Most hospitals have cardiac arrest teams that respond to in-hospital cardiac arrests using modern technology and standardised protocols. However, survival to hospital discharge in patients with in-hospital cardiac arrests has remained stable at between 14.7% (United States) and 16.7% (United Kingdom) for 30 years.1 As several studies of in-hospital cardiac arrests suggest that signs of clinical and physiological instability may precede the arrest,2-4 introducing an intensive care-based hospital-wide preventive approach (a medical emergency team [MET]) might decrease the incidence of cardiac arrests and, consequently, hospital mortality. We tested this hypothesis by conducting a prospective trial comparing these outcome measures before and after introducing a MET.
The MET system was structured so that any member of the hospital staff could activate it. The MET included the duty intensive care fellow and a designated intensive care nurse. If available, the receiving medical registrar was encouraged to attend. An ICU consultant was available from 08:00 until 20:00, and attended if requested. After hours, an intensive care consultant was available within 15–30 minutes for attendance if required.
The criteria for MET activation (Box 1) were displayed prominently in each ward. The MET was activated by a pager call and by a public announcement internal communication call “Medical emergency team to Ward X”. The MET carried an emergency pack with drugs and equipment for resuscitation and endotracheal intubation.
A computerised statistical package (Statview) was used for data analysis and descriptive statistics.5 Fisher’s exact test was used for comparisons between the “before” and “after” periods, and the χ2 test was used for three-way comparison of the “before” period, the “after” (intervention) period, and the additional seasonal control period.
Box 2 shows the number and distribution of medical and surgical admissions during the period before and after the MET was introduced. There were no significant differences.
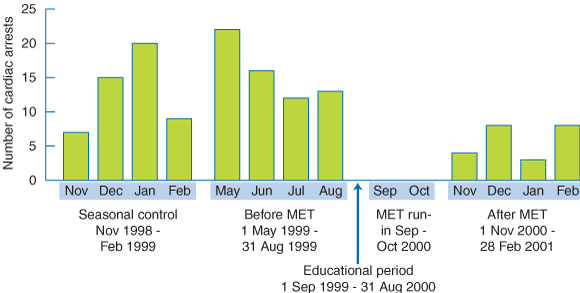
In the “before” period, there were 8974 medical admissions, compared with 8377 in the intervention period. There were 33 cardiac arrests among medical patients in the “before” period, compared with 11 in the intervention period (relative risk reduction [RRR], 66%; P = 0.002).
There were 12 116 surgical admissions in the “before” period, compared with 12 544 in the intervention period. The number of cardiac arrests in surgical patients decreased from 30 to 11 (RRR, 63%; P = 0.003). Therefore, the total reduction in the number of cardiac arrests was from 63 to 22 (RRR, 65%; P < 0.001). None of the patients suffering a cardiac arrest and receiving treatment had “do not resuscitate” orders explicitly written in the patient progress notes.
In the same 4-month period (seasonal control period) 2 years before the introduction of the MET, there were 51 cardiac arrests, which was not significantly different from the number of cardiac arrests in the “before” period (P = 0.3), but significantly different from the number in the intervention period (P = 0.001). Monthly cardiac arrest data during these periods are presented in Box 4.
In the intervention period, there were 99 MET calls triggered by different, and sometimes multiple, criteria for physiological instability (“worried about the patient”, 46; haemoglobin desaturation on pulse oximetry, 37; change in conscious state, 28; low systolic blood pressure, 35; heart rate change, 20; respiratory rate change, 18; and oliguria, 2).
The MET attended each call within a mean (SD) period of 4.5 (2.2) minutes, and was in attendance for a mean (SD) period of 19 (18) minutes. Different units within the hospital activated the MET in a relatively uniform way (Box 5), but the MET was significantly more frequently activated during the evening (48 calls from 16:00 to midnight versus 31 from 08:00 to 16:00 versus 20 from midnight to 08:00; P < 0.001).
The MET initiated and completed a variety of therapeutic, investigational and procedural interventions (Box 6). Of the 99 MET calls, 18 resulted in an emergency ICU or high dependency unit (HDU) admission, with a total of 109 days and 18 days spent in ICU and HDU, respectively. The ICU/HDU stay for these patients ranged from 1 to 31 days, with 10 patients staying for 3 days or less.
Of the 24 patients who died after a MET call, 10 were designated “not for resuscitation” before the call, and two were so designated after the MET call. Three patients had a cardiac arrest at the time of the MET call and died during the call. The other nine patients, who were for full resuscitation, died a median of 19 days after the call (range, 2–57 days). Considering “not for resuscitation” orders and cardiac arrests separately, survival after a MET call was 89.6%, and none of the patients who were for full resuscitation died within 24 hours of a MET call.
There were 37 in-hospital deaths related to cardiac arrests in the “before” period and 16 in the intervention period (RRR for cardiac arrest deaths, 56%; P = 0.008). There were a total of 302 inpatient deaths in the “before” period compared with 222 deaths in the intervention period (RRR, 26%; P = 0.004) (Box 3). In the same 4-month period 2 years before the introduction of the MET, there were 275 deaths (P = 0.27 compared with the “before” period; P = 0.018 compared with the intervention period).
After cardiac arrest, and in the absence of any change in the cardiac arrest treatment protocol, survivors in the “before” period required a total of 163 ICU bed-days and 1353 hospital bed-days, and survivors in the intervention period required 33 ICU bed-days (RRR, 80%; P < 0.001) and 159 hospital bed-days (RRR, 88%; P < 0.001) (Box 3).
We found that the incidence of in-hospital cardiac arrests decreased by two-thirds after the introduction of a MET. This reduction, in both medical and surgical patients, is internally consistent and suggests a widespread impact, irrespective of admission diagnosis. It is also consistent with previous observations that between 50% and 84% of in-hospital cardiac arrests are preceded by physiological instability.2-4,6,7 By appropriately responding to physiological instability, most in-hospital cardiac arrests can be prevented.
With the MET there was also a more than 50% reduction in the number of cardiac arrest-related deaths, and a reduced number of postcardiac-arrest bed-days. For our institution, this would mean a yearly decrease of close to 3500 bed-days. This suggests that a MET is associated with major cost savings and increased hospital efficiency.
Our institution was able to continue to implement and sustain the MET system after the study period by adding one dedicated MET fellow to the intensive care staff allocation. This staff member responds to MET calls and collects data, making possible continuing education and auditing.
Introduction of the MET was associated with a 26% reduction in overall hospital mortality (three lives/1000 admissions). To our knowledge, this is the first before-and-after study of any intervention that shows an impact on all-cause hospital mortality. This effect was only partly accounted for by the impact of the MET on cardiac arrests. The MET might, therefore, confer other benefits, such as increasing awareness of the consequences of physiological instability. It is also possible that the educational program to introduce the MET had an impact on the care of acutely unwell patients.
It is important to consider our study’s limitations. First, this trial was not double blind, or placebo-controlled or randomised. It is not possible to have a double-blind MET intervention, and introducing “sham” intervention as placebo was ethically untenable, and “contamination” (so-called Hawthorne effect) would have been inevitable. Finally, a traditional, patient randomisation study of the MET would be ethically, scientifically and logistically impossible in a single hospital.
Our favourable findings may have been due to a high incidence of cardiac arrests in the control period or an abnormally low seasonal incidence in the intervention period. Australian data show a cardiac arrest incidence ranging from 36 to 51 per 10 000 admissions.2,8 In the “before” period, there were 30 cardiac arrests per 10 000 hospital admissions, and there was no statistically significant seasonal variation in the incidence of cardiac arrests in our hospital. Furthermore, the 4-month MET intervention period included, by chance, 3 months immediately after the start of the working year for new interns (a possible seasonal bias against the MET), whereas the control period did not.
The reduction in cardiac arrests was not due to “reclassification” of cardiac arrests into MET calls. There were three true cardiac arrests which occurred during a MET intervention, and these arrests were counted as such. Furthermore, most of the interventions were technically “simple”, suggesting that timely intervention (within minutes rather than hours) at the time of deterioration might also decrease the complexity of care required. Together with the dominance of the “worried” criterion for activation of the MET, this also suggests that some relatively simple acute interventions may appear too technically demanding to junior medical or nursing staff, or that such staff might lack the experience to recognise that these interventions are needed immediately.
Similarly, it is possible that our overall in-hospital mortality was high during the “before” period and was simply restored to standard levels by the MET, or fell because of seasonal variation. In Australia, data from other large hospitals show an overall crude mortality rate between 138 and 184 deaths per 10 000 admissions.8 Our crude mortality rate in the “before” period was 143 deaths per 10 000 admissions, and there was no statistically significant seasonal variation in in-hospital mortality.
Our findings within a single institution might not apply to other hospitals. Institution-specific heuristics and unique administrative features may have enhanced the impact of the MET approach. However, our institution has all the organisational, structural and logistic features of a typical tertiary referral hospital. Another possibility is that our implementation of a MET may have differed from that of other institutions,9-11 but whether implementation has an impact on its efficacy is not known. We believe that our approach is simple and low cost. It is also possible that the decrease in cardiac arrests was secondary to some other improvements in patient care between the “before” and “after” periods. However, there were no changes in the structure, referral pattern or activity of our hospital, with the total number of admissions during the two study periods remaining essentially unchanged (< 1% change in the denominator for the study outcomes). Furthermore, there were no changes in “not for CPR” policy, hospital admission policy, discharge practices or surgical casemix during the study. We are also not aware of any improvements or advances in medical or surgical treatment that could explain a greater than 60% reduction in cardiac arrests and a 25% reduction in overall mortality.
Another recent study showing a possible beneficial effect of a MET9 had several methodological shortcomings.12 Despite other indirect supportive evidence,10,11,13-17 the MET approach has not yet been adopted by most hospitals in Australia or elsewhere, and controversy continues concerning its safety and effectiveness.8,12
In conclusion, introducing an ICU-based MET in a teaching hospital decreased the incidence of and deaths from cardiac arrests, postcardiac-arrest bed-days, as well as overall mortality. Further testing of this approach is now needed in a variety of hospital and geographical settings.
1: Criteria for initiation of a MET call*
If one of these is present call 7777 and ask for the MET
Staff member is worried about the patient
Acute change in heart rate to < 40 or > 130 beats/min
Acute change in systolic blood pressure to < 90 mmHg
Acute change in respiratory rate to < 8 or > 30 breaths/min
Acute change in pulse oximetry saturation to < 90%, despite oxygen administration
Acute change in conscious state
Acute change in urine output to < 50 mL in 4 hours.
*Criteria were listed on a large red poster placed prominently in all wards.
2: Hospital population, patients having major surgery* and types of surgery, before and after introducing the medical emergency team (MET). Data are number of patients or procedures (95% CI), unless otherwise indicated
|
Before MET (1/5/99 – 31/8/99) |
After MET (1/11/00 – 28/2/01) |
|||||||||
Medical admissions |
8 974 (8 834–9 114) |
8 377 (8 239–8 516) |
|||||||||
Surgical admissions |
12 116 (11 976–12 256) |
12 544 (12 405–12 683) |
|||||||||
Patients receiving major surgery |
1 127 (1 065–1 189) |
1 067 (1 006–1 128) |
|||||||||
Men |
660 (628–692) |
613 (582–644) |
|||||||||
Women |
467 (434–500) |
454 (422–484) |
|||||||||
Mean age (years) |
60.7 (59.5–61.9) |
60.2 (59–61.4) |
|||||||||
Patients > 75 years |
315 (286–344) |
281 (253–309) |
|||||||||
Major surgical procedures |
1 369 (1 301–1 437) |
1 313 (1 246–1 380) |
|||||||||
Cardiac surgery |
188 (163–213) |
141 (119–163) |
|||||||||
Thoracic surgery |
142 (120–164) |
117 (97–137) |
|||||||||
General surgery |
288 (259–317) |
318 (288–348) |
|||||||||
Orthopaedic surgery |
253 (225–281) |
285 (256–314) |
|||||||||
Vascular surgery |
160 (137–183) |
132 (111–153) |
|||||||||
Neurosurgery |
147 (125–169) |
111 (92–130) |
|||||||||
Plastic surgery |
77 (61–93) |
84 (67–101) |
|||||||||
Other (includes liver transplantation) |
114 (94–134) |
125 (104–146) |
|||||||||
*Major surgery is defined as any surgery requiring a hospital stay > 48 hours. |
|||||||||||
3: Changes in number of cardiac arrests, bed-days and mortality, before and after introducing the medical emergency team (MET)
|
Before MET |
After MET |
Difference
|
Relative risk ratio |
|||||||
No. of cardiac arrests |
63 |
22 |
41 (23–59) |
0.35 (0.22–0.57) |
|||||||
Deaths from cardiac arrest |
37 |
16 |
21 (7–35) |
0.43 (0.26–0.70) |
|||||||
No. of days in ICU after cardiac arrest |
163 |
33 |
130 (110–150) |
0.20 (0.13–0.33) |
|||||||
No. of days in hospital after cardiac arrest |
1353 |
159 |
1194 (1119–1269) |
0.11 (0.09–0.13) |
|||||||
Inpatient deaths |
302 |
222 |
80 (37–123) |
0.74 (0.70–0.79) |
|||||||
ICU = intensive care unit. |
|||||||||||
5: Proportion of medical emergency team (MET) calls from the different hospital units
General surgery | 21% |
Neurology | 13% |
Cardiology | 9% |
Nephrology | 8% |
Cardiothoracic surgery | 8% |
Othopaedic surgery | 6% |
Spinal | 6% |
Plastic surgery | 4% |
Oncology | 4% |
General medicine | 3% |
Other (vascular surgery, thoracic medicine, haematology, neurosurgery, liver transplant) | 18% |
6: Number of interventions and procedures implemented by the Medical Emergency Team
Interventions | ||
Nasopharyngeal/oropharyngeal suctioning and additional oxygen | 21 | |
Administration of IV fluid bolus | 18 | |
Administration of IV frusemide bolus | 11 | |
Initiation of non-invasive positive pressure ventilation by mask | 9 | |
Nebulised salbutamol | 8 | |
Temporary ventilation by bag and mask | 6 | |
Suctioning of tracheostomy tube | 6 | |
Initiation of IV glyceryltrinitrate infusion | 6 | |
Administration of anticonvulsants | 5 | |
Administration of IV vasopressors | 5 | |
Insertion of a Guedel airway | 4 | |
Administration of IV morphine | 4 | |
Insertion of a urinary catheter | 4 | |
Cardioversion | 3 | |
Administration of IV β blockers or digoxin | 3 | |
Administration of IV naloxone | 2 | |
Transfer to operating room with ongoing resuscitation | 2 | |
Administration of IV metoclopramide | 2 | |
Administration of IV ranitidine | 2 | |
Administration of IV insulin or glucose | 2 | |
Insertion of new tracheostomy tube | 1 | |
Insertion of minitracheostomy tube | 1 | |
Acute transfusion of red cells | 1 | |
Administration of dexamethasone | 1 | |
Administration of intravenous magnesium | 1 | |
Administration of atropine | 1 | |
Removal of central venous catheter | 1 | |
Acute investigations | ||
Chest x-ray | 14 | |
Electrocardiogram | 16 | |
Computed tomography scan | 4 | |
Arterial blood gases | 36 | |
Urea, creatinine, electrolytes and liver function tests | 40 | |
Invasive procedures | ||
IV line insertion | 18 | |
Arterial line insertion | 5 | |
Endotracheal intubation | 3 | |
Central venous catheter insertion | 3 |
IV = intravenous.
Received 18 February 2003, accepted 2 July 2003
- Rinaldo Bellomo1
- Donna Goldsmith2
- Shigehiko Uchino3
- Jonathan Buckmaster4
- Graeme K Hart5
- Helen Opdam6
- William Silvester7
- Laurie Doolan8
- Geoffrey Gutteridge9
- Austin and Repatriation Medical Centre, Melbourne, VIC.
We thank our Medical Records Department for their assistance with this project. This study was funded by a grant from the Quality Improvement Branch of the Acute Health Care section of the Victorian Department of Human Services.
None identified.
- 1. Saklayen M, Liss H, Markert R. In-hospital cardiopulmonary resuscitation. Medicine 1995; 74: 163-175.
- 2. Buist MD, Jarmolowski E, Burton PR, et al. Recognising clinical instability in hospital patients before cardiac arrest or unplanned admission to intensive care. Med J Aust 1999; 171: 22-25.
- 3. Franklin C, Mathew J. Developing strategies to prevent in-hospital cardiac arrest: analyzing responses of physicians and nurses in the hours before the event. Crit Care Med 1994; 22: 244-247.
- 4. Shein RMH, Hazday N, Pena M, et al. Clinical antecedents to in-hospital cardiopulmonary arrest. Chest 1990; 98: 1388-1392.
- 5. Statview [computer program], version 4. Berkeley, Calif: Abacus Concepts Inc, 1996.
- 6. Hodgetts TJ, Kenward G, Vlachonikolis IG, et al. The identification of risk factors for cardiac arrest and formulation of activation criteria to alert a medical emergency team. Resuscitation 2002; 54: 125-131.
- 7. Smith AF, Wood J. Can some in-hospital cardio-respiratory arrests be prevented? A prospective survey. Resuscitation 1998; 37: 133-137.
- 8. Bristow P, Hillman KM, Chey T, et al. Rates of in-hospital arrests, deaths, and intensive care admissions: the effect of a medical emergency team. Med J Aust 2000; 173: 236-240. <eMJA full text>
- 9. Buist M, Moore GE, Bernard SA, et al. Effects of a medical emergency team on reduction of incidence of and mortality from unexpected cardiac arrests in hospital: preliminary study. BMJ 2002; 324: 387-390.
- 10. Lee A, Bishop G, Hillman K, Daffum K. The medical emergency team. Anaesth Intensive Care 1995; 23: 183-186.
- 11. Hurihan F, Bishop G, Hillman KM, et al. The medical emergency team: a new strategy to identify and intervene in high-risk patients. Clin Intensive Care 1995; 6: 269-272.
- 12. Smith GB, Nolan J. Medical emergency teams and cardiac arrests in hospital. Results may have been due to education of ward staff. BMJ 2002; 324: 1215.
- 13. Goldhill DR, White SA, Sumner A. Physiological values and procedures in the 24 hours before ICU admission from the ward. Anaesthesia 1999; 54: 529-534.
- 14. McQuillan P, Pilkington S, Allan A, et al. Confidential inquiry into quality of care before admission to intensive care. BMJ 1998; 316: 1853-1858.
- 15. Hillman KM. Redefining resuscitation. Aust N Z J Med 1998; 28: 759-760.
- 16. Daly FFS, Sidney KL, Fatovich DM. The medical emergency team (MET): a model for the district general hospital. Aust N Z J Med 1998; 28: 795-798.
- 17. Henderson SO, Ballestreros D. Evaluation of a hospital-wide resuscitation team: does it increase survival for in-hospital cardiopulmonary arrest? Resuscitation 2001; 48: 111-116.





Abstract
Objective: To determine the effect on cardiac arrests and overall hospital mortality of an intensive care-based medical emergency team.
Design and setting: Prospective before-and-after trial in a tertiary referral hospital.
Patients: Consecutive patients admitted to hospital during a 4-month “before” period (May–August 1999) (n = 21 090) and a 4-month intervention period (November 2000 –February 2001) (n = 20 921).
Main outcome measures: Number of cardiac arrests, number of patients dying after cardiac arrest, number of postcardiac-arrest bed-days and overall number of in-hospital deaths.
Results: There were 63 cardiac arrests in the “before” period and 22 in the intervention period (relative risk reduction, RRR: 65%; P < 0.001). Thirty-seven deaths were attributed to cardiac arrests in the “before” period and 16 in the intervention period (RRR: 56%; P = 0.005). Survivors of cardiac arrest in the “before” period required 163 ICU bed-days versus 33 in the intervention period (RRR: 80%; P < 0.001), and 1353 hospital bed-days versus 159 in the intervention period (RRR: 88%; P < 0.001). There were 302 deaths in the “before” period and 222 in the intervention period (RRR: 26%; P = 0.004).
Conclusions: The incidence of in-hospital cardiac arrest and death following cardiac arrest, bed occupancy related to cardiac arrest, and overall in-hospital mortality decreased after introducing an intensive care-based medical emergency team.