Colorectal cancer is the commonest reported internal malignancy among non-smokers in Australia,1 and is ideally suited to population-screening strategies. While there is debate about the most appropriate technique for average-risk screening,2 colonoscopic surveillance has been advocated for individuals at higher risk, such as those with a strong family history of colorectal cancer.3-6 About 10%–20% of patients with colorectal cancer will eventually have a family member with the disease, and the relative risk of colorectal cancer increases with the number of relatives with the disease and the earlier the age of onset in the affected relative(s).3,7,8
The National Health and Medical Research Council (NHMRC) of Australia has recently published guidelines for the screening and management of colorectal cancer.5 Patients are stratified into three risk categories determined by their family history of the disease, and recommendations are made regarding the appropriate surveillance and follow-up methods and the age at which surveillance should begin (Box 1).
patient demographics and referral source;
abdominal symptoms, if any;
age, relationship and number of relatives with colorectal neoplasia and/or the following malignancies: ovarian, gastric, endometrial, hepatobiliary, small-bowel, renal pelvis or ureter;
findings at colonoscopy (in particular, polyp size); and
histology of lesions identified. Advanced adenomas were defined as large adenomas (≥ 1 cm) or adenomas with villous change.
Follow-up recommendations were recorded by retrospectively reviewing endoscopy reports.
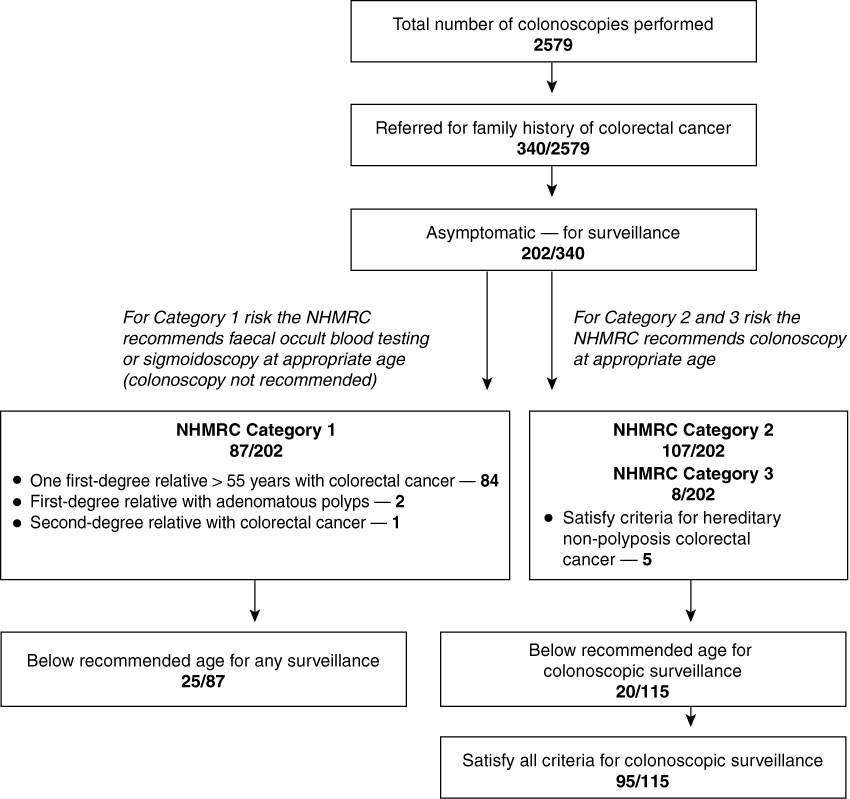
There were 2579 colonoscopies performed during the study period. A family history of colorectal cancer was the basis of referral in 340 patients and 202 of these had no abdominal symptoms. These 202 patients for surveillance comprised 83 men (41%) and 119 women (59%), with a mean age of 50 years (95% CI, 48.3–51.6 years). Only 95 of the 202 patients (47%) satisfied all criteria for colonoscopic surveillance. A further 20 (22%) satisfied the criteria but were below the recommended age for surveillance. Of the remaining 87 patients (43%), who did not satisfy the criteria, 25 were also below the recommended age for surveillance. Details of patient stratification are given in Box 2.
Colonoscopy was performed in 201 of the 202 surveillance cases (one patient declined the procedure when informed of the risks), and was complete to the caecum or terminal ileum in 199 cases. There were no complications from colonoscopy. Findings at colonoscopy, grouped by whether or not the patient satisfied the guidelines, are given in Box 3. Colorectal neoplasms, including advanced adenomatous polyps, were more common in patients whose referral concurred with the guidelines than in those whose referral did not concur (21% v 15%), but the difference was not statistically significant.
The NHMRC Guidelines for the prevention, early detection and management of colorectal cancer were published and distributed to all Australian GPs and selected specialists in 1999.5 We found that only 47% of patients referred for surveillance colonoscopy on the basis of a family history of colorectal cancer concurred fully with these guidelines. Although patients with a family history are at increased risk of colorectal cancer, the guidelines provide criteria for determining the strength of the family history, and the patient has to reach a particular age before surveillance colonoscopy is recommended. Importantly, however, the guidelines aim "to provide information on which decisions can be made, rather than dictate a specific form of treatment", and that they are "inclusive, not prescriptive". The guidelines are not rigid standards of care, and procedures that are discordant with the guidelines are not necessarily inappropriate. However, as colonoscopy is a relatively scarce resource in Western Australia, with elective waiting periods for diagnostic colonoscopy (a higher-priority investigation than surveillance) of over three months in all three adult teaching hospitals (unpublished data), a relatively prescriptive approach to interpreting the guidelines could be advocated.
The conflicting nature of several prominent guidelines, including recommendations by the American College of Gastroenterology and previous Australian guidelines.6,9 For example, the American College of Gastroenterology recommends surveillance colonoscopy for all patients with a first-degree relative with colorectal cancer.6 Similarly, several experts advocate colonoscopic screening for average-risk subjects, with some arguing that it is the most cost-effective option;2,10,11 and
That a screening test for an individual needs to be distinguished from a population-based screening program.12 Patient-driven surveillance colonoscopies (where the family history of colorectal cancer falls outside the guidelines) may be reasonable on an individual basis, especially given that the NHMRC guidelines are more restrictive than other published guidelines. Indeed, the difficulties of a fully informed discussion with patients seeking advice on screening for colorectal cancer are not trivial.13
Previous Australian studies have examined the response of GPs to various national colorectal cancer screening guidelines. Olynyk et al found that patients would often have been referred at a younger age, and at a higher screening frequency, than recommended by the guidelines.14 Another study found that GPs' views and use of colorectal cancer screening tests varied widely and were often inconsistent with available evidence.15 Our study differs from these in that we have assessed actual practice rather than questionnaire-based opinions or postulated practice. Furthermore, our patients may represent only the tip of the iceberg, being patients who have actually been referred, undergone preparation and attended for colonoscopy.
1: Summary of National Health and Medical Research Council (NHMRC) categories and surveillance recommendations for patients with a family history of colorectal cancer and follow-up recommendations for patients with adenomatous polyps
CATEGORY 1: Average or slightly above average risk (relative risk up to 2-fold)
No family history of colorectal cancer; or one first- or second-degree relative with colorectal cancer at age 55 years or older.
CATEGORY 2: Moderately increased risk (relative risk, 3–6-fold)
One first-degree relative with colorectal cancer before age 55 years (without the potentially high-risk features listed below); or two first- or second-degree relatives on the same side of the family with colorectal cancer diagnosed at any age (without potentially high-risk features).
Three or more first-or second-degree relatives on the same side of the family with colorectal cancer (suspected hereditary non-polyposis colorectal cancer [HNPCC]); or suspected familial adenomatous polyposis; or family member with identified high-risk genetic mutation.
Follow-up recommendations for patients with adenomatous polyps
Recommended interval to repeat colonoscopy:
3 months — after piecemeal removal, or excision of malignant adenoma, or incompletely excised large adenoma.
within a year — incomplete or inadequate examination (eg, in a subject with multiple adenomas).
3 years — for large adenomas (> 1 cm), high-grade dysplasia, villous change, three or more adenomas, or aged 60 years or over with a first-degree relative with colorectal cancer; and
4–6 years — in subjects without the preceding risk factors.
Received 6 August 2001, accepted 26 November 2001
- 1. Australian Institute of Health and Welfare (AIHW) and Australasian Association of Cancer Registries (AACR). Cancer in Australia 1991-1994 (with projections to 1999). Canberra: AIHW, 1998 (Cancer Series No. 7).
- 2. Bolin TD, Lapsley HM, Korman MG. Screening for colorectal cancer: what is the most cost-effective approach? Med J Aust 2001; 174: 298-301.
- 3. Winawer SJ, Fletcher RH, Miller L, et al. Colorectal cancer screening: clinical guidelines and rationale. Gastroenterology 1997; 112: 594-642.
- 4. St John DJ, Macrae FA, Young GP. Colorectal cancer screening: clinical guidelines and rationale. Gastroenterology 1997; 113: 1422 (discussion 1424).
- 5. National Health and Medical Research Council. Clinical practice guidelines for the prevention, early detection and management of colorectal cancer. Canberra: Ausinfo, 1999.
- 6. Rex DK, Johnson DA, Lieberman DA, et al. Colorectal cancer prevention 2000: screening recommendations of the American College of Gastroenterology. American College of Gastroenterology. Am J Gastroenterol 2000; 95: 868-877.
- 7. St John DJ, McDermott FT, Hopper JL, et al. Cancer risk in relatives of patients with common colorectal cancer. Ann Intern Med 1993; 118: 785-790.
- 8. Fuchs CS, Giovannucci EL, Colditz GA, et al. A prospective study of family history and the risk of colorectal cancer. N Engl J Med 1994; 331: 1669-1674.
- 9. Bolin T, Collopy B, Cowen A, et al. Colorectal cancer: prevention, diagnosis and treatment. Sydney: The Gut Foundation and Colorectal Surgical Society of Australia; 1993.
- 10. Podolsky DK. Going the distance — the case for true colorectal-cancer screening. N Engl J Med 2000; 343: 207-208.
- 11. Burt RW. Colon cancer screening. Gastroenterology 2000; 119: 837-853.
- 12. Young GP. Confusion about secondary prevention for bowel cancer: resolving issues at the front line. Med J Aust 1999; 170: 102-103.
- 13. Ward M. Preventing colon cancer: the problem with guidelines or the perils of prevention. Med J Aust 1997; 166: 201-204.
- 14. Olynyk JK, Aquilia S, Platell CF, et al. Colorectal cancer screening by general practitioners: comparison with national guidelines. Med J Aust 1998; 168: 331-334.
- 15. Sladden MJ, Ward JE. Australian general practitioners' views and use of colorectal cancer screening tests. Med J Aust 1999; 170: 110-113. <eMJA full text>





Abstract
Objectives: To assess whether referrals for surveillance colonoscopy and subsequent follow-up recommendations for patients with a family history of colorectal cancer concurred with the published National Health and Medical Research Council (NHMRC) guidelines.
Design: A prospective audit of patients with a family history of colorectal cancer referred for surveillance colonoscopy. Follow-up recommendations were assessed retrospectively.
Setting and subjects: All patients referred to a major teaching hospital for surveillance colonoscopy on the basis of a family history of colorectal cancer from 2 January 2000 – 15 April 2001.
Main outcome measures: Concurrence of referrals and recommendations with NHMRC guidelines.
Results: Of 340 patients referred because of a family history of colorectal cancer, 202 (83 men, 119 women) were asymptomatic. Their mean age was 50 years (95% CI, 48.3–51.6 years). The family history of 95 (47%) of these patients satisfied the NHMRC criteria for colonoscopic surveillance. Another 20 patients (17%) satisfied the criteria, but were referred before the recommended age to commence surveillance. Analysis by referral source showed that the proportion of referrals meeting NHMRC guidelines was higher from specialists than from general practitioners (75% v 45%), and this difference was significant. Follow-up recommendations, when made, concurred with NHMRC guidelines in 81% of cases.
Conclusions: Further education of the medical community is required to increase understanding of colorectal screening strategies and ensure appropriate resource allocation.