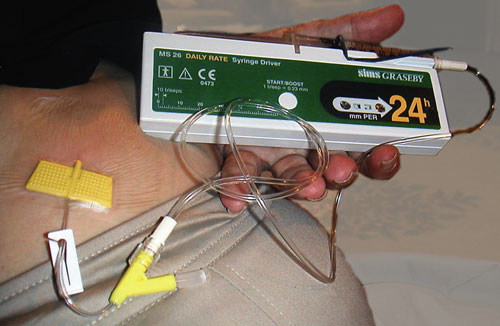
For comfort, convenience and therapeutic advantage, many palliative care patients have their medications administered with syringe drivers (Box 1).1,2 The patency of the subcutaneous site is limited principally by inflammation, with sites lasting between 12 hours and 20 days, depending on types or combinations of medications, rates of infusion and whether metal needles or plastic cannulas are used.3-5 Changing the site causes patient and carer distress, and financial costs related to nursing time and equipment replacement, especially for rural and remote community-based patients. Anecdotally, it has been suggested that site lifespan can be extended by adding a small amount of the steroid dexamethasone to the syringe driver. However, this suggestion has never been rigorously tested.
We aimed to test the effect of dexamethasone on longevity of syringe driver cannulation sites and to contribute to the development of evidence-based protocols for palliative care medicine.
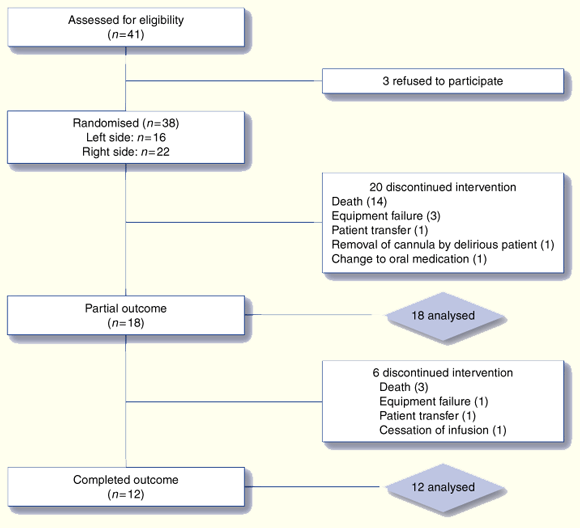
Thirty-eight patients were recruited (Box 2). All but one participant had a primary diagnosis of a solid carcinoma; the remaining patient had motor neurone disease. Patient ages ranged from 48 to 89 years with a mean of 68.6 years. There were 25 men and 13 women.
The number of drugs per patient delivered through the syringe drivers varied from one to four in each 24-hour period. Drugs infused are detailed in Box 3. No visible precipitates were observed in any of the test or control syringes.
Twenty of the patients (13 men and 7 women) were regarded as non-completers because their participation concluded before either of the syringe driver sites had broken down. Patients not completing the study did not differ significantly from those who did in age, sex, location or side of test site, or number of drugs contained within the syringe driver.
Six patients (5 men and 1 woman) partially completed the study. In these six patients, the dexamethasone-treated site remained patent although the control site had broken down when they concluded participation. Partial completers did not differ significantly from those completing in terms of age, sex, location or side of test site, or number of drugs contained within the syringe driver.
Twelve patients (7 men and 5 women) fully completed the study, in that the time for both test and control sites to break down was quantified to the nearest 4 hours.
The analyses were conducted in two phases (Box 4). The first phase included data from the 18 patients who at least partially completed the study. This analysis may underestimate effect size as six patients ended participation with their dexamethasone-treated site still intact. The second phase involved data from the 12 subjects who fully completed the study.
For the 20 patients who discontinued before either site had broken down, sites lasted from a minimum of 1 day to a maximum of 20 days, with a mean of 8.2 days (95% CI, 5.7–10.7).
For the 18 patients who partially or fully completed the study, there was a significant extension of site lifespan for dexamethasone-treated sites (Box 4; t = 3.59; df = 17; P = 0.002). Among these 18 patients, control sites lasted between 1.8 and 22.0 days and dexamethasone-treated sites lasted between 2.8 and 26.5 days.
For the 12 patients who fully completed the study, the addition of 1 mg dexamethasone to the syringe driver extended site lifespan by an average of 3.9 days (Box 4). This result is significant despite the smaller sample (t = 2.59; df = 11, P = 0.025). In this sample, control sites lasted between 1.8 and 22.0 days and dexamethasone-treated sites lasted between 3.5 and 24.0 days. No significant effects on site lifespan due to class, dose or numbers of drugs in syringe drivers were found.
Ten of the 38 participants received additional systemic dexamethasone, either orally or subcutaneously, at some stage during the study. Among the 18 who partially or fully completed the trial, seven received systemic dexamethasone, and of these, five fully completed the study. Six of the seven patients had daily doses between 2 and 16 mg subcutaneously, and one patient received 4 mg orally. In these patients, sites remained viable for longer periods: control sites lasted for 12.5 days, compared with a mean of 6.2 days in patients not receiving systemic dexamethasone. However, the outcome of interest was the difference between control and test sites.
The potentially confounding effect of systemic dexamethasone was controlled statistically by including it as a centred covariate in a regression analysis.6 The average benefit gained from the addition of 1 mg dexamethasone to the syringe driver was maintained after covariate adjustment. The mean benefit was 3.6 days increase in site life (95% CI, 1.4–5.9; P = 0.003), with no effect of systemic dexamethasone (P = 0.96).
To date, the use of dexamethasone to maintain the subcutaneous sites of syringe drivers has been piecemeal, centre-dependent and based on anecdotal evidence. Some centres use dexamethasone only with drugs that are considered irritants, such as ketamine hydrochloride, promethazine or morphine tartrate, whereas others use it only for patients whose sites are known to break down quickly. This study provides evidence (Level II according to the National Health and Medical Research Council [NHMRC] scale of weighting of evidence7) that the routine addition of 1 mg dexamethasone to the syringe drivers of palliative care patients significantly extends the lifespan of the subcutaneous sites.
Although the results suggest that the dose of 1 mg subcutaneous dexamethasone is acting locally, systemic effects may be expected with long-term use. The impact of these effects, though difficult to research in a palliative population,8 requires consideration. It would be clinically valuable to extend the findings of our study, to determine, for instance, whether 1 mg is the optimal minimal dose of dexamethasone to be added to syringe drivers, or whether topical steroid preparations could be substituted to achieve a similar benefit. The influence of other factors, such as the overall performance status of the patient, their nutritional status, pharmacological variables and prognosis, also require investigation. Such investigations would require larger sample sizes and would be appropriate for multisite studies.
The sterile subcutaneous abscess that occurred in our study was in association with the daily infusion of promethazine and dexamethasone. Although promethazine given subcutaneously is relatively irritant, it is nonetheless an effective anti-emetic and, in certain circumstances, the benefits gained from subcutaneous infusion outweigh the risks. We consider that, in this particular individual, the abscess was more likely due to the promethazine or perhaps the combination of the promethazine and dexamethasone, rather than the dexa-methasone alone.
It is difficult to conduct randomised controlled trials in palliative care patients, partly because death so frequently intervenes. Also, palliative care patients exhibit enormous variability in their condition, rate of disease progression, rapidly changing pharmacotherapeutics, previous treatment, age, socioeconomic status, past medical history and current psychological stressors. Our study indicates that well-designed rigorous clinical protocols can be applied in this difficult and vulnerable population. A strength of our study was that each individual acted as his or her own control, thus eliminating most potentially confounding clinical variables. This protocol could be adapted to investigate many common pharmacological questions in palliative clinical practice. The need for such investigations is reflected in the wide variation of pharmacological practices across palliative centres,8,9 and the lack of data on the compatibility and stability of drug mixtures in syringe drivers.10
Our findings are likely to be of par-ticular benefit to community-based patients, especially in rural and remote communities. Extending the lifespan of subcutaneous sites will decrease the frequency of medical interventions, decrease the operational management costs of maintaining patients in environments distant to palliative care service providers, and may contribute to an increased quality of life for patients and their carers.
3: Drugs infused through syringe drivers during the study
Class of drug |
Drug |
Proportion (n = 38) |
Range of maximum daily doses |
||||||||
Opioids* |
|
86.8% |
|
||||||||
|
Morphine sulfate |
34.0% |
7.5–95 mg |
||||||||
|
Hydromorphone |
34.0% |
4–80 mg |
||||||||
|
Fentanyl |
11.0% |
580–750 μg |
||||||||
|
Sufentanyl |
11.0% |
150–650 μg |
||||||||
|
Morphine tartrate |
3.0% |
120 mg |
||||||||
Anti-emetics* |
|
57.9% |
|
||||||||
|
Metoclopramide |
34.0% |
10–60 mg |
||||||||
|
Promethazine |
29.0% |
12.5–25 mg |
||||||||
Butyrophenones |
Haloperidol |
39.5% |
1–5 mg |
||||||||
Anticonvulsants |
Clonazepam |
26.3% |
1–4 mg |
||||||||
Benzodiazepines |
Midazolam |
23.7% |
2.5–20 mg |
||||||||
Antispasmodics |
Hyoscine butylbromide |
7.9% |
30–60 mg |
||||||||
Anaesthetic agents |
Ketamine hydrochloride |
7.9% |
150–300 mg |
||||||||
Somatostatin analogue |
Octreotide |
5.3% |
0.5 mg |
||||||||
* Within-category percentages do not sum to total if patients had more than one drug per category at any time over the course of the study. |
|||||||||||
4: Lifespan of sites, and difference, as a function of addition of 1 mg dexamethasone
|
Mean site lifespan (95% CI) of subcutaneous sites |
Mean increase in days of viability (95% CI) for dexamethasone site |
|||||||||
|
n |
Control |
Dexamethasone |
||||||||
Partial completion (at least one site failed) |
18 |
8.6 (5.4–11.8) |
12.3 (8.5–16.0) |
3.6 (1.5–5.8) |
|||||||
Full completion (both sites failed) |
12 |
8.6 (4.7–12.5) |
12.5 (8.1–16.9) |
3.9 (0.6–7.2) |
|||||||
Received 23 August 2003, accepted 10 February 2003
- Liz Reymond1
- Pat Treston2
- Margaret A Charles3
- Jan Bowman4
- 1 Mount Olivet Hospice, Kangaroo Point, QLD.
- 2 School of Psychology, University of Sydney, Sydney, NSW.
- 3 Mater Hospital, South Brisbane, QLD.
The authors gratefully acknowledge the patients, carers and nurses who made the study possible. Particular thanks to the research nurses Fiona Israel and Jan Paton for their professional and personal input. The study was funded by the Registrar Scholarship and Research Fund, Royal Australian College of General Practitioners. Faulding Pharmaceutical donated the dexamethasone.
None identified.
- 1. Beswick DT. Use of syringe drivers in terminal care. Pharm J 1987; 238: 656-658.
- 2. Dover SB. Syringe drivers in terminal care. BMJ 1987; 294: 553-555.
- 3. Currow D, Cooney N. Comparison of metal versus Vialon subcutaneous catheters in a palliative care setting. Palliat Med 1994; 8: 333-336.
- 4. Dawkins L, Britton D, Johnson I, et al. A randomized trial of winged Vialon cannulae and metal butterfly needles. Int J Palliat Nurs 2000; 6: 110-116.
- 5. Ross JR, Saunders Y, Cochrane M, Zeppetella G. A prospective within-patient comparison between metal butterfly needles and Teflon cannulae in sub-cutaneous infusion of drugs to terminally ill hospice patients. Palliat Med 2002; 16: 13-16.
- 6. Pocock SJ, Assmann SE, Enos LE, Kasten LE. Subgroup analysis, covariate adjustment and baseline comparisons in clinical trial reporting: current practice and problems. Stat Med 2002; 21: 2917-2930.
- 7. National Health and Medical Research Council. A guide to the development, implementation and evaluation of clinical practice guidelines. Canberra: NHMRC, 1999.
- 8. Hardy JR, Rees E, Ling J, et al. A prospective survey of the use of dexamethasone on a palliative care unit. Palliat Med 2001; 15: 3-8.
- 9. O'Doherty CA, Hall EJ, Schofield L, Zeppetella G. Drugs and syringe drivers: a survey of adult specialist palliative care practice in the United Kingdom and Eire. Palliat Med 2001; 15: 149-154.
- 10. Drummond SH, Peterson GM, Galloway JG, Keefe PA. National survey of drug use in palliative care. Palliat Med 1996; 10: 119-124.





Abstract
Objective: To assess the effect of adding 1 mg dexamethasone to syringe drivers on the viability time of subcutaneous cannulation sites in palliative care patients.
Design: Prospective, double-blind, randomised, controlled trial in which patients received half their daily infused medications plus 1 mg dexamethasone in 1 mL saline through one subcutaneous site (test site) and the other half of their medications plus 1 mL saline through another symmetrically placed site (control site).
Participants and setting: Palliative care patients from the inpatient units at two hospices, recruited between 1999 and 2002.
Main outcome measure: Difference in time that the test and control sites remained viable.
Results: 38 patients consented and were randomised. Twenty did not complete the trial because their participation in the study finished before either site broke down. Eighteen patients either partially completed (at least one site broke down) or fully completed (both sites broke down) the trial. In these 18 patients, test sites lasted 3.6 days longer than control sites (95% CI, 1.5–5.8 days; P = 0.002). Twelve patients fully completed the trial. In this group, test sites lasted 3.9 days longer than control sites (95% CI, 0.6–7.2 days; P = 0.025).
Conclusions: The addition of 1 mg dexamethasone to syringe drivers significantly extends the viability time of subcutaneous cannulation sites in palliative care patients.