Indapamide hemihydrate, a non-thiazide diuretic, is available in Australia in a 2.5 mg immediate-release (IR) formulation and a 1.5 mg sustained-release (SR) formulation. In July 2000, when it became known that supply of 500 mg chlorothiazide was to be discontinued, the 2.5 mg IR preparation of indapamide began to be promoted as a substitute for chlorothiazide. Recently, a combination product (perindopril erbumine and indapamide hemihydrate) has also been registered.
Clinically significant hyponatraemia has been attributed to indapamide as well as to the thiazide diuretics. The symptoms of hyponatraemia may begin with nausea and malaise, and progress to headache, lethargy, confusion and obtundation. Stupor, seizures and coma may occur if the serum concentration falls acutely below 120 mmol/L, or decreases rapidly.1 There is also a risk of pontine myelinolysis if the hyponatraemia is treated inappropriately.2 Hypokalaemia may result in muscle weakness and cardiac arrhythmias; symptoms generally only appear with severe hypokalaemia (less than 2.5 mmol/L).3
We undertook this study to review the reporting of indapamide-associated electrolyte disturbances in Australia. We used chlorothiazide and the combination diuretic hydrochlorothiazide (50 mg) and amiloride hydrochloride (5 mg) as comparator drugs.
The information analysed was obtained from the computerised records of Australian adverse drug reaction reports held by the Adverse Drug Reactions Advisory Committee (ADRAC). Chlorothiazide was used in Australia before the establishment of the computerised database in November 1972, and only reports received after that date were examined. The first report for the hydrochlorothiazide and amiloride combination was submitted in January 1975 and the first for indapamide in August 1984. We only considered reports in which the diuretic was the sole suspected drug.
Detailed computerised summaries of each report were retrieved, and checked against the original reports to safeguard against error during data entry. The five most commonly reported adverse reactions for each of the drugs were identified using the preferred terms from the World Health Organization Adverse Reaction Terminology.4 There was some overlap between drugs, resulting in our analysing the 11 reactions shown in Box 1. Demographic information was extracted from each report (such details were not available in only a small number of reports) and other symptoms and signs recorded in the reports were noted.
The Royal College of Pathologists of Australasia gives the following reference intervals: 5
serum sodium, 135–145 mmol/L;
serum potassium, 3.8–4.9 mmol/L; and
plasma potassium, 3.4–4.5 mmol/L.
Reports to ADRAC describing electrolyte disturbances do not always include laboratory results or reference intervals. For our primary analysis, reports describing hyponatraemia or hypokalaemia were accepted at face value.
The number of reports of each reaction as a proportion of total reports for each of the three drug products was calculated, together with the 95% confidence intervals. Two proportions were considered to be statistically significantly different if their 95% confidence intervals did not overlap.
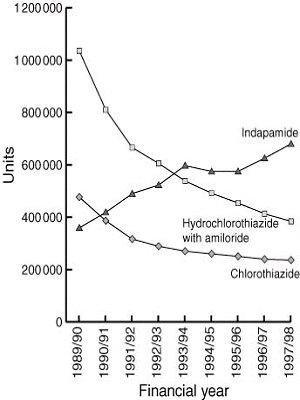
Usage data were obtained for the three diuretics (Mr Peter McManus, Pharmaceutical Benefits Branch, personal communication) by a method in which Pharmaceutical Benefits Scheme (PBS) data and results of a survey of pharmacies are combined.6 Box 2 shows that, for the financial years 1989/90 to 1997/98, use of indapamide increased at a time when use of the other two drugs decreased markedly.
As shown in Box 1, 136 reports involving chlorothiazide, 390 involving the hydrochlorothiazide and amiloride combination and 401 involving indapamide had been received to 30 September 2000. Both the hydrochlorothiazide and amiloride combination and indapamide had significantly higher proportions of reports of hyponatraemia than chlorothiazide. Indapamide had a significantly higher proportion of reports of hypokalaemia than the other two drugs.
For all three drugs, at least 80% of reports of hyponatraemia were in people aged 65 or over (chlorothiazide, 2/2 [100%]; hydrochlorothiazide and amiloride, 52/63 [83%]; indapamide, 69/84 [82%]), and at least 78% of patients were female (chlorothiazide, 2/2 [100%]; hydrochlorothiazide and amiloride, 49/63 [78%]; indapamide, 67/84 [80%]). For indapamide, most patients with hypokalaemia were aged 65 or over (53/87 [61%]), and most were female (61/87 [70%]).
For all three drugs, at least 50% of reports described patients with serum sodium concentrations ≤ 120 mmol/L. In the 84 reports with indapamide, as well as hyponatraemia symptoms mentioned included nausea (15 reports), confusion (13), vomiting (11), dizziness (9), anorexia (8), fatigue (5), malaise (4), syncope (4), somnolence (4), headache (2) and convulsions (2). One or more of these signs and symptoms was mentioned in 39 of the 84 reports.
At least 72 of the 84 reports of hyponatraemia with indapamide related to the use of 2.5 mg IR daily (dose explicitly stated in 53; stated as one tablet daily for a brand available only as 2.5 mg IR or reported before the 1.5 mg SR formulation was marketed in 19). Only two reports explicitly stated another daily dose. At least 77 of 87 reports of hypokalaemia with indapamide related to use of 2.5 mg IR tablets (dose explicitly stated in 59; stated as one tablet daily and reported when only 2.5 mg available in 18). Only one report explicitly stated another daily dose.
Of the 87 reports of hypokalaemia with indapamide, 35 patients also had hyponatraemia. A serum potassium concentration was included in 67 of the 87 reports (10 [15%], < 2.5 mmol/L; 34 [51%], 2.5 to < 3.0 mmol/L; 23 [34%], 3.0–3.5 mmol/L). Six of the 10 reports with a serum potassium concentration < 2.5 mmol/L mentioned other symptoms, including confusion, convulsions, ventricular fibrillation and syncope.
Indapamide has been implicated as a cause of severe hyponatraemia in a small number of case reports,7 and our findings support this. It is striking that there was a greater proportion of Australian reports of hyponatraemia with indapamide than with the hydrochlorothiazide and amiloride combination. Further, this proportion is many times greater than that for chlorothiazide. The reports show that indapamide-associated hyponatraemia resembles that caused by hydrochlorothiazide with amiloride in that it can be severe, and is reported predominantly in elderly women.8
Voluntary reporting systems do not provide a basis for calculating incidence or robust estimates of risk. The experience reported here relates almost solely to 2.5 mg IR indapamide tablets, the use of which has increased in recent years. Whether similar electrolyte disturbances will be observed with 1.5 mg SR indapamide tablets, subsidised on the PBS since 1 November 2001, requires further postmarketing surveillance.
Increasing use of indapamide in place of chlorothiazide will expose more patients to the risk of hyponatraemia. Changes in the conscious or mental states in patients, especially elderly women, taking indapamide should prompt timely measurement of the serum sodium concentration.
1: Numbers and proportions (with 95% confidence intervals) of commonly reported suspected adverse reactions in which the diuretic was the sole suspected drug
Number of reports |
Chlorothiazide |
Hydrochlorothiazide with amiloride |
Indapamide |
||||||||
Total |
427 |
763 |
644 |
||||||||
Diuretic sole suspected drug |
136 |
390 |
401 |
||||||||
Hyponatraemia |
2 (1.5%; 0.2%–5.2%) |
63 (16.2%; 12.6%–20.2%) |
84 (20.9%; 17.1%–25.3%) |
||||||||
Hypokalaemia |
6 (4.4%; 1.6%–9.4%) |
25 (6.4%; 4.2%–9.3%) |
87 (21.7%; 17.8%–26.1%) |
||||||||
Nausea |
5 (3.7%; 1.2%–8.4%) |
31 (7.9%; 5.5%–11.1%) |
51 (12.7%; 9.6%–16.4%) |
||||||||
Pruritus |
15 (11.0%; 6.3%–17.5%) |
28 (7.2%; 4.8%–10.2%) |
25 (6.2%; 4.1%–9.1%) |
||||||||
Fatigue |
2 (1.5%; 0.2%–5.2%) |
15 (3.8%; 2.2%–6.3%) |
28 (7.0%; 4.7%–9.9%) |
||||||||
Dizziness |
5 (3.7%; 1.2%–8.4%) |
23 (5.9%; 3.8%–8.7%) |
31 (7.7%; 5.3%–10.8%) |
||||||||
Headache |
5 (3.7%; 1.2%–8.4%) |
11 (2.8%; 1.4%–5.0%) |
29 (7.2%; 4.9%–10.2%) |
||||||||
Photosensitivity reaction |
18 (13.2%; 8.0%–20.1%) |
79 (20.3%; 16.4%–24.6%) |
8 (2.0%; 0.9%–3.9%) |
||||||||
Rash (not otherwise specified) |
10 (7.4%; 3.6%–13.1%) |
16 (4.1%; 2.4%–6.6%) |
19 (4.7%; 2.9%–7.3%) |
||||||||
Maculopapular rash |
9 (6.6%; 3.1%–12.2%) |
24 (6.2%; 4.0%–9.0%) |
23 (5.7%; 3.7%–8.5%) |
||||||||
Erythematous rash |
11 (8.1%; 4.1%–14.0%) |
14 (3.6%; 2.0%–5.9%) |
13 (3.2%; 1.7%–5.5%) |
||||||||
- Michael D Chapman
- Ross Hanrahan2
- John McEwen3
- John E Marley4
- 1 Department of General Practice, University of Adelaide, Adelaide, SA.
- 2 Adverse Drug Reactions Unit, Therapeutic Goods Administration, Woden, ACT.
- 3 The University of Newcastle, Newcastle, NSW.
Dr J Baker (Therapeutic Goods Administration; TGA) provided advice about statistical analysis. Dr I Boyd, Dr P Purcell, Ms J Robinson and Ms L Stear (Adverse Drug Reactions Unit, TGA) assisted with the review of adverse drug reaction reports. Dr R Hill and Dr M Hossain (Adverse Drug Reactions Unit, TGA) assisted with revision of the article.
- 1. Singer GG, Brenner BM. Fluid and electrolyte disturbances. In: Fauci AS, Braunwald E, et al, editors. Harrison's principles of internal medicine. 14th ed. New York: McGraw-Hill, 1998: 265-277.
- 2. Nielsen JM. Central pontine myelinolysis complicating hyponatraemia. Med J Aust 1987; 146: 492-496.
- 3. Adam WR. Fluid and electrolyte disorders. In: Whitworth JA, Lawrence JR, editors. Textbook of renal disease. 2nd ed. Edinburgh: Churchill Livingstone, 1994: 472-473.
- 4. Uppsala Monitoring Centre. Adverse reaction terminology. Uppsala: World Health Organization Collaborating Centre for International Drug Monitoring; 1999.
- 5. The Royal College of Pathologists of Australasia. Manual of use and interpretation of pathology tests. Sydney: The Royal College of Pathologists of Australasia, 1997.
- 6. Edmonds DJ, Dumbrell DM, Primrose JG, et al. Development of an Australian drug utilisation database. Pharmacoeconomics 1993; 3: 427-432.
- 7. Chan TY. Indapamide-induced severe hyponatraemia and hypokalaemia. Ann Pharmacother 1995; 29: 1124-1128.
- 8. Boyd IW, Mathew TH, Rohan AP. Hyponatraemia due to the combination of hydrochlorothiazide and amiloride (Moduretic): Australian spontaneous reports 1977–1988. Med J Aust 1990; 152: 308-309.





Abstract
Objectives: To review Australian adverse drug reaction reports describing hyponatraemia and hypokalaemia attributed to indapamide and compare the characteristics of the patients with those in Australian reports implicating two other diuretic products (hydrochlorothiazide and amiloride hydrochloride; chlorothiazide).
Design: Descriptive analysis using reports from the database of the Adverse Drug Reactions Advisory Committee (ADRAC).
Main outcome measures: Numbers of reports of hyponatraemia and hypokalaemia; proportion of such reports in total reports of adverse reactions to each drug; severity of electrolyte disturbances.
Results: Between August 1984 and September 2000, 84 Australian reports of hyponatraemia and 87 reports of hypokalaemia, in which indapamide was the sole suspected drug, were submitted to ADRAC. Most reports involved an indapamide dose of 2.5 mg daily. There was a significantly greater proportion of reports of hyponatraemia with indapamide and with the hydrochlorothiazide and amiloride combination than with chlorothiazide; hypokalaemia was significantly more common for indapamide than for the other two drugs. Of the 87 reports of hypokalaemia with indapamide, 35 patients also had hyponatraemia. For all three drugs, at least 80% of reports of hyponatraemia were in people aged 65 or over, and electrolyte disturbance was most commonly reported in elderly women.
Conclusions: Hyponatraemia and hypokalaemia have been described in 20.9% and 21.7%, respectively, of reports to ADRAC in which indapamide was the sole suspected drug. The electrolyte disturbances can be severe.