Haugen BR, Lin EC. Isotope imaging for metastatic thyroid cancer. Endocrinol Metab Clin North Am 2001; 30: 469-492.
- 20. AACE/AAES medical/surgical guidelines for clinical practice: management of thyroid carcinoma. American Association of Clinical Endocrinologists. American College of Endocrinology. Endocr Pract 2001; 7: 202-220.
- 21. Modigliani E, Franc B, Niccolisire P. Diagnosis and treatment of medullary thyroid cancer. Baillieres Best Pract Res Clin Endocrinol Metab 2000; 14: 631-649.
- 22. Giuffrida D, Gharib H. Anaplastic thyroid carcinoma: current diagnosis and treatment. Ann Oncol 2000; 11: 1083-1089.
- 23. Burman KD, Ringel MD, Wartofsky L. Unusual types of thyroid neoplasms. Endocrinol Metab Clin North Am 1996; 25: 49-68.





Abstract
Thyroid nodules are common clinically (prevalence, about 5%) and even more common on ultrasound examination (about 25%).
About 5% of thyroid nodules are malignant.
Most thyroid cancers are well-differentiated papillary or follicular tumours with an excellent prognosis (10-year survival, 80%–95%).
The incidence of papillary thyroid cancer appears to be increasing on the east coast of Australia.
Fine-needle aspiration biopsy of the thyroid is the most cost-effective diagnostic tool.
Recommended initial management of all follicular carcinomas and of papillary carcinomas > 1.0 cm is total thyroidectomy followed by radioiodine ablation.
Most patients should be managed postoperatively with doses of thyroid hormone sufficient to suppress plasma levels of thyroid-stimulating hormone.
Recurrences can occur many years after initial therapy, and follow-up should be lifelong.