The past 20 years has seen an increase in the incidence of cancer in all age groups in Victoria. This increase has been more marked in adolescents and young adults than in any other age group. Cancer incidence increased by 30% in those aged 10–24 years between 1993 and 2001.1,2 A similar trend has been reported overseas.3 In addition, mortality rates in adolescents and young adults with cancer have been slow to decline.
Published studies have consistently shown that children and adolescents with a variety of cancer types have higher survival rates if treated in clinical trials and at specialised centres.4-6 Entry into clinical trials appears to be a more important determinant of outcome than the place of treatment.7,8 Given the poor outcomes for adolescents and young adults with cancer, access to, and treatment in, national clinical trials is of paramount importance in this age group.
The proportion of adolescents and young adults managed at specialised tertiary centres and treated in clinical trials in Victoria is not known. We felt it important to document current management, and to examine the outcomes of adolescents and young adults with cancer who receive their treatment in Victoria.
A total of 679 adolescents and young adults with cancer were identified from the Victorian Cancer Registry for the 5 years from 1992 to 1996. Fourteen patients were excluded: eight were not treated in Victoria; five had an incorrect diagnosis of cancer; and one received no treatment. This left 665 patients eligible for study entry. Questionnaires were completed for 576 (357 male, 219 female) eligible patients (87%).
The distribution of cancers and location of treatment for each age group is shown in Box 1.
Treatment was categorised according to management in a clinical trial, according to a treatment protocol, or neither. The results for each age group and all cancers are shown in Box 2 (a). Significantly more adolescents aged 10–19 years were treated within clinical trials in paediatric teaching hospitals than adult hospitals (38% and 3%, respectively; P < 0.005; 95% CI for difference, 25%–41%). Only 4% of young adults aged 20–24 years were treated within clinical trials.
There was no significant difference in overall 5-year survival between the three age groups for all cancers combined.
Treatment and 5-year survival were examined separately for each of the six cancers studied. Overall survival did not differ significantly between age groups for each cancer (Box 3).
Brain tumours: Few adolescents and young adults with brain tumours were treated within clinical trials (Box 2 [b]).
Bone tumours: Significantly more adolescents aged 10–15 years than those aged 16–19 years were treated within clinical trials (46% v 5.5%; P = 0.003; 95% CI for difference, 21%–64%; Box 2 [c]). Survival appeared to be improved if patients were treated at a paediatric hospital (Box 2 [c]), although these differences are completely confounded with age differences (almost all patients over 16 years were treated at adult hospitals).
Germ cell tumours: Few patients were treated in clinical trials (Box 3). There were no major differences in survival according to place of treatment.
Leukaemia and lymphoma: Recruitment into clinical trials decreased significantly with increasing age (Box 3). Survival was not associated with place of treatment.
Soft tissue tumours: There were insufficient numbers of patients with soft tissue tumours for statistical analysis.
Several international studies have shown that the outcome for adolescents and young adults with cancer is worse than for younger children.3,9 Our study has provided previously unavailable information on the patterns of management and outcomes of adolescents and young adults with cancer in Victoria. The study was not designed to examine factors other than recruitment to clinical trials that might be associated with cancer outcomes in this population, as differences between treatment locations were heavily confounded with age differences. Possible effects of treatment regimens are better examined within the context of randomised trials.
In our study, the overall 5-year survival in adolescents aged 16–19 years was not significantly different from that of patients aged 10–15 and 20–24 years.
Adolescents and young adults with cancer are treated at both adult and paediatric centres. In keeping with reports from the United States,10 83% of Victorian adolescents aged 10–15 years, and 14% of those aged 16–19 years are treated at paediatric institutions and have access to paediatric clinical trials. We found significantly fewer adolescents and young adults aged 16–19 years and 20–24 years were recruited to clinical trials when compared with those aged 10–15 years. Again, this result is in keeping with US reports, showing enrolment into both paediatric and adult cooperative group clinical trials was consistently lower in adolescents aged 15–21 years than in children aged 0–14 years.11 It is unclear why adolescents and young adults with cancer have such low rates of recruitment into clinical trials.
A recent British study confirmed that most oncologists and haematologists are committed to the concept of prospective randomised clinical trials.12 However, despite this, less than half the eligible patients aged 15–29 years were actually enrolled in national leukaemia trials. Reasons for lack of recruitment included clinician preference for one arm of a national trial, concurrent regional studies or non-trial protocols, and concerns about administrative issues. In our study, which includes patients enrolled in regional trials, we did not explore the reasons for lack of recruitment into clinical trials, but it is clear that barriers do exist.
In adolescents and young adults with acute lymphoblastic leukaemia, improved survival has been reported for patients treated on intensive paediatric protocols, suggesting that adolescents and young adults with this cancer may benefit from an “aggressive” paediatric-type regimen.13 In our study, the number of adolescents and young adults with cancer was too small to examine the influence of treatment on outcome. However, Kaplan–Meier survival analysis showed the 5-year survival for all cancers combined was similar across all age groups and was independent of place of treatment.
In adolescents and young adults with bone tumours, we demonstrated that treatment at a paediatric teaching hospital, where recruitment into clinical trials was more common, was associated with improved outcome. The pathophysiology, natural history and response to treatment in osteosarcoma is not influenced by age,14 but the prognosis in Ewing’s sarcoma does appear to worsen with age.15 In our study it was not possible to separate the influence of increasing age from the potentially adverse influence of poor recruitment into clinical trials on patient survival.
For some tumour types, such as germ cell tumours and lymphoma, where well established treatment protocols are in place, recruitment into clinical trials appears to have less effect on overall survival. In Victoria, most patients with germ cell tumours and lymphomas were treated according to well-established protocols. Survival appeared uniformly high across all age groups and settings.
About 1% of adolescents and young adults in our study failed to complete planned therapy because of non-compliance with treatment. We probably underestimated the extent of the problem. Despite completing planned therapy, adolescents and young adults may not have complied with oral self-administered chemotherapy, which can be a significant component of therapy for some cancers. Published studies of non-compliance with oral chemotherapy in children and adolescents have reported rates between 10%16 and 59%.17 Compliance is known to decrease with duration of therapy,18 and has a significant effect on patient survival.19 Non-compliance may account, in part, for the poor survival rates reported in adolescents and young adults with cancer.
We examined outcome in terms of survival, and did not attempt to address treatment-related morbidity or quality-of-life, both of which are major concerns for adolescents and young adults. Although short-term and long-term adjustment difficulties have been described,11 there is a paucity of information on quality-of-life in adolescents and young adults with cancer. Increasingly, clinical trials are incorporating measures of quality-of-life and, as survival improves, these will become a more important focus of outcome.
Our study has confirmed that, in Victoria, adolescents and young adults are less likely to be enrolled in clinical cancer trials than younger children. There is clearly a need to address this problem.
One reason for lack of recruitment into clinical trials might be the absence of suitable open studies.20 Many of the cancers seen in patients aged 10–24 years are common in the paediatric age group, but more than 25% of young adults will have tumours not routinely studied or treated by paediatric oncology cooperatives, and which adult oncologists regularly manage. Enrolment into appropriate adult trials should be encouraged. Appropriate management of adolescents and young adults requires communication and cooperation between all adult and paediatric specialists involved in cancer treatment. This could be achieved by developing a cancer resource network to provide information about current clinical studies to paediatric and adult oncologists, other specialists, adolescents and young adults with cancer and their families.9
1: The distribution of cancers and location of treatment for 576 adolescents and young adults with cancer
|
Age group |
||||||||||
|
10–15 years |
16–19 years |
20–24 years |
||||||||
Completed questionnaires |
167/177* (94%)† |
144/165* (87%)† |
265/323* (82%)† |
||||||||
Distribution of cancers (No. [%]‡) |
|
|
|
||||||||
Bone tumours |
26 (16%) |
18 (13%) |
16 (6%) |
||||||||
Brain tumours |
35 (21%) |
19 (13%) |
41 (16%) |
||||||||
Germ cell tumours |
10 (6%) |
28 (19%) |
72 (27%) |
||||||||
Leukaemia |
43 (26%) |
23 (16%) |
26 (10%) |
||||||||
Lymphoma |
40 (24%) |
49 (34%) |
90 (34%) |
||||||||
Soft tissue tumours |
13 (8%) |
7 (5%) |
20 (8%) |
||||||||
Treatment location† |
|
|
|
||||||||
Paediatric hospital |
139 (83%) |
20 (14%) |
4 (2%) |
||||||||
Adult hospital |
25 (15%) |
121 (84%) |
253 (96%) |
||||||||
Private rooms |
3 (2%) |
3 (2%) |
8 (3%) |
||||||||
* Denominator is total number of eligible patients. † P = 0.023. ‡ Percentage of patients with completed questionnaires. |
|||||||||||
2: Management of all 576 adolescents and young adults with cancer, those with bone tumours (survival also shown) and those with brain tumours according to treatment regimen and place of treatment
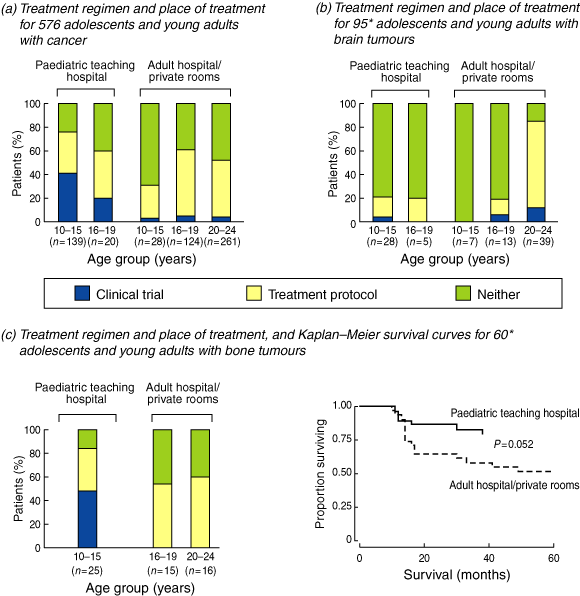
* Groups totalling three patients or fewer (eg, three patients aged 16–19 years with bone tumours treated at a paediatric hospital; one patient aged 10–15 years with a bone tumour treated at an adult hospital) were not included.
3: Percentage of adolescents and young adults in clinical trials, and overall 5-year survival in all age groups by tumour type
|
Age group |
||||||||||
|
10–15 years |
16–19 years |
20–24 years |
||||||||
Bone tumours |
|
|
|
||||||||
Number |
26 |
18 |
16 |
||||||||
Clinical trial |
12 (46%)* |
1 (5.5%)* |
0* |
||||||||
Overall survival (95% CI) |
81% (59%–91%) |
44% (22%–65%) |
63% (35%–81%) |
||||||||
Brain tumours |
|
|
|
||||||||
Number |
35 |
19 |
41 |
||||||||
Clinical trial |
1 (3%) |
1 (6%) |
1 (2%) |
||||||||
Overall survival (95% CI) |
83% (66%–92%) |
58% (33%–76%) |
68% (52%–80%) |
||||||||
Germ cell tumours |
|
|
|
||||||||
Number |
10 |
28 |
72 |
||||||||
Clinical trial |
0 |
0 |
4 (6%) |
||||||||
Overall survival (95% CI) |
80% (41%–95%) |
93% (74%–98%) |
85% (74%–91%) |
||||||||
Leukaemias |
|
|
|
||||||||
Number |
43 |
23 |
26 |
||||||||
Clinical trial |
28 (65%)* |
3 (13%)* |
3 (12%)* |
||||||||
Overall survival (95% CI) |
56% (40%–69%) |
61% (38%–77%) |
50% (30%–67%) |
||||||||
Lymphomas |
|
|
|
||||||||
Number |
40 |
49 |
90 |
||||||||
Clinical trial |
16 (40%)* |
5 (10%)* |
2 (2%)* |
||||||||
Overall survival (95% CI) |
85% (70%–93%) |
80% (65%–88%) |
82% (73%–89%) |
||||||||
Soft tissue tumours |
|
|
|
||||||||
Number |
13 |
7 |
20 |
||||||||
Clinical trial |
1 (8%) |
0 |
1 (5%) |
||||||||
Overall survival (95% CI) |
77% (44%–92%) |
71% (26%–92%) |
75% (50%–89%) |
||||||||
* P < 0.05 |
|||||||||||
Received 19 February 2003, accepted 8 September 2003
- Anne E Mitchell1
- Deborah L Scarcella2
- Gemma L Rigutto3
- David M Ashley4
- Vicky J Thursfield5
- Graham G Giles6
- Maree Sexton7
- 1 Department of Haematology and Oncology, Royal Children’s Hospital, Melbourne, VIC.
- 2 Cancer Epidemiology Centre, The Cancer Council, Melbourne, VIC.
- 3 Paediatric Radiation Oncology, Peter MacCallum Cancer Institute, Melbourne, VIC.
We thank Andrea Dozzi, Michael Ng, Nikki Cross and Gillian Hoogeveen for their assistance with data collection, and Professor John Carlin and Susan Donath for statistical advice. We gratefully acknowledge the support of The Cancer Council Victoria and the doctors who completed the questionnaires.
Anne Mitchell was supported by a grant from the Bluey Day Appeal, and Associate Professor Ashley was supported by a National Health and Medical Research Council practitioner fellowship. The supporting sources had no involvement in the study design, data collection, analysis and interpretation, or in writing the article.
- 1. Giles G, Whitfield K, Thursfield V, Staples M, editors. Canstat: cancer in Victoria 1993. Melbourne: Anti-Cancer Council of Victoria Epidemiology Centre; Anti-Cancer Council of Victoria, 1996.
- 2. Giles G, Thursfield V, editors. Canstat: cancer in Victoria 2001. Melbourne: The Cancer Council Victoria Epidemiology Centre; The Cancer Council Victoria, 2003.
- 3. Bleyer WA, Tejeda H, Murphy SB, et al. National cancer clinical trials: children have equal access; adolescents do not. J Adolesc Health 1997; 21: 366-373.
- 4. Wagner HP, Dingeldein-Bettler I, Berchthold W, et al. Childhood NHL in Switzerland: incidence and survival of 120 study and 42 non-study patients. Med Pediatr Oncol 1995; 24: 281-286.
- 5. Meadows AT, Kramer S, Hopson R, et al. Survival in childhood acute lymphoblastic leukaemia: effect of protocol and place of treatment. Cancer Invest 1983; 1: 49-55.
- 6. Stiller CA, Benjamin S, Cartwright RA, et al. Patterns of care and survival for adolescents and young adults with acute leukaemia — a population-based study. Br J Cancer 1999; 79: 658-665.
- 7. Stiller CA, Eatock EM. Patterns of care and survival for children with acute lymphoblastic leukaemia diagnosed between 1980 and 1994. Arch Dis Child 1999; 81: 202-208.
- 8. Stiller CA, Draper GJ. Treatment centre size, entry to clinical trials, and survival in acute lymphoblastic leukaemia. Arch Dis Child 1989; 64: 657-661.
- 9. Bleyer WA. Cancer in older adolescents and young adults: epidemiology, diagnosis, treatment, survival, and importance of clinical trials. Med Pediatr Oncol 2002; 38: 1-10.
- 10. Ross JA, Severson RK, Pollock BH, Robison LL. Childhood cancer in the United States: a geographical analysis of cases from the paediatric cooperative clinical trials group. Cancer 1996; 77: 201-207.
- 11. Reaman HG, Bonfiglio J, Krailo M, et al. Cancer in adolescents and young adults. Cancer 1993; 71 Suppl 10: 3206-3209.
- 12. Benjamin S, Kroll ME, Cartwright RA, et al. Haematologist’s approaches to the management of adolescents and young adults with acute leukaemia. Br J Haematol 2000; 111: 1045-1050.
- 13. Boissel N, Auclerc MF, Lheritier V, et al. Should adolescents with acute lymphoblastic leukemia be treated as old children or young adults? Comparison of the French FRALLE-93 and LALA-94 trials. J Clin Oncol 2003; 21: 774-780.
- 14. Meyers PA, Gorlick R. Osteosarcoma. Pediatr Clin North Am 1997; 44: 973-989.
- 15. Rodriguez-Galindo C, Spunt SL, Pappo AS. Treatment of Ewing sarcoma family of tumors: current status and outlook for the future. Med Pediatr Oncol 2003; 40: 276-287.
- 16. Lancaster D, Lennard L, Lilleyman JS. Profile of non-compliance in lymphoblastic leukaemia. Arch Dis Child 1997; 76: 365-366.
- 17. Smith SD, Roase D, Trueworthy RC, Lowman JT. A reliable method for evaluating drug compliance in children with cancer. Cancer 1979; 43: 169-173.
- 18. Tebbi CK, Cummings KM, Zevon MA, et al. Compliance of pediatric and adolescent cancer patients. Cancer 1986; 58: 1179-1184.
- 19. Richardson JL, Shelton DR, Krailo M, Levine AM. The effect of compliance with treatment on survival among patients with hematologic malignancies. J Clin Oncol 1990; 8: 356-364.
- 20. Goldman S, Stafford C, Weinthal J, et al. Older adolescents vary greatly from children in their route of referral to the pediatric oncologist and national trials. In: Grunberg SM, editor. Proceedings of the American Society of Clinical Oncology; 2000, May 20–23; New Orleans, Louisiana. Alexandria, Va: American Society of Clinical Oncology, 2000: 450a.





Abstract
Objectives: To describe the location of treatment, recruitment to clinical trials and outcomes for adolescents and young adults treated for cancer in Victoria.
Design and setting: Retrospective review of all adolescents and young adults aged 10–24 years diagnosed with cancer between 1992 and 1996, identified from the Victorian Cancer Registry.
Main outcome measures: Treatment regimen (clinical trial, treatment protocol or neither), compliance with treatment and 5-year survival.
Results: Questionnaires were completed for 576 of 665 eligible adolescents and young adults (87% response rate). Recruitment into clinical trials decreased with increasing age. Adolescents aged 10–19 years were more likely to be recruited to a clinical trial if treated at a paediatric hospital. For all cancers, 5-year survival was similar across the age groups and was not influenced by the place of treatment. Only 1% of adolescents and young adults failed to complete planned therapy due to non-compliance.
Conclusions: Despite a similar incidence of cancer to that in younger children, adolescents and young adults with cancer are poorly recruited into clinical trials in Victoria. Establishment of a cancer resource network in Victoria may provide information to both paediatric and adult oncologists about currently available clinical trials.