|
Research
Outcome of a screening program for vancomycin-resistant
enterococci in a hospital in Victoria
M Lindsay Grayson, Elizabeth A Grabsch, Paul D R Johnson, Dianne Olden,
Melissa Aberline, H Y Li, Geoffrey Hogg, Marguerite Abbott and
Peter G Kerr
MJA 1999; 171: 133-136
See also Ferguson, Robertson et al & Collignon
Abstract -
Introduction -
Methods -
Results -
Discussion -
Acknowledgements -
References -
Authors' details
-
-
More articles on Infectious diseases and parasitology
|
Abstract |
Objective: To screen for faecal colonisation with
vancomycin-resistant enterococci (VRE) among potentially at-risk
patients.
Design: Infection control screening program.
Setting: Monash Medical Centre (a tertiary care
hospital), Melbourne, Victoria, in the seven months from June
1997.
Patients: Patients in the Renal, Oncology and Intensive
Care (ICU) Units.
Main outcome measures: Presence of VRE in a rectal swab or
faecal specimen taken at admission and at regular intervals during
inpatient stay; presence of vancomycin-resistance genes
(vanA, vanB and vanC) assessed by polymerase
chain reaction (PCR); genetic clonality of isolates assessed by
pulsed-field gel electrophoresis (PFGE).
Results: 574 patients (356 renal, 134 ICU and 84 oncology)
were screened; 12 were colonised with VRE -- nine renal inpatients,
two having peritoneal dialysis or in-centre haemodialysis, and one
ICU patient. Nine isolates were Enterococcus faecalis
(seven positive for vanB and two negative for all three
resistance genes) and three were Enterococcus faecium (all
positive for vanB). Eight were high-level gentamicin
resistant. PFGE suggested genetic clonality between the index
isolate and five other isolates from renal patients. No specific
clinical practice was associated with VRE colonisation. Attempts to
clear rectal carriage with oral ampicillin/amoxycillin or
bacitracin were of limited success. Although antibiotic
prescribing in the Renal Unit was generally consistent with defined
protocols, use of vancomycin and third-generation cephalosporins
has been further restricted.
Conclusions: Renal inpatients in our institution appear
most at risk of VRE colonisation (4.6% overall) and therefore of VRE
infection. Routine screening, especially of potentially high-risk
patients, should be considered in major Australian hospitals.
|
| | Introduction |
Vancomycin-resistant enterococci (VRE) were first reported in the
United Kingdom, Europe and the United States in 1988,1-6 and in
Australia in 1994.7 In the United States, VRE
have become common and potentially fatal nosocomial pathogens; they
account for 14% of enterococcal bacteraemias in intensive care
patients, while VRE bacteraemia has an attributable mortality of
30%-46%.4,8 In Australia, cases of VRE
infection have now been reported from hospitals in all States and
Territories except the Australian Capital Territory and
Tasmania.9,10 In May 1997, a patient receiving renal haemodialysis at Monash
Medical Centre, Melbourne, developed a VRE urinary tract infection
(see Box 1). This was the first identified case of VRE infection at this
institution. As the patient had had close contact with other patients
and had suffered diarrhoea, we were concerned about a significant
nosocomial outbreak of VRE infection. Clinical infection is almost
always associated with faecal colonisation with this organism. We
therefore assessed the extent of faecal VRE colonisation among renal
and other high-risk patients by active screening, and isolated
infected or colonised patients. We describe the results of this
screening program and our infection control measures, and highlight
some of the clinical issues.
|
| |
Methods |
An infection control screening program for faecal colonisation with
VRE was implemented in the Renal, Oncology and Intensive Care Units at
Monash Medical Centre, Melbourne, Victoria, for the seven months
from June 1997. These patients were chosen as experience
(local, US and European) suggested they were at greatest risk of VRE
infection.4-6,8,10 | |
Patients |
In the Renal Unit, screening was planned for all patients receiving
care in the renal ward and in-centre haemodialysis or continuous
ambulatory peritoneal dialysis and, when possible, for dialysis
patients managed at home. In the Oncology and Intensive Care Units
(ICU), screening was planned for an arbitrary number of 100
consecutive patients admitted to each unit.
All inpatients were screened on admission and discharge. In the renal
ward this was later modified to on admission and a regular day of the
week (Tuesday). Patients managed by the in-centre haemodialysis
unit were screened every three months, and other outpatients were
screened at least once.
| |
Screening |
Rectal swabs were obtained using standard cotton-alginate-tipped
sterile swabs from all patients except neutropenic oncology
patients. In these, a perianal swab or faecal specimen was
substituted for a rectal swab because of their increased risk of
septicaemia after rectal trauma.13 Specimens were plated on
media specifically selective for vancomycin-resistant
enterococci (bile esculin azide agar with 6 µg/mL vancomycin)
and cultured for up to 72 hours. Esculin-positive isolates with
possible resistance to vancomycin were identified, and single
colonies of each morphology type were assessed to identify
Enterococcus faecalis and Enterococcus faecium
(the pathogenic enterococcal species most commonly associated with
vancomycin resistance). Assessment included Gram stain, tests of
motility and pigment production, the pyrrolidonyl arylamidase test
(Murex Diagnostics Ltd, Dartford, UK) and streptococcal latex
grouping.14 | |
VRE assessment |
VRE isolates were tested further for antibiotic susceptibility.
Minimum inhibitory concentrations (MICs) were determined for
vancomycin, teicoplanin and ampicillin and high-level gentamicin
(MIC > 500 µg/mL) using the E test (AB Biodisk, Dalvagen,
Sweden). Production of β-lactamase was assesssed by nitrocephin
disc (Becton Dickinson Microbiology Systems, Cockeysville, MD,
USA).
Enterococcal species was confirmed and presence of
vancomycin-resistance genes vanA, vanB or vanC was
assessed by polymerase chain reaction (PCR) genetic probe using a
modification of techniques described previously.10,15 Genetic
similarity (ie, potential clonality) of VRE isolates was assessed by
pulsed-field gel electrophoresis (PFGE) using a method modified
after Miranda et al.16
Factors that were potentially associated with VRE colonisation were
assessed retrospectively.6,8 Statistical analyses
were by χ2 or t test.
|
| |
Results |
| |
Patients |
Screening was undertaken on 574 patients -- 356 renal, 134 ICU and 84
oncology patients. Renal patients comprised:
- 194 of 238 inpatients in the renal ward (82%);
- 66 of 82 peritoneal dialysis and in-centre haemodialysis patients
(80%);
- 94 outpatients (mostly satellite and home haemodialysis
patients); and
- 2 of 180 renal transplantation patients.
In general, consecutive oncology and ICU patients were screened;
none refused screening. In the Oncology Unit, the target number (100
patients) was not attained because of a protocol lapse.
| |
VRE colonisation | |
Faecal colonisation with VRE was found in 12 patients, including the
index patient -- 11 renal patients (3% of renal patients tested) and
one ICU patient (0.7% of ICU patients tested). Their characteristics
are shown in Box 2. No VRE colonisation was found in oncology patients.
The 11 renal patients with VRE colonisation comprised nine
inpatients (9/194 [5%]) and two having peritoneal dialysis or
in-centre haemodialysis (2/66 [3%]). No non-dialysis renal
outpatients were colonised. VRE colonisation was found on the
initial rectal swab for seven patients and after a series of negative
cultures for the remaining five patients.
| |
Characteristics of VRE isolates | |
Characteristics of the 12 VRE isolates are shown in Box 2. Nine of the 12
were E. faecalis -- seven testing positive for vanB,
including the index isolate (vancomycin MICs, 12-32 µg/mL),
and two testing negative for vanA, vanB and
vanC (vancomycin MICs, 6 and 8 µg/mL, respectively).
The other three isolates were E. faecium -- all testing
positive for vanB (vancomycin MICs, 16, 64 and > 256
µg/mL, respectively). All isolates were susceptible to
teicoplanin. While all E. faecalis isolates were
susceptible to ampicillin, all E. faecium isolates were
resistant (MICs > 256 µg/mL). Six of the nine E.
faecalis isolates and two of the three E. faecium
isolates had high-level resistance to gentamicin. None of the 12
produced detectable β-lactamase.
Eleven isolates were assessed by PFGE. Results are shown in
the Figure. Six of the seven vanB E. faecalis isolates,
including the index isolate, appeared genetically similar (lanes
4-8 and index isolate in lane 9). Three of these similar isolates, plus
one dissimilar vanB E. faecalis isolate (not shown), were
isolated from patients who had been nursed together in a four-bed
area. One of these patients (with vanB E. faecalis) had been
nursed with the index patient six months before screening positive.
The three E. faecium isolates and the non-ABC E. faecalis
isolate (lanes 1-3 and 10, respectively) showed a variety of
electrophoretic patterns on PFGE, suggesting they were genetically
dissimilar from each other.
| |
Factors potentially associated with VRE | |
Among the 12 patients with VRE colonisation, nine had received
vancomycin in the previous month, four of whom had also received a
third-generation cephalosporin (ceftriaxone). Information on
previous antibiotics was not available for patients without VRE
colonisation.
Among the six renal patients found to have VRE colonisation on their
initial swab (including the index case), five had been inpatients
during the previous three months, compared with 127 of the 350
non-colonised patients (36%). Mean duration of preswab inpatient
stay for these six colonised patients was 17.5 days (range, 0-62),
compared with 2.6 days (range, 0-43) for non-colonised patients
(P < 0.001).
| |
VRE control measures | |
All patients identified with faecal VRE colonisation were nursed in a
single room according to infection control guidelines.11 In January
1998, an eight-bed VRE isolation facility was established.
Continued, less rigorous screening of renal inpatients
identified four new cases of VRE colonisation in the following three
months (not described here), but these VRE strains were dissimilar on
PFGE to the previous 12 strains.
Antibiotic usage patterns were also reviewed. Most antibiotic use in
the Renal Unit was found to be consistent with the unit's protocols.
These were amended to further restrict use of third-generation
cephalosporins and glycopeptides to specific situations, such as
nosocomial pneumonia and serious staphylococcal infections.
In seven patients, an attempt was made to "clear" faecal VRE
colonisation with either ampicillin or amoxycillin (variable
doses, depending on renal function) or oral bacitracin (25
000 units four times a day for 7-14 days).17,18 In four of these
patients, follow-up rectal swabs were taken, and in two (one taking
ampicillin and one bacitracin) VRE was no longer detected 18 and 13
days, respectively, after therapy.
At completion of the study, nine of the 12 patients with VRE
colonisation had died, although, other than the index patient, none
had developed VRE infection.
|
| |
Discussion |
A screening program introduced at Monash Medical Centre after
identification of VRE infection in a renal patient found faecal VRE
colonisation in another 10 renal patients (3% of renal patients
overall) and one ICU patient (0.7%). Isolates from six of the renal
patients, including the index patient, were genetically similar and
probably clonal. No patients except the index patient developed VRE
infection.
VRE is now a major nosocomial pathogen in many US and European centres,
but until recently relatively few clinical VRE infections had been
reported in Australia.6,8-10 To our knowledge, this
is the first Australian report of a systematic screening program for
VRE among potentially at-risk patients.
Our results were consistent with those of previous studies, which
suggested that 10-20 patients are likely to have faecal colonisation
for every case of clinical VRE infection.6,8,19,20 Although risk
factors for VRE infection have been identified by US and European
investigators,6,8the factors
associated with VRE colonisation are less clear and may vary
depending on the epidemiology of VRE in different countries.
Antibiotic prescribing patterns, nosocomial transmission and use
of antibiotics (eg, avoparcin) in the veterinary industry
appear of varying importance in different regions.8,10
Our study did not allow valid assessment of all factors potentially
associated with VRE colonisation in our patients. Nevertheless, the
fact that renal patients with VRE colonisation spent significantly
more days in hospital in the previous three months than patients
without colonisation raises the possibility that the hospital
environment or illness-related factors influenced the likelihood
of VRE colonisation.
Resistance to vancomycin among enterococci is generally due to
presence of one of four resistance genes -- vanA,
vanB, vanC and vanD. These genes result in
synthesis of abnormal precursors in the peptidoglycan layer of the
bacterial cell wall, thereby reducing the affinity with which
vancomycin binds to this target site.8VanA is associated
phenotypically with resistance to vancomycin (MIC > 64
µg/mL) and teicoplanin (MIC > 16 µg/mL), and is the
most common genotype found in Europe and some centres in the
US.8VanB is
associated with medium-level resistance to vancomycin (MIC > 4
µg/mL) but susceptibility to teicoplanin. Consistent with
our findings, it is the predominant genotype noted in
Australia.9,10VanC is
associated with naturally occurring low-level resistance to
vancomycin and susceptibility to teicoplanin among less pathogenic
enterococcal species, while vanD, which is phenotypically
similar, has been occasionally noted in some E. faecium
isolates.8,21,22 It is possible that
our two non-ABC E. faecalis isolates contain vanD,
but we are currently unable to test for this gene.
Presence of faecal VRE colonisation among 5% of renal inpatients at
our institution (3% of renal patients overall) was higher than
expected, but suggested that nosocomial transmission of VRE was not
yet a widespread problem. Nevertheless, our PFGE data suggested that
six of the seven vanB E. faecalis strains were clonal, raising
infection control issues for the Renal Unit. As reported
previously,17,18 we found attempts to
clear faecal VRE carriage with antibiotic therapy were unsuccessful
and not worthwhile. The screening program and establishment of VRE
isolation facilities to readily cohort and barrier-nurse patients
with VRE colonisation appeared to assist in limiting nosocomial VRE
transmission, while continuing to provide medical care for patients
in a compassionate manner.
The incidence of faecal VRE colonisation that we found among
high-risk patients at our institution suggests that routine
screening for faecal VRE colonisation should now be considered by
other similar Australian hospitals.
|
Acknowledgements | |
We wish to acknowledge the contribution of the nursing staff of the
Renal, Oncology and Intensive Care Units and the Outpatient
Department in obtaining rectal cultures.
|
| |
References |
- Leclercq R, Derlot E, Duval J, Courvalin P. Plasmid-mediated
resistance to vancomycin and teicoplanin in Enterococcus
faecium. N Engl J Med 1988; 319: 157-161.
-
Uttley AHC, Collins CH, Naidoo J, George RC. Vancomycin-resistant
enterococci. Lancet 1988; 1: 57-58.
-
Clark NC, Cooksey RC, Hill BC, et al. Characterization of
glycopeptide-resistant enterococci from U.S. hospitals.
Antimicrob Agents Chemother 1993; 37: 2311-2317.
-
Centers for Disease Control. Nosocomial enterococci resistant to
vancomycin -- United States, 1989-1993. MMWR Morb Mortal Wkly
Rep 1993; 42: 597-599.
-
Frieden TR, Munsiff SS, Low DE, et al. Emergence of
vancomycin-resistant enterococci in New York City. Lancet
1993; 342: 76-79.
-
Boyce JM. Vancomycin-resistant enterococcus. Detection,
epidemiology, and control measures. Infect Dis Clin North Am
1997; 11: 367-384.
-
Kamarulzaman A, Tosolini FA, Boquest AL, et al. Vancomycin
resistant Enterococcus faecium infection in a liver
transplant recipient [abstract]. Aust N Z J Med 1995; 25: 560.
-
Eliopoulos GM. Vancomycin-resistant enterococci. Mechanism and
clinical relevance. Infect Dis Clin North Am 1997; 11:
851-865.
-
Bell J, Turnidge J, Coombs G, O'Brien F. Emergence and epidemiology
of vancomycin-resistant enterococci in Australia. Commun Dis
Intell 1998; 22: 249-252.
-
Bell JM, Paton JC, Turnidge J. Emergence of vancomycin-resistant
enterococci in Australia: phenotypic and genotypic
characteristics of isolates. J Clin Microbiol 1998; 36:
2187-2190.
-
Standing Committee on Infection Control (SCIC), Department of
Human Services, Victoria. Guidelines for the management of patients
with confirmed vancomycin-resistant enterococci (VRE)
infection/colonisation. Melbourne: Department of Human Services,
1996.
-
Moellering RC Jr. The Garrod lecture. The enterococcus: a classic
example of the impact of antimicrobial resistance on therapeutic
options. J Antimicrob Chemother 1991; 28: 1-12.
-
Weinstein JW, Tallapragada S, Farrel P, Dembry L-M. Comparison of
rectal and perirectal swabs for detection of colonisation with
vancomycin-resistant enterococci. J Clin Microbiol 1996;
34: 210-212.
-
Facklam RR, Sahm DF. Enterococcus. In: Murray PR, Baron EJ,
Pfaller MA, et al, editors. Manual of clinical microbiology. 6th ed.
Washington: ASM Press, 1995: 308-314.
-
Dutka-Malen A, Evers S, Courvalin P. Detection of glycopeptide
resistance genotypes and identification to the species level of
clinically relevant enterococci by PCR. J Clin Microbiol
1995; 33: 24-27.
-
Miranda AG, Singh KV, Murray BE. A fingerprinting of
Enterococcus faecium by pulsed-field gel electrophoresis
may be a useful epidemiologic tool. J Clin Microbiol 1991; 29:
2752-2757.
-
O'Donovan CA, Fan-Havard P, Tecson-Tumang FT, et al. Enteric
eradication of vancomycin-resistant Enterococcus faecium
with oral bacitracin. Diagn Microbiol Infect Dis 1994; 18:
105-109.
-
Chia JKS, Nakata MM, Park SS, et al. Use of bacitracin therapy for
infection due to vancomycin-resistant Enterococcus
faecium. Clin Infect Dis 1995; 21: 1520.
-
Jordens JZ, Bates J, Griffith DT. Faecal carriage and nosocomial
spread of vancomycin-resistant Enterococcus faecium. J
Antmicrob Chemother 1994; 34: 515-528.
-
Montecalvo MA, deLaencastre H, Carraher M, et al. Natural history
of colonization with vancomycin-resistant Enterococcus
faecium. Infect Control Hosp Epidemiol 1995; 16:
680-685.
-
Leclercq R, Courvalin P. Resistance to glycopeptides in
enterococci. Clin Infect Dis 1997; 24: 545-556.
-
Perichon B, Reynolds P, Courvalin P. VanD-type
glycopeptide-resistant Enterococcus faecium BM4339.
Antimicrob Agents Chemother 1997; 41: 2016-2018.
(Received 15 Dec 1998, accepted 23 Apr 1999)
|
| | Authors' details |
Infectious Disease and Clinical Epidemiology Department, Monash
Medical Centre, Melbourne, VIC.
M Lindsay Grayson, MD, FRACP, Director, and Professor of
Epidemiology and Preventive Medicine, Monash University,
Melbourne, VIC;
Elizabeth A Grabsch, BSc, GradDipClinEpid,
Infection Control Scientist;
Paul D R Johnson, PhD, FRACP,
Infectious Disease Physician, and Senior Lecturer,
Microbiology Department, Monash University, Melbourne, VIC;
Dianne Olden, PhD, Research Scientist.
Infection Control Unit, Monash Medical Centre, Melbourne, VIC.
Melissa Aberline, RN, BSc, Infection Control Nurse.
Microbiological Diagnostic Unit, Melbourne University,
Melbourne, VIC.
H Y Li, MMed, Scientist;
Geoffrey Hogg, FRACP, FRCPA,
Director.
Nephrology Department, Monash Medical Centre, Melbourne, VIC.
Marguerite Abbott, RN, BAppSci, Nurse Director;
Peter G
Kerr, PhD, FRACP, Deputy Director.
Reprints will not be available from the authors.
Correspondence:
Professor M L Grayson, Infectious Diseases and Clinical
Epidemiology Department, Monash Medical Centre, 246 Clayton Road,
Clayton, VIC 3168.
Email: Lindsay.GraysonATmed.monash.edu.au
|
| | 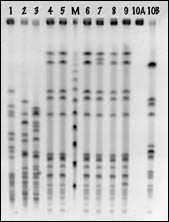 | Pulsed-field gel electrophoresis of vancomycin-resistant enterococcal isolates.
Lanes 1-3: E. faecium (vanB) isolates from renal patients.
Lanes 4-9: E. faecalis (vanB) isolates from renal patients, including index isolate (lane 9).
Lane M: Molecular weight markers.
Lane 10: E. faecalis (non-ABC) isolate from renal patient.
|
| | Back to text | | |
1: Case history of the index patient
In May 1997, a 26-year-old woman presented to Monash Medical Centre with a fever and urinary tract infection caused by vancomycin-resistant enterococci (VRE). She had faecal colonisation with the same VRE strain. She had endstage renal failure requiring in-centre haemodialysis three times a week.
Ten days later, the patient presented in status epilepticus with faecal incontinence that led to substantial faecal contamination of the Emergency Department and the in-centre Haemodialysis Unit. Appropriate cleaning protocols11 were implemented in each area, and limited environmental cultures suggested no contamination.
Five months later, the patient developed symptomatic VRE bacteraemia after surgical insertion of a femoral Goretex arteriovenous fistula. The VRE isolate was phenotypically identical to the initial urinary isolate. As it had high-level gentamicin resistance, she was treated with continuous-infusion ampicillin, continuing for 10 weeks because of the possibility of graft sepsis.12 The patient continued to show faecal VRE colonisation until her death (not related to VRE) in September 1998.
|
| | Back to text |
|





