Prudent use of antibiotics is central to controlling the increasing problem of antibiotic resistance in bacteria. Promoting prudent use is a core strategy advocated by the Australian Joint Expert Advisory Committee on Antibiotic Resistance (JETACAR)1 and is of major public health significance.
Use of broad-spectrum antibiotics has been repeatedly linked to the emergence of antibiotic-resistant organisms. In particular, use of third-generation cephalosporins is a risk factor for infection with methicillin-resistant Staphylococcus aureus, Clostridium difficile, enterococci and resistant gram-negative bacilli.2,3 These antibiotics, along with vancomycin and metronidazole, have been significantly associated with vancomycin-resistant enterococci (VRE).4 Periods of increased cephalosporin use have coincided with increased rates of VRE isolation.5
In 1999, a statewide evaluation of use of the third-generation cephalosporins cefotaxime and ceftriaxone (CEFX) was undertaken in Victorian hospitals.6 These antibiotics were found to be used more frequently at the Royal Melbourne Hospital than at other comparable teaching hospitals, with only 26% of courses being concordant with accepted indications in the national antibiotic guidelines.9 The agents were often used for surgical prophylaxis, with all these courses non-concordant with guidelines. The most common indication was respiratory tract infections, with most of these courses non-concordant with guidelines (50% of cases showed no abnormality on chest x-ray, and 11% did not meet the criteria for severe pneumonia) (unpublished data).
We sought to improve CEFX use at the Royal Melbourne Hospital by developing a computerised antimicrobial approval system with an educational component. We assessed CEFX use before and after introduction of this intervention.
A multidisciplinary subcommittee of the hospital's Drug and Therapeutics Committee was formed, comprising a drug usage evaluation pharmacist, a clinical pharmacist (infectious diseases), an infectious diseases physician, an infectious diseases registrar, an emergency physician and information technology specialists. This subcommittee developed a multifaceted strategy to improve CEFX prescribing.
CEFX was added to the hospital's list of restricted antimicrobial agents. It was removed from operating theatres and general wards, but not from the emergency department (ED), intensive care unit (ICU) or infectious diseases ward. Prescriptions for CEFX were required to be endorsed with an Antimicrobial Approval Number (AAN). This is a code comprising the authoriser's initials, date of prescription and duration of approved use.
A web-based form for generating these numbers was introduced for CEFX prescriptions for indications recommended in the national antibiotic guidelines.7 This form was interactive and linked to the antibiotic guidelines. It required prescribers to choose the drug and indication from drop down lists (Box 1).
Computer-generated AANs were not provided for indications not listed. Prescribers seeking AANs for these indications were asked to telephone the infectious diseases registrar.
A 24-hour grace period was allowed if prescribers were unable to gain approval through the web-based system or to contact an infectious diseases registrar. The antimicrobial approval system did not apply to the ICU, where other initiatives were undertaken to assist antimicrobial prescribing.
Feedback was sought from hospital medical staff to identify problems two months after the system was introduced.
The progress of the system was monitored in two ways. Firstly, monthly gross use figures for CEFX (defined daily doses [DDDs] per 1000 occupied acute case bed days) were generated from the pharmacy dispensing system and reported to the Antimicrobial Subcommittee.
Secondly, a concurrent, observational evaluation of CEFX use in patients commencing a course of these drugs was conducted during seven days in September 1999 (pre-intervention) and 14 days in July 2001 (about four months post-intervention) using the Methods described by Robertson et al.6 Courses were assessed for concordance with the current edition of Therapeutic guidelines: antibiotic (version 10 in 1999, and version 11 in 2001).7,9
Prescribing data were fed back to doctors once the approval system was established. Hospital units were given reports on their use of broad-spectrum cephalosporins and the proportion of use for which approval numbers were obtained in the previous month. Sub-sequently, the correlation between the indication "severe community-acquired pneumonia" and abnormal chest x-ray was examined. Letters were sent to prescribers who twice or more entered severe pneumonia as the indication when the chest x-ray was formally reported as normal at the time of prescribing.
Monthly CEFX use in the hospital fell to about half previous use on introduction of the system, a result sustained for 15 months (Box 2). While there was no significant change in CEFX use in the ICU (where the intervention was not undertaken) (P = 0.68), there was a significant reduction in use in the wards (mean, 38.3 DDDs/1000 bed days pre-intervention v 17.6 DDDs/1000 bed days post-intervention; P = 0.002).
The number of AANs per month ranged from 39 to 71, accounting for 36% to 62% of grams of CEFX prescribed. However, despite regular feedback in the ED, only 4%–17% of grams of CEFX prescribed were accounted for by AANs.
The proportion of approvals that were obtained using the web-based form rather than by telephone increased from 56% in April 2001 (one month after introduction of the system) to 90% in September 2001 (six months after its introduction) and remained in the 85%–95% range thereafter.
Results of the indication-linked audits of CEFX use are summarised in Box 3. After the intervention, use became more consistent with national antibiotic guidelines (P < 0.002), and use for surgical prophylaxis was nil. In particular, the proportion of patients treated empirically with CEFX for respiratory tract infection who did not have an abnormality on chest x-ray decreased from 50% (18/36) pre-intervention to 27% (11/41) post-intervention.
Use of other broad-spectrum β-lactam antibiotics and quinolones showed no consistent increase. In contrast, both gentamicin and benzylpenicillin use increased significantly — gentamicin from a mean of 30.0 DDDs/1000 bed days pre-intervention to 48.3 DDDs/1000 bed days post-intervention (P = 0.0001), and benzylpenicillin from a mean of 20.7 DDDs/1000 bed days to 28.4 DDDs/1000 bed days (P = 0.01) (Box 2).
We found that introduction of a multifaceted strategy, including a simple computerised antimicrobial approval system with an educational component, was successful in reducing CEFX use in our hospital and increasing concordance with prescribing guidelines. CEFX use for surgical prophylaxis was effectively eliminated, while CEFX use for respiratory infections without evidence of pneumonia on chest x-ray was markedly decreased. The changes were sustained over 15 months of follow-up.
Features of the strategy that may have contributed to its success include educational programs, restrictions on CEFX availability in the wards, and the antimicrobial approval system itself. Education alone (lectures and tutorials) has been generally ineffective in achieving sustained change,12 but was essential to our strategy to ensure that changes in prescribing practice were acceptable and understood by prescribers. Restricted availability on wards had more direct effects on pharmacists and nurses who obtained the drugs, reminding them of the new process, than prescribers. The approval system aims to be a disincentive to non-considered use of broad-spectrum agents, to educate through links to national guidelines, and to act as a surveillance tool allowing feedback to clinicians. Sustainability is one of the greatest challenges in modifying prescribing. We believe the ongoing approval system together with feedback to clinicians explained much of the sustained improvement.
Commitment to the program by key staff, adequate staff time for educational programs and endorsement by senior hospital clinicians and management were fundamental to its success.
Antimicrobial approval and restriction systems have been effective in controlling antibiotic use elsewhere, but may require significant resources and, if inefficient, can cause great inconvenience.13 Some hospitals in the United States have successfully used computer-assisted decision support systems linked with electronic prescribing to implement antibiotic practice guidelines.14,15 These sophisticated systems generate alerts if inappropriate agents are chosen for identified pathogens and give recommended antibiotic choices by indication and local consensus guidelines. However, these systems were "purpose built", extremely costly to implement, and not easily set up elsewhere. Our system is simpler and has been inexpensive to develop to this stage. However, it would benefit from expansion to other restricted drugs and improved reporting capacity.
A recent review of strategies to improve hospital prescribing reported that "multifaceted interventions aimed at barriers to good prescribing have the greatest chance of success".13 Multifaceted strategies have been recommended to contain antibiotic resistance by the World Health Organization16 and JETACAR.1 Such a strategy, including guidelines, educational activities, and rapid feedback to prescribers about inappropriate use, was successful in reducing overuse of third-generation cephalosporins at the John Hunter Hospital, Newcastle, NSW.17
We developed our strategy with the knowledge that many experienced hospital clinicians disagreed with some recommendations of the national antibiotic guidelines. The pre-intervention audit helped identify non-controversial areas of inappropriate use. These were targeted by educational programs. We accepted less than full compliance, hoping that it would increase with time and education. There has been little apparent resentment by clinicians.
The approval system was least used in the ED. As ED consultants predicted that time pressures in this department would make a web-based approval system impractical, a simple paper-based alternative was developed, and educational sessions arranged, but compliance was limited. We tried to improve participation through involving a clinical pharmacist in the ED and through physician feedback, with little success. As many antibiotic courses were commenced in the ED, this is an important ongoing challenge.
Introduction of the system was complicated by non-standardised software in ward computers, which was overcome relatively easily. For routine indications, prescribers overwhelmingly used the web-based rather than the telephone approval system. Informal feedback on acceptability to prescribers at educational meetings showed that they occasionally "cheated", particularly when consultants indicated to junior staff a preference for CEFX for non-standard indications.
A concern is that a computerised system may diminish discussion about antimicrobial resistance and use between prescribers and infectious diseases physicians. However, the system aimed to encourage discussion of non-standard indications, and use of infectious diseases consultative services did not decrease, while the "presence" of infectious diseases physicians at educational meetings increased.
Although we did not incorporate the effects of autocorrelation or any seasonal effects in our analysis, we believe the reduction in cephalosporin use is compelling. The greatest potential benefit of improved prescribing is in reducing the development of antibiotic-resistant organisms, but surveillance of this resistance would have required more resources than were available.
We did not attempt detailed costing of the intervention, but note that software development costs were about $6000, while education was included in the routine activities of the Infectious Diseases Service, Pharmacy and Drug Usage Evaluation Program. The post-intervention audit involved about 12 person-weeks, and ongoing maintenance and audit occupy one person-day per month. The increase in gentamicin use increased demand on the aminoglycoside dosing service.
The approval system has potential benefits beyond improving use of broad-spectrum cephalosporins. The web-based form may save time for both prescribers and authorising doctors compared with telephone-based systems. Other hospitals have expressed interest in the system; a multicentre system would potentially allow research on the epidemiology of antibiotic use and its relationship to antibiotic resistance genes. The system is a potential model for promoting optimal use of other restricted or expensive pharmaceuticals within the hospital. An expanded system incorporating other restricted antibiotics and linking prescribing to laboratory Results and decision support systems is currently being developed.
1: Web-based form for generating Antimicrobial Approval Numbers for ceftriaxone/cefotaxime prescriptions

Prescribers were required to log into the form using their hospital network log-in code as identification, to enter the patient's medical record number and check that correct patient details were displayed, and then to choose the drug and indication from drop down lists.

Each listed indication was linked to the relevant section of the antibiotic guidelines accessible on the Clinicians Health Channel,8 a health information portal provided by the Victorian Department of Human Services. The form was interactive. For example, prescribers who wished to use ceftriaxone for community-acquired pneumonia had to indicate that the chest x-ray showed a pulmonary infiltrate and that specified criteria for severe pneumonia were met.
2: Use of ceftriaxone/cefotaxime and other antibiotics at Royal Melbourne Hospital before and after introduction of the antimicrobial approval system
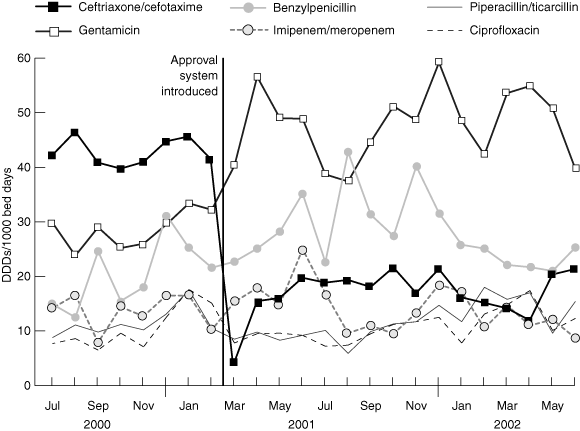
DDD = defined daily dose. After introduction of the approval system in March 2001, there was a significant decrease in use of ceftriaxone/cefotaxime (P = 0.002) and significant increases in use of gentamicin (P = 0.0001) and benzylpenicillin (P = 0.01).
3: Ceftriaxone/cefotaxime use and concordance with national antibiotic guidelines at Royal Melbourne Hospital and other hospitals
|
Royal Melbourne Hospital |
Other Melbourne teaching hospitals6 |
|||||||||
|
A |
B |
C |
D |
|||||||
Study date |
Jul 2001 |
Sep 1999 |
Sep 1999 |
Sep 1999 |
Sep 1999 |
Sep 1999 |
|||||
Length of enrolment (days) |
14 |
7 |
7 |
7 |
7 |
7 |
|||||
Number of courses |
74 |
79 |
45 |
39 |
39 |
22 |
|||||
Defined daily doses/1000 bed days |
22 |
35 |
26 |
22 |
39 |
12 |
|||||
Number of courses started in operating theatres |
0 |
17 (22%) |
16 (36%) |
0 |
0 |
0 |
|||||
Concordance with guidelines* |
|
|
|
|
|
|
|||||
All courses |
51%† |
26%† |
33% |
28% |
18% |
45% |
|||||
Empirical courses for respiratory tract infection |
17/41 (41%) |
8/36 (22%) |
11/27 (41%) |
8/23 (35%) |
4/20 (20%) |
6/13 (46%) |
|||||
* Therapeutic guidelines: antibiotic (11th edition7 in 2001 and 10th edition9 in 1999). † P < 0.002 Fisher's exact test. |
|||||||||||
Received 2 September 2002, accepted 6 February 2003
- Michael J Richards1
- Lyn-Li Lim2
- Marion B Robertson3
- Nicholas R Jones4
- Simone E Taylor5
- Margarida M Duarte6
- Dale A Kerr7
- Graham J Stanton8
- Peter D Ritchie9
- Jonathan G A Dartnell10
- 1 Royal Melbourne Hospital, Parkville, VIC.
- 2 Therapeutic Guidelines Limited, North Melbourne, VIC.
We acknowledge the assistance of Dr Jim Black (Victorian Infectious Diseases Service, Royal Melbourne Hospital) in performing the statistical analysis.
None identified. Development of the antimicrobial approval system was funded within the budget of the Royal Melbourne Hospital.
- 1. Joint Expert Technical Advisory Committee on Antibiotic Resistance (JETACAR). The use of antibiotics in food-producing animals: antibiotic-resistant bacteria in animals and humans. Report of JETACAR. Commonwealth Department of Health and Aged Care and Commonwealth Department of Agriculture, Fisheries and Forestry — Australia. Canberra: Ausinfo, 1999. Available at: http://www.health.gov.au/pubs/jetacar.pdf (accessed Mar 2003).
- 2. Paterson DL, Playford EG. Should third-generation cephalosporins be the empirical treatment of choice for severe community-acquired pneumonia in adults? Med J Aust 1998; 168: 344-348.
- 3. Ferguson JK. Vancomycin-resistant enterococci: causes and control. Med J Aust 1999; 171: 117-118.
- 4. Dahms RA, Johnson EM, Statz CL, et al. Third-generation cephalosporins and vancomycin as risk factors for postoperative vancomycin-resistant enterococcus infection. Arch Surg 1998; 133: 1343-1346.
- 5. Quale J, Landman D, Atwood E, et al. Experience with a hospital-wide outbreak of vancomycin-resistant enterococci. Am J Infect Control 1996; 24: 372-379.
- 6. Robertson MB, Korman TM, Dartnell JGA, et al. Ceftriaxone and cefotaxime use in Victorian hospitals. Med J Aust 2002; 176: 524-529. <eMJA full text>
- 7. Writing Group. Therapeutic guidelines: antibiotic. 11th ed. Melbourne: Therapeutic Guidelines Limited, 2000.
- 8. Clinicians Health Channel. Victorian Government Department of Human Services. Available at: http://www.clinicians.vic.gov.au/ (accessed Mar 2003).
- 9. Writing Group. Therapeutic guidelines: antibiotic. 10th ed. Melbourne: Therapeutic Guidelines Limited, 1998.
- 10. Dean A, Dean J, Coulombier D, et al. Epi Info, version 6: a word processing, database, and statistics program for public health on IBM-compatible microcomputers. Atlanta: Centers for Disease Control and Prevention, 1996.
- 11. Altman D. Practical statistics for medical research. London: Chapman and Hall, 1991.
- 12. Bero LA, Grilli R, Grimshaw JM, et al. Closing the gap between research and practice: an overview of systematic reviews of interventions to promote the implementation of research findings. The Cochrane Effective Practice and Organization of Care Review Group. BMJ 1998; 317: 465-468.
- 13. Dartnell JGA. Activities to improve hospital prescribing. Aust Prescriber 2001; 24: 29-31.
- 14. Pestotnik SL, Classen DC, Evans RS, Burke JP. Implementing antibiotic practice guidelines through computer-assisted decision support : clinical and financial outcomes. Ann Intern Med 1996; 124: 884-890.
- 15. Shojania KG, Yokoe D, Platt R, et al. Reducing vancomycin use utilizing a computer guideline: Results of a randomized controlled trial. J Am Med Inform Assoc 1998; 5: 554-562.
- 16. World Health Organization. WHO global strategy for containment of antimicrobial resistance 2001. Available at: http://www.who.int/emc/amrpdfs/WHO_Global_Strategy_English.pdf (accessed Mar 2003).
- 17. MacDonald J, Ferguson J. Hospital education influences prescribing at John Hunter Hospital. Aust Prescriber 2001; 24: 32.





Abstract
Objective: To achieve sustained improvement in use of cefotaxime and ceftriaxone (CEFX) in a major teaching hospital, as measured against national antibiotic guidelines.
Design and setting: Pre- and post-intervention survey of CEFX use in the Royal Melbourne Hospital, a tertiary hospital in Melbourne, Victoria.
Intervention: Web-based antimicrobial approval system linked to national antibiotic guidelines was developed by a multidisciplinary team and implemented in March 2001.
Main outcome measures: Change in rate of CEFX use (defined daily doses [DDDs] per 1000 acute occupied bed days) over 8 months pre- and 15 months post-intervention; concordance of indication for CEFX with national antibiotic guidelines pre- and post-intervention.
Results: CEFX use decreased from a mean of 38.3 DDDs/1000 bed days pre-intervention to 15.9, 18.7 and 21.2 DDDs/1000 bed days at 1, 4 and 15 months post-intervention. Concordance with national antibiotic guidelines rose from 25% of courses pre-intervention to 51% within 5 months post-intervention (P < 0.002). Gentamicin use also increased, from a mean of 30.0 to 48.3 DDDs/1000 bed days (P = 0.0001).
Conclusion: The web-based antimicrobial approval system achieved a sustained reduction in CEFX use over 15 months as well as increased prescribing concordance with antibiotic guidelines. It has potential for linking to electronic prescribing and for wider use for other drugs, as well as for research into the epidemiology of antibiotic use.