The most severely disadvantaged population group in Australia are remote-dwelling Aboriginals and Torres Strait Islanders.1 Their age distribution2 and the illness patterns in their children resemble those in developing countries. In the Northern Territory, the Australian State with the highest proportion of Aboriginal people,2 respiratory illness is the most common hospital separation diagnosis in infants and children aged 1–5 years,3 and accounts for 15.2% of all hospital admissions of Aboriginals (the highest category after childbirth and pregnancy).2 However, these data under-represent the true burden of acute respiratory disease, as many of the children with pneumonia are treated in their remote communities.
There are no data to quantify respiratory morbidity after hospitalisation for acute pneumonia in this population. Respiratory illness in childhood is a risk factor for chronic respiratory morbidity and pulmonary dysfunction in adulthood,4 and childhood pneumonia is a risk factor for bronchiectasis later in life. There is an unacceptably high rate of chronic suppurative lung disease in Aboriginal children living in remote communities.5,6
A follow-up chest radiograph in patients with pneumonia can detect other pulmonary disease and hence determine appropriate management.7 However, the use of chest radiography in uncomplicated pneumonia (and particularly the need for a follow-up x-ray film) has been debated.8 Canadian guidelines for management of pneu-monia in children encourage the use of chest radiography,9,10 but UK guidelines discourage its use.10 Published Australian paediatric guidelines do not exist as yet, but it is common practice for children hospitalised in Australian paediatric centres to have an initial and follow-up chest radiograph.
For children in remote communities, a follow-up chest radiograph often involves a plane flight, overnight accommodation, major disruption to family life and high costs. If a predischarge chest radiograph could predict ongoing respiratory morbidity, this could potentially reduce the economic and social costs associated with following up all children.
The aim of our study was to assess follow-up respiratory care by (i) evaluating short-term outcomes, and (ii) determining whether a predischarge chest radiograph can predict respiratory morbidity of children hospitalised with alveolar lobar changes on a chest radiograph.
Alice Springs Hospital is the only hospital in Alice Springs (population, 27 629) and the central Australian region, covering an area of about one million km2. Aboriginal Australians account for about a third of the population of central Australia,3 and most live in small, remote communities.
A new diagnosis: the child had not previously been diagnosed with the condition at the index presentation.
Reactive airways disease: the child had recurrent wheezing that reversed with β2-agonist therapy.
Bronchiectasis: chest CT scans in the absence of acute illness showed the changes of bronchiectasis according to the criteria of Naidich et al.11
Chronic suppurative lung disease: the child had a prolonged (> 4 months) history of productive and/or moist cough that responded to intravenous antibiotic treatment, but a chest CT scan did not show the changes of bronchiectasis.
Data were analysed using the Statistical Package for the Social Sciences (SPSS).12 Groups were compared using the Mann–Whitney test, and 2-tailed P levels less than 0.05 were considered significant. Relative risks were calculated according to Altman.13 For calculation of weight and height z scores, data based on the World Health Organization growth reference14 were used.
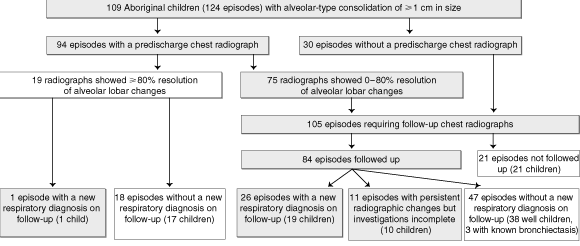
Over the 6-month period, 1002 paediatric separations were recorded (763 Aboriginal, 239 non-Aboriginal), with a respiratory condition as the principal diagnosis in 381. The radiological cri-teria were fulfilled in 136, but eight episodes were excluded (severe neurological illness or aspiration-like event in five, no consent in three), leaving 128 episodes for inclusion in the study. These 128 episodes occurred in 113 children, as 14 children had recurrent admissions (13 children with two admissions and one child with three admissions). Four children (single episodes) were not Aboriginal and, given the small number, were omitted from the follow-up data. Data were analysed for number of episodes (n = 124) or number of children (n = 109). A flowchart of the outcomes for the 109 children in our study is given in Box 1.
Comorbidities were common (Box 2), as was previous hospitalisation for respiratory infections (71% of episodes). In 112 episodes (90.3%), respiratory illness was the primary reason for admission; in the remainder, the reasons for admission were gastroenteritis (7; 5.6%), febrile convulsion (2; 1.6%), and one each (0.8%) surgery, abdominal pain, and "failure to thrive". None of the children who had recurrent admissions were previously known to have had bronchiectasis.
A predischarge chest radiograph was available in only 94 episodes (75.8%). "Complete" resolution was present in 19 episodes (15.3%). A follow-up radiograph was required in the remaining 105 episodes (Box 1). Of these, only 33 episodes (31.4%) had a "successful" follow-up (follow-up within 2 months). The two most common reasons were no appointment requested by the junior doctors, as detailed in the discharge summary (38 episodes), and the child not attending the appointment (21 episodes).
Within a year after the acute admission, follow-up occurred (84 episodes) or was no longer required (19 episodes with "near-complete" resolution on the predischarge radiograph) for a total of 103 episodes (83.1%), with "opportunistic" follow-up occurring in 38 episodes. However, not all children had their respiratory follow-up completed within the study period, as 10 children (11 episodes) were still being evaluated for persistent symptoms accompanied by radiological changes (Box 1). Respiratory diagnoses in the 78 children in whom follow-up was completed are shown in Box 3.
Our study showed that Aboriginal children admitted to hospital have a high burden of previous respiratory illness and comorbidities. Of the children with completed follow-up, a new treatable respiratory condition was found in 25.6% within 12 months of their index illness. When the predischarge chest radiograph showed "minimal" resolution, there was a high likelihood of chronic respiratory disease being detected at the subsequent follow-up, although our results should be interpreted with caution, given the wide confidence intervals.
Aetiology and carriage studies have been performed in central Australian Aboriginal children with acute lower respiratory infection,15,16 but there are no published clinical follow-up data. In contrast to the situation in developing countries, morbidity rather than mortality is the major concern in children with acute lower respiratory infections in disadvantaged communities living in affluent countries.17
Although our eventual follow-up rate of 83.1% of episodes (with extra effort being made for our study) was acceptable, the follow-up rate at 2 months was low at 31.4%. This, combined with the high rate (25.6%) of respiratory morbidity in children followed up, strongly suggests that an appropriate program to improve respiratory and other ambulatory services would be beneficial for the early diagnosis and appropriate management of these children. Even if it is assumed that the 21 children lost to follow-up did not have any respiratory morbidity, the proportion of children with new respiratory morbidity is still high at 22.7% (20 of 88 children).
To our knowledge, this is the first study to document the need for vigilant follow-up of Indigenous children hospitalised with lobar alveolar changes. There are several possible reasons for the high rates of disease found on follow-up.
Firstly, we suspect that many of the radiological changes noted were chronic rather than acute. Other disadvantaged groups in affluent countries, such as Alaskan Inuit children in the United States,18 also have high levels of chronic respiratory morbidity, as do children in developing countries.
Secondly, Aboriginal children living in remote communities have high rates of purulent nasal and ear discharge,19 as well as dense nasopharyngeal colonisation with respiratory pathogens.16 Microaspiration of these secretions may contribute not just to high rates of lower respiratory infections, but also to the prevalence of chronic suppurative lung disease.
Thirdly, an increased risk of lower respiratory infections is associated with a greater number of house occupants, poor quality housing, macro- and micromalnutrition, inadequate water supply, and exposure to biomass combustion and tobacco smoke, conditions which apply to many remote Indigenous communities.20 These conditions may also contribute to incomplete resolution of lower respiratory tract infections.
A critical question for future research is whether treatment with antibiotics for 5 days is inadequate, resulting in persistent low-grade bacterial bronchitis. It is possible that, as described by Cole,21 incomplete eradication of bacteria in early bacterial pneumonia, as well as persistent airway inflammation, promotes the development of bronchiectasis.
In ambulatory settings, chest radiographs have limited value in acute res-piratory infections.22 Guideline recommendations against using predischarge and repeat chest radiographs are based generally on Level D evidence (expert opinion or a lower evidence level).9 Our prospective study is limited by a small numbers of subjects. However, children whose predischarge chest radiograph showed "minimal" resolution of changes appeared to have a significantly increased risk of bronchiectasis, chronic suppurative lung disease, and other respiratory morbidity, on follow-up within 12 months of their index illness. However, the risk was not reduced when "near-complete" resolution occurred. The lack of a predictive value of "near-complete" resolution may relate to the short-term nature of the study, as subsequent respiratory infections, rather than the index illness, could influence the development of chronic suppurative lung disease. A predischarge chest radiograph, if predictive of morbidity, would indeed be highly beneficial in remote or rural settings. The question of whether delayed clearing predicts bronchiectasis or new respiratory morbidity cannot be answered from our study, as repeated timed chest radiographs could not be done in our setting. For the otherwise well child, follow-up radiographs are not recommended, but such recommendations may be inappropriate for central Australian Aboriginal children, as many have recurrent pneumonia, comorbidities and socioeconomically poor living conditions. Clinical predictors (other than malnutrition and exposure to pollutants) for the development of chronic respiratory morbidity, not identifiable in this limited study, would be beneficial in determining which children need radiological follow-up.
Until further studies are performed our findings suggest that, in our setting, all children with alveolar lobar radiological abnormalities should be followed up clinically, and those with persistent symptoms, or whose predischarge chest radiograph shows "minimal" resolution, should also have a follow-up chest radiograph. There is an urgent need to reduce lower respiratory infections and to determine better means of preventing long-term respiratory complications in such populations.
2: Characteristics of 109 Aboriginal children, admitted to hospital over a 6-month period, who had alveolar lobar abnormalities (124 episodes)
Variable |
Median (range) or number (%) |
||||||||||
Patient characteristics (n = 109) |
|
||||||||||
Sex (M : F) |
68 : 41 |
||||||||||
Number with weight for age z score less than – 2SD |
15 (13.8%) |
||||||||||
Median weight for age z score |
– 0.88 (– 3.15, 2.58) |
||||||||||
Median height for age z score |
– 0.63 (– 4.30, 6.80) |
||||||||||
Median weight for height z score |
– 0.48 (– 3.80, 2.26) |
||||||||||
Gestation at birth |
|
||||||||||
Number at term (> 37 weeks) |
86 (78.9%) |
||||||||||
Number preterm (30–36 weeks) |
12 (11.0%) |
||||||||||
Number preterm (24–29 weeks) |
1 (0.9%) |
||||||||||
Unknown |
10 (9.2%) |
||||||||||
Median birthweight (kg) |
3.19 (0.79, 3.24) |
||||||||||
Number with known bronchiectasis |
3 (2.7%) |
||||||||||
Number with a mother who smoked (excludes pitjuri [traditional tobacco] chewing) |
21 (19.3%) |
||||||||||
Episode characteristics (n = 124) |
|
||||||||||
Patient's median age (years) |
1.78 (0.02, 13.30) |
||||||||||
Number collected by Royal Flying Doctor Service |
63 (50.8%) |
||||||||||
Median duration of hospitalisation (days) |
5 (2, 48) |
||||||||||
Number requiring oxygen therapy |
32 (25.8%) |
||||||||||
Median length of time oxygen required (hours) |
0 (0, 168) |
||||||||||
Number given prior antibiotics |
48 (38.7%) |
||||||||||
Median neutrophil count (x109/L) |
13.1 (1.5, 44.2) |
||||||||||
Median time taken for temperature to return to normal (hours) |
0 (0, 120) |
||||||||||
Number with comorbidities |
|
||||||||||
Anaemia |
64 (51.5%) |
||||||||||
Chronic suppurative otitis media |
45 (36.3%) |
||||||||||
Diarrhoea |
37 (29.8%) |
||||||||||
Urinary tract infection |
2 (1.6%) |
||||||||||
Median number of previous admissions |
2 (0–21) |
||||||||||
Median time since last admission (years) |
0.52 (0, 9) |
||||||||||
Number with previous admission for bronchiolitis |
54 (43.5%) |
||||||||||
Number with previous admission for pneumonia |
69 (55.6%) |
||||||||||
3: Follow-up respiratory diagnoses in the 78 children whose follow-up was completed*
Respiratory diagnosis at follow-up |
No. of children |
||||||||||
No respiratory morbidity documented |
55 |
||||||||||
Bronchiectasis |
|
||||||||||
New diagnosis |
9 |
||||||||||
Previously diagnosed |
3 |
||||||||||
Chronic suppurative lung disease |
5 |
||||||||||
Reactive airway disease |
5 (2)† |
||||||||||
Pulmonary abscess |
1 |
Bronchiolitis obliterans |
0 (1)† |
||||||||
|
* At least one new diagnosis was found in 20 of the 78 children (25.6%) with follow-up completed.
| |||||||||||
- Anne B Chang1
- John P Masel2
- Naomi C Boyce3
- Paul J Torzillo4
- 1 Royal Children's Hospital, Brisbane, QLD.
- 2 Flinders University, Northern Territory Clinical School, Alice Springs, NT.
- 3 Royal Prince Alfred Medical Centre, Newtown, NSW.
The successful follow-up of these children was only possible through the dedication and assistance of Carmel Hattch and Jennie Thurley, to whom we are very grateful.
None identified.
- 1. Eades SJ. Reconciliation, social equity and Indigenous health. Med J Aust 2000; 172: 468-469. <MJA full text>
- 2. Fisher D, Ruben AR. Funding of Northern Territory public hospitals. Aust Health Rev 2002; 25: 189-193.
- 3. Dempsey KE, Condon JR. Mortality in the Northern Territory 1979-1997. Darwin, Territory Health Services, 1999.
- 4. Cooreman J, Redon S, Levallois M, et al. Respiratory history during infancy and childhood, and respiratory conditions in adulthood. Int J Epidemiol 1990; 19: 621-627.
- 5. Chang AB, Grimwood K, Mulholland EK, Torzillo PJ. Bronchiectasis in Indigenous children in remote Australian communities [position statement]. Med J Aust 2002; 177: 200-204. <MJA full text>
- 6. Chang AB, Masel JP, Boyce NC, et al. Non-CF bronchiectasis-clinical and HRCT evaluation. Pediatr Pulmonol 2003. In press.
- 7. Franquet T. Imaging of pneumonia: trends and algorithms. Eur Respir J 2001; 18: 196-208.
- 8. Grant CC. Pneumonia in children: becoming harder to ignore. N Z Med J 1999; 112: 345-347.
- 9. Jadavji T, Law B, Lebel MH, et al. A practical guide for the diagnosis and treatment of paediatric pneumonia. CMAJ 1997; 156: S703-S711.
- 10. Russell G. Community acquired pneumonia. Arch Dis Child 2001; 85: 445-446.
- 11. Naidich DP, McCauley DI, Khouri NF, et al. Computed tomography of bronchiectasis. J Comput Assist Tomogr 1982; 6: 437-444.
- 12. SPSS [computer program], version 7.5. Chicago, Ill: SPSS Inc, 1997.
- 13. Altman DG. Practical statistics for medical research. London: Chapman & Hall, 1991: 266.
- 14. Epi Info 2000, version 1.1. Atlanta, Ga: Centers for Disease Control and Prevention, 2000.
- 15. Torzillo P, Dixon J, Manning K, et al. Etiology of acute lower respiratory tract infection in Central Australian Aboriginal children. Pediatr Infect Dis J 1999; 18: 714-721.
- 16. Gratten M, Manning K, Dixon J, et al. Upper airway carriage by Haemophilus influenzae and Streptococcus pneumoniae in Australian Aboriginal children hospitalised with acute lower respiratory infection. Southeast Asian J Trop Med Public Health 1994; 25: 123-131.
- 17. Grant CC, Scragg R, Tan D, et al. Hospitalization for pneumonia in children in Auckland, New Zealand. J Paediatr Child Health 1998; 34: 355-359.
- 18. Singleton R, Morris A, Redding G, et al. Bronchiectasis in Alaska Native children: causes and clinical courses. Pediatr Pulmonol 2000; 29: 182-187.
- 19. Torzillo PJ, Waterford JE, Hollows FC, Jones DL. Respiratory disease amongst Aborigines in the Pilbara. Int J Epidemiol 1983; 12: 105-106.
- 20. Health is life. Report on the Inquiry into Indigenous Health. House of Representatives Standing Committee on Family and Community Affairs, Parliament of the Commonwealth of Australia, 2000. Available at: http: //www.aph.gov.au/house/committee/fca/indhea/reportfinal.pdf (accessed Mar 2003).
- 21. Cole, PJ. Inflammation: a two-edged sword — the model of bronchiectasis. Eur J Respir Dis Suppl 1986; 147: 6-15.
- 22. Swingler GH, Zwarenstein M. Chest radiograph in acute lower respiratory infections in children. Cochrane Review 2003; The Cochrane Library, Issue 1, 2003. Oxford: Update Software.





Abstract
Objectives: To describe the short-term outcomes in Aboriginal children admitted to hospital with radiological alveolar lobar changes; and determine whether predischarge chest radiography can predict respiratory morbidity found at follow-up.
Design, participants, setting: Prospective cohort study of Aboriginal children admitted to Alice Springs Hospital between October 2000 and April 2001 with alveolar lobar abnormalities (area of consolidation, ≥ 1 cm) on chest radiographs. Participants were to have a predischarge radiograph and be followed up for 12 months.
Main outcome measures: Comorbidities, follow-up rate, and new respiratory disease found at follow-up.
Results: Of 113 children hospitalised with radiological alveolar lobar changes, 109 were Aboriginal. Their median age was 1.8 years (range, 0.2 months–13.3 years), and 124 episodes were recorded. Comorbidities were common in these children (anaemia, 51.5%; suppurative otitis media, 37.3%). The follow-up rate one year after admission was 83.1% of episodes. New treatable chronic respiratory morbidity was found in 20 (25.6%) of the 78 children with completed follow-up. Predischarge chest radiographs were predictive of all chronic respiratory morbidity when they showed no or minimal resolution (0–20% resolution) (relative risk, 7.43; 95% CI, 2.07–26.60).
Conclusions: Central Australian Aboriginal children admitted to hospital with alveolar changes on chest radiographs have a substantial burden of chronic respiratory illness, and should be clinically followed up for early detection and management of chronic respiratory morbidity. A predischarge radiograph is useful, and patients whose radiograph shows no or minimal resolution should have a follow-up x-ray film.