|
Research
Reliability of sentinel node status in predicting axillary lymph
node involvement in breast cancer
James Kollias, P Grantley Gill, Barry E Chatterton, Vivian E Hall,
Melissa A Bochner, Brendon J Coventry and Gelareh Farshid
MJA 1999; 171: 461-465
For editorial comment, see Ung & Wetzig
Abstract -
Introduction -
Methods -
Results -
Discussion -
References -
Authors' details
-
-
More articles on Oncology
|
| |
Abstract |
Objectives: To assess the reliability of
determining sentinel node status in staging regional lymph nodes in
breast cancer.
Design and setting: Prospective validation study in a
major public teaching hospital, comparing histological sentinel
node status with that of remaining axillary nodes.
Patients: 117 women who underwent sentinel node biopsy
and axillary dissection for primary breast cancer between 1995 and
1998.
Main outcome measures: Intraoperative success rate in
sentinel node identification; false negative rate; predictive
value of negative sentinel node status; overall accuracy of sentinel
node status.
Results: The sentinel node was identified at operation in
95 patients (81.2%). Tumour involvement of the sentinel node was
demonstrated in 29 of 31 women (93.5%; 95% CI, 79%-99%). Sixty-four of
the 66 women in whom the sentinel node was negative for tumour showed no
further involvement of remaining axillary nodes (standard
haematoxylin-eosin histological assessment), giving a predictive
value of negative sentinel node status of 97% (95% CI, 89%-100%). The
overall accuracy in 95 women in whom sentinel node status was compared
with axillary node status was 97.9%.
Conclusions: Histopathological examination of the
sentinel node is an accurate method of assessing axillary lymph node
status in primary breast cancer and is likely to be incorporated into
future surgical management of women with primary breast cancer.
|
| | Introduction |
Axillary lymph node status is the most important prognostic
indicator in early breast cancer, and the detection of nodal
metastases is a key factor in recommending adjuvant systemic therapy
after surgery.1,2 Surgical removal and
histopathological assessment of these nodes remains the only
accurate way of determining their involvement with tumour. Axillary
dissection also reduces the risk of regional recurrence of breast
cancer in the axilla,3 as the risk is inversely
related to the number of axillary nodes removed.4 However, axillary lymph node dissection is not without morbidity:
seroma formation, wound infection, damage to nerves, and reduced
shoulder mobility. Of particular importance is lymphoedema, which
occurs in 15% and 30% of women.5-8 As a consequence, other,
less invasive methods of assessing axillary node status have been
investigated (eg, mammography, ultrasound and colour doppler
imaging, magnetic resonance imaging [MRI] and positron emission
tomography [PET] scanning), but have yet to achieve the accuracy of
surgical staging.
Axillary node sampling -- removal of a small number of Level 1 nodes
(those below the lower border of the pectoralis minor muscle)
-- is associated with fewer complications, and has been proposed
as an alternative to complete axillary dissection for staging of the
axilla.9,10 However, its efficacy
has been questioned.11
With the advent of population-based mammographic screening
programs, there has been a dramatic decrease in tumour size and lymph
node involvement in women diagnosed with early breast
cancer.12,13 Thus, an increasing
proportion of women will undergo axillary dissection only to find
that their lymph glands are free of disease. Ideally, there should be a
method of providing accurate assessment of axillary lymph node
status without the need for axillary dissection.
The sentinel lymph node (the first draining node within a lymph node
basin) is the first to receive lymphatic drainage from a tumour site.
Selective biopsy of this node allows the detection of metastases in
clinically normal nodes with a low false negative rate, and has been
used in patients with operable breast cancer by several
groups.14-19 Their findings
indicate that the status of the sentinel node(s) can accurately
predict that of the fully dissected axilla. We report our experience
of lymphoscintigraphy, intraoperative sentinel node mapping and
sentinel node biopsy in 117 women with primary operable breast
cancer. Our aims were:
- To assess the success rate of
lymphoscintigraphy and intraoperative lymph node mapping in
identifying the sentinel node; and
- To assess the accuracy of sentinel node biopsy in staging the
axillary nodes.
|
| |
Methods |
| |
Patients |
A consecutive series of 117 women treated for primary breast cancer at
the Royal Adelaide Hospital Breast Unit between June 1995 and August
1998 entered a prospective evaluation of the technique of sentinel
lymph node biopsy in breast cancer. Ethical approval for the study was
provided by the Human Ethics Committee of the Royal Adelaide
Hospital. All women gave written informed consent to participate in
the study.
Eligibility criteria were:
- Operable primary breast
cancer (tumour, < 5 cm in diameter), detected clinically and by
imaging, and confirmed by cytology, core biopsy or open biopsy;
- Clinically impalpable axillary lymph nodes; and
- The usual surgical indications for axillary dissection (ie,
invasive, operable cancer).
Patients were excluded if their condition did not fulfil these
criteria; if they were pregnant or currently breastfeeding; if there
was a high clinical suspicion or preoperative verification of
axillary nodal involvement; or if they had metastatic breast
carcinoma or a preoperative diagnosis of ductal carcinoma-in-situ.
The women's ages ranged from 31 to 82 years (median, 60 years). Their
clinical characteristics are summarised in Table 1.
During the period of study, no eligible women refused entry to the
study.
| |
Isotope injection technique | |
The radiopharmaceutical used was 99mTc-labelled antimony
sulfide colloid ("Lymph-Flo", Royal Adelaide Hospital
Radiopharmacy). The colloid underwent filtration through a
0.2-µ sterile filter, ensuring more than 80% of the filtered
particles were smaller than 20 nm. A 32-mm, 25-gauge needle was used to
inject 40 MBq of tracer to four sites surrounding the palpable margin
of the breast lesion. If the lesion was not palpable, ultrasound
localisation was performed, and the injection was given in a similar
manner under ultrasound guidance. In the initial stages of the study,
0.5 mL of tracer was injected in each of 82 patients. For the remaining
35 women, the injected volume was increased to 4 mL in four divided
doses. In these latter women, the injection site was lightly
massaged, and they were instructed to move their arms to encourage
lymphatic movement. All radioisotope injections were given on the
morning of the day of surgery.
| |
Lymphoscintigraphy and lymph node mapping | |
After injection, serial anterior and appropriate lateral images
were obtained with a large-field-of-view gamma camera (GE XRT,
General Electric) at about 15-minute intervals until the initial
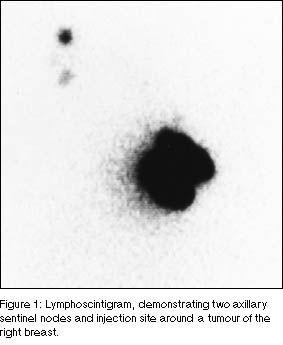
draining node (or nodes) was visualised (Figure 1). The surface projection of the sentinel node was then marked on
the skin with a radioactive marker. Orthogonal projections were made
by the established technique of "triangulation"; the marks were
joined by a straight line to indicate the base of a right-angled
triangle with the node at the apex. Body outline was marked with a
radioactive marker, or a transmission image was performed by holding
a "flood" source behind the patient.
The intraoperative probe (RMD CTC 4 with audible guidance system,
Gammasonics, Melbourne) was calibrated in the Nuclear Medicine
Department to the counts detected at the skin surface.
| |
Surgical technique | |
After completion of lymphoscintigraphy and sentinel node mapping,
the patient and hand-held gamma probe were transferred to the
operating theatre. In 66 patients, 1-2 mL of 2.5% Patent Blue V dye
(Guerbet Laboratories, France; distributed by Fauldings
Australia, Adelaide) was injected into the breast parenchyma or
subdermal fat overlying the tumour to facilitate intraoperative
identification of the sentinel node. Blue dye alone was used
in 19 patients before a gamma probe was available.
At operation, a 2-cm transverse axillary incision was made in
accordance with the planned axillary lymph node dissection, but
taking into account the preoperative skin markings indicating the
location of the sentinel node at lymphoscintigraphy. An attempt was
made to identify the node in vivo before commencement of
axillary dissection. The node was identified by its blue colour (if
dye was used) and/or by the hand-held gamma probe (in a sterile
sheath). The probe enabled detection of individual nodes with
radioactivity levels significantly greater than those of the
axillary fat (Figure 2). Sometimes more than one sentinel
node was identified. If both dye and radioisotope were used for
lymphatic mapping, the blue node corresponded to the most
radioactive node.
Once the sentinel node was removed, its activity was reassessed ex
vivo and it was sent for histological examination separately
from the main axillary nodal specimen. The axillary fat was then
examined with the gamma probe in vivo to exclude any residual
activity suggesting further sentinel nodes. The axillary skin
incision was then lengthened and a level I and II axillary lymph node
dissection was performed. The resected axillary tissue was examined
ex vivo using the probe to identify any further radioactive or
blue lymph nodes not identified during in-vivo examination.
| |
Histopathological examination | |
All specimens were examined by duty histopathologists at the
Institute of Medical and Veterinary Science. The histological
tumour features were classified according to tumour size and
grade,20 and presence or absence of
vascular invasion.21 Generally, sentinel
nodes were submitted in their entirety for histological evaluation.
Those larger than 1.5 cm were sliced before paraffin embedding. Each
node was placed in an individual cassette. At least one section of each
node was stained with haematoxylin-eosin (H&E) and examined with
light microscopy. Immunohistochemical analysis (antikeratin
antibody CAM 5.2, Becton Dickinson) was performed in H&E-stained
sections suspected of having metastatic tumour deposits.
The axillary fat was fixed in formalin and the nodes were later
isolated from the fat after clearance in Carnoy's solution. Each node
was placed in an individual cassette and larger nodes were sliced
before being embedded in paraffin. At least one H&E-stained section
of each node was examined.
| | Statistical analysis | |
A false negative sentinel node was defined as an excised sentinel
lymph node which contained no microscopically detectable tumour,
but which was associated with at least one tumour-positive node in the
remaining resected axillary tissue. The false negative rate and the
predictive value of negative sentinel node status were calculated
together with 95% confidence intervals. The kappa (κ) statistic for
paired data was used to assess the level of agreement between sentinel
node status and axillary node status.22 A score of -1
indicates perfect disagreement and + 1 indicates perfect agreement.
The corresponding z and P values were calculated. Univariate
analysis was used to assess clinical and histological factors that
predicted intraoperative sentinel node localisation. Fisher's
exact and χ2 tests were used for other
analyses between groups.
|
| |
Results |
| |
Lymphoscintigraphy | |
The sentinel node was identified on preoperative
lymphoscintigraphy in 74 of 117 women (63.2%). One sentinel node was
identified in 52 women, two were identified in 20 women, and in two
further women three and four sentinel nodes were identified,
respectively. The sentinel node was identified outside the lower
axilla in nine patients (Table 2). A significant
increase in sentinel node identification at lymphoscintigraphy was
noted after the injection of larger isotope volumes into the breast
(77% v 57%; χ2 = 4.15; P = 0.04),
while rates of intraoperative detection of the sentinel node also
increased (91% v 76%; χ2 = 3.4; P = 0.06).
| |
Intraoperative sentinel node identification | |
The sentinel node was identified in 95 patients (81.2%) at operation.
In 66 women, one sentinel node was identified, two were identified in
20 women, three in eight women, and in one four sentinel nodes were
identified. The sentinel node was identified in 35 of the 51 women in
whom radioisotope alone was used (68.6%), compared with 18 of 19 women
in whom blue dye alone was used (94.7%) and 42 of 47 women in whom both
isotope and blue dye were used (89.4%) (χ2 = 9.6; P = 0.008). Of
the clinical and histological factors assessed for predicting
intraoperative sentinel node identification, only a positive
preoperative lymphoscintigram was significant (χ2 = 28.7; P <
0.001) (Table 3).
| |
Predictive value of sentinel node(s) | |
In 95 patients in whom the sentinel node was identified, 31 had
metastatic tumour involvement of axillary nodes (32.6%). Tumour
involvement of the sentinel node was demonstrated in 29 of these 31
women (93.5%; 95% CI, 79%-99%), giving a false negative rate of 6.5%.
The sentinel node was the only positive node in 13 of 31 women (41.9%).
Of 66 women with a negative sentinel node, 64 had no tumour involvement
in the remainder of the axillary nodes (by standard H&E histological
assessment), giving a predictive value of negative sentinel node
status of 97% (95% CI, 89%-100%). The overall accuracy in 95 patients
in whom sentinel node status was compared with axillary node status
was 97.9% (κ, 0.95; z = 9.3; P < 0.001) (Table 4). Of the 22 women in whom the sentinel
node was not identified at operation, six had nodal metastases on
histological examination of the dissected axillary nodes.
|
| |
Discussion |
The concept of the sentinel lymph node is based on the premise that the
first lymph node to receive lymphatic drainage from a tumour site
should be the first site of lymphatic spread; that "skip metastases"
do not occur; and that the absence of tumour metastases in the sentinel
node implies the absence of lymph node metastases in the entire
lymphatic basin. This concept was first described in penile
carcinoma in 197723 and was later studied in
patients with cutaneous melanoma.24 Previous detailed
pathological studies of axillary nodes in women with breast cancer
have demonstrated a skip metastasis rate of less than 5%.25,26 Our results confirm that the status of the sentinel lymph node(s)
predicts the overall axillary lymph node status with a high degree of
accuracy, and can thus be used to limit the morbidity associated with
axillary surgery. More importantly, the predictive value of a
tumour-free sentinel node was 97%. As such, women identified with a
sentinel node free of metastatic tumour can be reassured that further
axillary lymph node involvement is highly unlikely. Other studies of
sentinel node biopsy in breast cancer (using blue dye and radioactive
isotope techniques) have shown sentinel node status to accurately
determine axillary lymph node status in more than 95% of
women.14-19
We still need to deal with the problem that 3% of patients exhibited
tumour-positive axillary nodes when the biopsied sentinel node was
negative. The optimal method of pathological assessment of the
sentinel node remains unresolved and was not addressed in our study.
This issue was discussed at the Adelaide Workshop on Sentinel Node
Biopsy in Breast Cancer27 and is the subject of
further studies by one of us (G F). Giuliano et al28 have found
that immunohistochemical studies of sentinel nodes showed
micrometastases in an additional 11% of women whose sentinel node was
tumour negative on light microscopy. However, similar assessment of
two women with false negative results in our study did not reveal
metastases.
The implications of micrometastases detected by sensitive
immunohistochemical and polymerase chain reaction (PCR)
techniques for multidisciplinary care are unknown. They are
currently being investigated in trials in the United States (Merrick
Ross, Associate Professor of Surgical Oncology, M D Anderson
Hospital, Texas, USA, personal communication). Until the answers to
this question are available, a large UK trial (ALMANAC) is assessing
sentinel node status by conventional microscopy (R Mansell,
Professor of Surgery, Cardiff University, UK, personal
communication), as this is the current method on which treatment
planning is based. These uncertainties emphasise the need for
Australian studies to incorporate detailed protocols for pathology
assessment of the sentinel node. The prognostic implications of a
false negative sentinel node are uncertain, but should be compared
with the considerable physical morbidity associated with axillary
dissection in lymph node negative women. There is a definite
error rate in routine pathological assessment of axillary
dissection specimens which may underestimate metastatic disease by
11%-30%,27,29 while unselective
sampling of the axilla fails to remove involved nodes in many
women.11 The false negative rate
must ultimately be minimised by maximal detection of the sentinel
node by scintigraphy, careful operative technique and optimal
pathological assessment, which requires an experienced
multidisciplinary team.
The concomitant intraoperative use of both blue dye and radionuclide
methods for lymphatic mapping was particularly useful for sentinel
node biopsy. Preoperative lymphoscintigraphy permits
identification of the sentinel node and subsequent planning of the
site of skin incision. Several radiolabelled colloids are currently
in use around the world, but the recent workshop in
Adelaide27 identified antimony
colloids as having excellent properties for lymphoscintigraphy.
This is the only agent available for this purpose in Australia and is
able to visualise sentinel nodes in the internal mammary chain as well
as in the axillary node group. The blue dye technique facilitated
visualisation of the sentinel node at the time of surgery and was
supplemented by the use of an intraoperative gamma probe. In all
patients in whom both blue dye and radionuclide were used, the blue
node corresponded to the "hot" node previously identified on
lymphoscintigraphy and identified intraoperatively with the
hand-held gamma probe. Furthermore, the identification of a
sentinel node at preoperative lymphoscintigraphy was the only
factor significantly associated with the intraoperative
identification of the sentinel node. Lymphoscintigraphy also
demonstrates the number and location of potential sentinel nodes
requiring biopsy. The initial rate of preoperative identification
of the sentinel node by lymphoscintigraphy in our series was lower
than that in published reports. However, this was overcome by
increasing the volume of the isotope injection and presumably
increasing tissue oncotic pressure, lymphatic uptake and drainage.
The importance of isotope volume in achieving successful
scintigraphic identification of the sentinel node has also been
suggested by others.30
Sentinel lymph node mapping and biopsy are likely to be incorporated
into clinical practice, provided they can be successfully performed
in most patients, and it can be shown that women with negative sentinel
nodes who undergo no further treatment to the axilla are not adversely
compromised in terms of disease-free and overall survival. This will
be best established by randomised controlled studies comparing
sentinel node biopsy with standard axillary surgical management. In
addition, these studies should address the implied assumption of
lower short and long term morbidity associated with this procedure,
the optimal methods of pathological assessment, and allow analysis
and comparison with clinicopathological variables in predicting
sentinel node status. Studies are currently being undertaken in
Europe, the United Kingdom and the United States and it is hoped that
Australian women can soon participate in similar trials in
Australia.
|
| |
References |
- Carter CL, Allen C, Henson DE. Relation of tumour size, lymph nodes
status and survival in 24,740 breast cancer cases. Cancer
1989; 63: 181-187.
-
Fisher ER, Anderson S, Redmond C, Fisher B. Pathologic findings
from the National Surgical Adjuvant Breast Project Protocol B-06: 10
year pathological and clinical prognostic discriminants.
Cancer 1993; 71: 2507-2514.
-
Fisher D, Woolmark N, Bauer M, et al. The accuracy of clinical nodes
staging and of limited axillary dissection as a determinant of
histological nodal status in carcinoma of the breast. Surg
Gynecol Obstet 1991; 152: 765-772.
-
Axellsson CK, Mouridsen HT, Zedeler K. Axillary dissection of
Level I and II lymph nodes is important in breast cancer
classification: The Danish Breast Cancer Cooperative Group (DBCG).
Eur J Cancer 1992; 28: 1415-1418.
-
Kissin MW, Querci-Della-Rovere G, Easton D, Westbury G. Risk of
lymphoedema following the treatment of breast cancer. Br J
Surg 1986; 73: 580-584.
-
Aitken RJ, Gayes MN, Rodger A, et al. Arm morbidity within a trial of
mastectomy and either node sample with selective radiotherapy or
axillary clearance. Br J Surg 1989; 76: 568-571.
-
Larson D, Weinstein M, Goldburg I, et al. Oedema of the arm as a
function of the extent of axillary surgery in patients with Stage 1-2
carcinoma of the breast treated with primary radiotherapy. Int J
Radiat Oncol Biol Phys 1986; 12: 1575-1582.
-
Liljegren G, Holmburg L. Arm morbidity after sector resection and
axillary dissection with or without postoperative radiotherapy in
breast cancer. Stage 1: Results from a randomised trial. Uppsala
Orebro Breast Cancer Study Group. Eur J Cancer 1997; 33:
193-199.
-
Steel RJC, Forrest APM, Gibson T, et al. The efficacy of lower
axillary sampling in obtaining lymph node status in breast cancer: a
controlled randomised trial. Br J Surg 1985; 72: 368-369.
-
Dixon JM, Dillon P, Anderson TJ, Chetty U. Axillary node sampling
in breast cancer: an assessment of its efficacy. Breast 1998;
7: 206-208.
-
Kissin MW, Thompson PH, Price AB, et al. The inadequacy of axillary
sampling in breast cancer. Lancet 1982; 1: 1210-1212.
-
Tabar L, Fagerberg G, Duffy SW, et al. Update of the Swedish
two-county program of mammographic screening for breast cancer.
Radiol Clin North Am 1992; 30: 187-210.
-
Cady B, Stone MD, Schuler JG, et al. The new era in breast cancer:
invasion, size and lymph node involvement dramatically decreased as
a result of mammographic screening. Arch Surg 1996; 131:
301-308.
-
Giuliano AE, Kirgan DM, Guenther JM, Morton DL. Lymphatic mapping
and sentinel lymphadenectomy in breast cancer. Ann Surg
1994; 220: 391-401.
-
Albertini JJ, Lyman GH, Cox C, et al. Lymphatic mapping and
sentinel node biopsy in the patient with breast cancer. JAMA
1996; 276: 1818-1822.
-
Veronesi U, Paganelli G, Galimberti V, et al. Sentinel node biopsy
to avoid axillary dissection in breast cancer with clinically
negative lymph nodes. Lancet 1997; 349: 1864-1867.
-
Borgstein PJ, Pijpers R, Comans EF, et al. Sentinel lymph node
biopsy in breast cancer: guidelines and pitfalls of
lymphoscintigraphy and gamma probe detection. J Am Coll Surg
1998; 186: 275-283.
-
Cox CE, Pendas S, Cox JM, et al. Guidelines for sentinel node biopsy
and lymphatic mapping of patients with breast cancer. Ann
Surg 1998; 227: 645-653.
-
O'Hea BJ, Hill ADK, El-Shirbiny AM, et al. Sentinel lymph node
biopsy in breast cancer: initial experience at Memorial
Sloan-Kettering Cancer Center. J Am Coll Surg 1998; 186:
423-427.
-
Elston CW, Ellis IO. Pathological prognostic factors in breast
cancer. The value of histological grade in breast cancer: experience
from a large study with long-term follow-up. Histopathology
1991; 19: 403-410.
-
Pinder SE, Ellis IO, Galea M, et al. Pathological prognostic
factors in breast cancer. Vascular invasion: relationship with
recurrence and survival in a large study with long-term follow-up.
Histopathology 1994; 24: 41-47.
-
Fliess JL. Statistical methods for rates and proportions. 2nd
edition. New York, NY: John Wiley and Sons, 1981.
-
Cabanas RM. An approach for the treatment of penile carcinoma.
Cancer 1977; 39: 456-466.
-
Morton DL, Wen D-R, Wong JH, et al. Technical details of
intraoperative lymphatic mapping for early stage melanoma. Arch
Surg 1992; 127: 392-399.
-
Berg JW. The significance of axillary node levels in the study of
breast cancer. Cancer 1955; 8: 776-778.
-
Veronesi U, Rilke F, Luimi A, et al. Distribution of axillary node
metastases by level of invasion: an analysis of 539 cases.
Cancer 1987; 59: 682-687.
-
Kollias J, Gill PG, Chatterton B, et al. Sentinel node biopsy in
breast cancer: recommendations for surgeons, pathologists,
nuclear physicians and radiologists in Australia and New Zealand.
Aust N Z J Surg 1999. In press.
-
Giuliano AE, Dale PS, Turner RR, et al. Improved axillary staging
of breast cancer with sentinel lymphadenectomy. Ann Surg
1995; 222: 387-399.
-
Hainsworth PJ, Tjandra JJ, Stillwell RG, et al. Detection and
significance of occult metastases in node negative breast cancer.
Br J Surg 1993; 80: 459-463.
-
Krag DN, Ashikaga T, Harlow SH, Weaver DL. Development of sentinel
node targeting technique in breast cancer patients. Breast J
1998; 4: 67-74.
(Received 22 Apr, accepted 9 Sep, 1999)
|
| | Authors' details |
Royal Adelaide Hospital and Women's Health Centre, Adelaide, SA.
James Kollias, MB BS, FRACS, Staff Surgeon,
Breast-Endocrine and Surgical Oncology Unit.
P Grantley Gill, FRACS, MD, Head, Breast-Endocrine and
Surgical Oncology Unit; and Associate Professor, University of
Adelaide.
Barry E Chatterton, MB BS, FRACP, Director, Department of
Nuclear Medicine.
Vivian E Hall, MB BS, FRACR, Radiologist, Department of
Radiology.
Melissa A Bochner, MB BS, FRACS, Senior Registrar,
Breast-Endocrine and Surgical Oncology Unit.
Brendon J Coventry, FRACS, PhD, Senior Surgeon,
Breast-Endocrine and Surgical Oncology Unit; and Senior Lecturer,
University of Adelaide.
Department of Tissue Pathology, Institute of Medical and Veterinary
Science, Adelaide, SA.
Gelareh Farshid, MB BS, FRCPA, Senior Lecturer, University
of Adelaide.
Reprints will not be available from the authors.
Correspondence:
Associate Professor P G Gill, Breast-Endocrine Surgical Oncology
Unit, Royal Adelaide Hospital, North Terrace, Adelaide, SA 5000.
cbatesbrownswordATmedicine.adelaide.edu.au
|
| |  | | Back to text | | 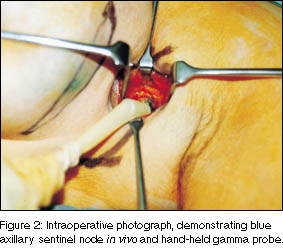 | | Back to text | | 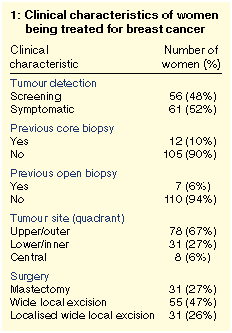 | | Back to text | |  | | Back to text | | |
3: Clinical and histological features predicting success in sentinel node identification at operation | | Variable | No. of women | Sentinel node identified (%) | χ2
(P) |
| | Age (years) | | < 50 | 33 | 30 (91%) | 2.84 | | > 50 | 84 | 65 (77%) | (0.09) | | Tumour site (quadrant) | | Upper/outer | 86 | 69 (80%) | 0.2 | | Lower/inner | 31 | 26 (84%) | (0.66) | | Tumour detection | | Screening | 56 | 42 (75%) | 2.7 | | Symptomatic | 61 | 53 (87%) | (0.1) | | Previous core biopsy | | Yes | 12 | 10 (83%) | 0.04 | | No | 105 | 85 (79%) | (0.84) | | Previous open biopsy | | Yes | 110 | 89 (81%) | 0.1 | | No | 7 | 6 (86%) | (0.75) | | Scintiscan result | | Positive | 74 | 71 (96%) | 28.7 | | Negative | 43 | 24 (56%) | (< 0.001) | | Operation | | Mastectomy | 31 | 24 (77%) | 1.23 | | Wide local excision | 55 | 47 (85%) | (0.54) | | Localised wide local excision | 31 | 24 (77%) | | Tumour size* | | < 2cm | 79 | 64 (81%) | 0.03 | | ≥ 2cm | 36 | 29 (81%) | (0.95) | | Tumour grade* | | 1 | 27 | 21 (78%) | 2.4 | | 2 | 53 | 46 (87%) | (0.31) | | 3 | 35 | 26 (74%) | | Lymphatic/vascular invasion | | Negative | 101 | 83 (82%) | 0.44 | | Positive | 16 | 12 (75%) | (0.5) | | Lymph node status | | Negative | 80 | 64 (80%) | 0.2 | | Positive | 37 | 31 (84%) | (0.6) |
| |
* Excludes two cases of ductal carcinoma-in-situ, diagnosed after excision.
|
| | Back to text | |  | | Back to text |
|









